Details of the Drug
General Information of Drug (ID: DMYDW6J)
| Drug Name |
PGD2
|
||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Synonyms |
Prostaglandin D2; prostaglandin D2; 11-Dehydroprostaglandin F2-alpha; 41598-07-6; BRN 2170623; UNII-RXY07S6CZ2; 11-Dehydroprostaglandin F2alpha; RXY07S6CZ2; CHEBI:15555; (5Z,13E,15S)-9alpha,15-Dihydroxy-11-oxoprosta-5,13-dienoate; (5Z,13E)-(15S)-9alpha,15-Dihydroxy-11-oxoprosta-5,13-dienoate; 9S,15S-dihydroxy-11-oxo-5Z,13E-prostadienoic acid; [3H]PGD2; (5E,13E)-9,15-DIHYDROXY-11-OXOPROSTA-5,13-DIEN-1-OIC ACID; Prosta-5,13-dien-1-oic acid, 9,15-dihydroxy-11-oxo-, (5Z,9-alpha,13E,15S)-; 11-dehydroprostaglandin F2-alpha
|
||||||||||||||||||||||
| Indication |
|
||||||||||||||||||||||
| Drug Type |
Small molecular drug
|
||||||||||||||||||||||
| Structure |
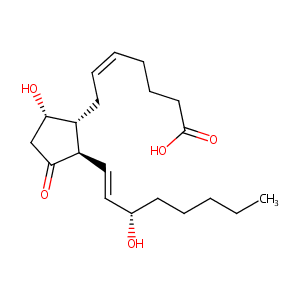 |
||||||||||||||||||||||
| 3D MOL is unavailable | 2D MOL | ||||||||||||||||||||||
| #Ro5 Violations (Lipinski): 1 | Molecular Weight (mw) | 352.5 | |||||||||||||||||||||
| Logarithm of the Partition Coefficient (xlogp) | 2.6 | ||||||||||||||||||||||
| Rotatable Bond Count (rotbonds) | 12 | ||||||||||||||||||||||
| Hydrogen Bond Donor Count (hbonddonor) | 3 | ||||||||||||||||||||||
| Hydrogen Bond Acceptor Count (hbondacc) | 5 | ||||||||||||||||||||||
| Chemical Identifiers |
|
||||||||||||||||||||||
| Cross-matching ID | |||||||||||||||||||||||
Molecular Interaction Atlas of This Drug
 Drug Therapeutic Target (DTT) |
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
 Drug Off-Target (DOT) |
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Molecular Interaction Atlas (MIA) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Molecular Expression Atlas of This Drug
| The Studied Disease | Discovery agent | |||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ICD Disease Classification | N.A. | |||||||||||||||||||||||
|
||||||||||||||||||||||||
| Molecular Expression Atlas (MEA) | ||||||||||||||||||||||||
References
