Details of the Drug
General Information of Drug (ID: DMVO4ED)
| Drug Name |
Ethoxzolamide
|
||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Synonyms |
ethoxzolamide; Ethoxyzolamide; Ethoxazolamide; Ethamide; Etoxzolamide; Cardrase; 6-Ethoxy-2-benzothiazolesulfonamide; 452-35-7; Glaucotensil; Diuretic C; 6-Ethoxy-1,3-benzothiazole-2-sulfonamide; Mingoral; 2-Benzothiazolesulfonamide, 6-ethoxy-; Redupresin; 6-Ethoxybenzo[d]thiazole-2-sulfonamide; 6-Ethoxyzolamide; 6-Ethoxybenzothiazole-2-sulfonamide; 6-Ethoxybenzothiazole-2-sulphonamide; U-4191; NSC 10679; UNII-Z52H4811WX; C9H10N2O3S2; HSDB 3268; CHEMBL18; EINECS 207-199-5; BRN 0212240; MLS000028637; AI3-50805; Cardrase; EZL; Cardrase (TN); Ethoxzolamide (EZA); 6-(ethyloxy)-1,3-benzothiazole-2-sulfonamide; 6-Ethoxy-benzothiazole-2-sulfonic acid amide; 6-ethoxy-1,3-benzothiazole-2-sulfonamide
|
||||||||||||||||||||||||||
| Indication |
|
||||||||||||||||||||||||||
| Therapeutic Class |
Diuretics
|
||||||||||||||||||||||||||
| Affected Organisms |
Humans and other mammals
|
||||||||||||||||||||||||||
| Drug Type |
Small molecular drug
|
||||||||||||||||||||||||||
| Structure |
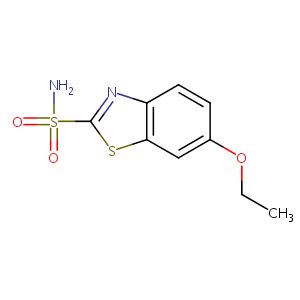 |
||||||||||||||||||||||||||
| 3D MOL | 2D MOL | ||||||||||||||||||||||||||
| #Ro5 Violations (Lipinski): 0 | Molecular Weight (mw) | 258.3 | |||||||||||||||||||||||||
| Logarithm of the Partition Coefficient (xlogp) | 2 | ||||||||||||||||||||||||||
| Rotatable Bond Count (rotbonds) | 3 | ||||||||||||||||||||||||||
| Hydrogen Bond Donor Count (hbonddonor) | 1 | ||||||||||||||||||||||||||
| Hydrogen Bond Acceptor Count (hbondacc) | 6 | ||||||||||||||||||||||||||
| ADMET Property | |||||||||||||||||||||||||||
| Chemical Identifiers |
|
||||||||||||||||||||||||||
| Cross-matching ID | |||||||||||||||||||||||||||
| Repurposed Drugs (RPD) | Click to Jump to the Detailed RPD Information of This Drug | ||||||||||||||||||||||||||
Molecular Interaction Atlas of This Drug
 Drug Therapeutic Target (DTT) |
|
|||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Molecular Interaction Atlas (MIA) | ||||||||||||||||||||||||||||||||
Molecular Expression Atlas of This Drug
| The Studied Disease | Glaucoma/ocular hypertension | |||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ICD Disease Classification | 9C61 | |||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
| Molecular Expression Atlas (MEA) | ||||||||||||||||||||||||||||||
References
