| 1 |
ClinicalTrials.gov (NCT00738972) Left Ventricular Hypertrophy Reduction With Statins in Hypertensives Patients (MK0653A-168)
|
| 2 |
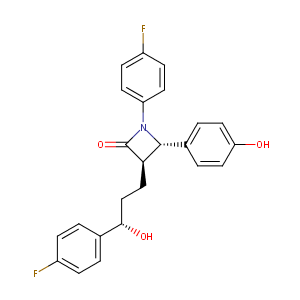
Ezetimibe FDA Label
|
| 3 |
URL: http://www.guidetopharmacology.org Nucleic Acids Res. 2015 Oct 12. pii: gkv1037. The IUPHAR/BPS Guide to PHARMACOLOGY in 2016: towards curated quantitative interactions between 1300 protein targets and 6000 ligands. (Ligand id: 6816).
|
| 4 |
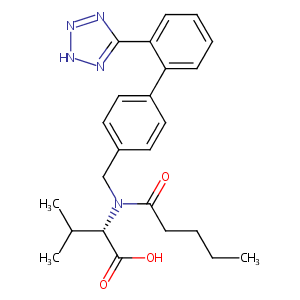
Valsartan FDA Label
|
| 5 |
URL: http://www.guidetopharmacology.org Nucleic Acids Res. 2015 Oct 12. pii: gkv1037. The IUPHAR/BPS Guide to PHARMACOLOGY in 2016: towards curated quantitative interactions between 1300 protein targets and 6000 ligands. (Ligand id: 3937).
|
| 6 |
Valsartan for Prevention of Acute Respiratory Distress Syndrome in Hospitalized Patients With SARS-COV-2 (COVID-19) Infection Disease
|
| 7 |
Identification of human UDP-glucuronosyltransferase enzyme(s) responsible for the glucuronidation of ezetimibe (Zetia). Drug Metab Dispos. 2004 Mar;32(3):314-20.
|
| 8 |
Update on patented cholesterol absorption inhibitors. Expert Opin Ther Pat. 2009 Aug;19(8):1083-107.
|
| 9 |
Intestinal expression of P-glycoprotein (ABCB1), multidrug resistance associated protein 2 (ABCC2), and uridine diphosphate-glucuronosyltransferase 1A1 predicts the disposition and modulates the effects of the cholesterol absorption inhibitor ezetimibe in humans. Clin Pharmacol Ther. 2006 Mar;79(3):206-17.
|
| 10 |
Pharmacokinetic and pharmacodynamic interactions between the immunosuppressant sirolimus and the lipid-lowering drug ezetimibe in healthy volunteers. Clin Pharmacol Ther. 2010 Jun;87(6):663-7.
|
| 11 |
Complex pharmacokinetic behavior of ezetimibe depends on abcc2, abcc3, and abcg2. Drug Metab Dispos. 2009 Aug;37(8):1698-702.
|
| 12 |
A LC-MS/MS method to quantify the novel cholesterol lowering drug ezetimibe in human serum, urine and feces in healthy subjects genotyped for SLCO1B1. J Chromatogr B Analyt Technol Biomed Life Sci. 2006 Jan 2;830(1):143-50.
|
| 13 |
Early identification of clinically relevant drug interactions with the human bile salt export pump (BSEP/ABCB11). Toxicol Sci. 2013 Dec;136(2):328-43.
|
| 14 |
Ezetimibe: a review of its metabolism, pharmacokinetics and drug interactions. Clin Pharmacokinet. 2005;44(5):467-94.
|
| 15 |
Drug interactions between the immunosuppressant tacrolimus and the cholesterol absorption inhibitor ezetimibe in healthy volunteers. Clin Pharmacol Ther. 2011 Apr;89(4):524-8.
|
| 16 |
Effects of ezetimibe and/or simvastatin on LDL receptor protein expression and on LDL receptor and HMG-CoA reductase gene expression: a randomized trial in healthy men. Atherosclerosis. 2008 May;198(1):198-207.
|
| 17 |
Carotenoid transport is decreased and expression of the lipid transporters SR-BI, NPC1L1, and ABCA1 is downregulated in Caco-2 cells treated with ezetimibe. J Nutr. 2005 Oct;135(10):2305-12. doi: 10.1093/jn/135.10.2305.
|
| 18 |
Compound heterozygosity for two non-synonymous polymorphisms in NPC1L1 in a non-responder to ezetimibe. Clin Genet. 2005 Feb;67(2):175-7. doi: 10.1111/j.1399-0004.2004.00388.x.
|
| 19 |
Valsartan improves adipose tissue function in humans with impaired glucose metabolism: a randomized placebo-controlled double-blind trial. PLoS One. 2012;7(6):e39930. doi: 10.1371/journal.pone.0039930. Epub 2012 Jun 29.
|
| 20 |
Radioligand binding assays: application of [(125)I]angiotensin II receptor binding. Methods Mol Biol. 2009;552:131-41.
|
| 21 |
Involvement of transporters in the hepatic uptake and biliary excretion of valsartan, a selective antagonist of the angiotensin II AT1-receptor, in humans. Drug Metab Dispos. 2006 Jul;34(7):1247-54.
|
| 22 |
Regulation of Organic Anion Transporting Polypeptides (OATP) 1B1- and OATP1B3-Mediated Transport: An Updated Review in the Context of OATP-Mediated Drug-Drug Interactions. Int J Mol Sci. 2018 Mar 14;19(3). pii: E855.
|
| 23 |
High-affinity interaction of sartans with H+/peptide transporters. Drug Metab Dispos. 2009 Jan;37(1):143-9.
|
| 24 |
Prediction of the overall renal tubular secretion and hepatic clearance of anionic drugs and a renal drug-drug interaction involving organic anion transporter 3 in humans by in vitro uptake experiments. Drug Metab Dispos. 2011 Jun;39(6):1031-8.
|
| 25 |
In vitro inhibition screening of human hepatic P450 enzymes by five angiotensin-II receptor antagonists. Eur J Clin Pharmacol. 2000 May;56(2):135-40.
|
| 26 |
Uric acid accumulation in the kidney triggers mast cell degranulation and aggravates renal oxidative stress. Toxicology. 2023 Jan 1;483:153387. doi: 10.1016/j.tox.2022.153387. Epub 2022 Dec 1.
|
| 27 |
Angiotensin II receptor blockade in normotensive subjects: A direct comparison of three AT1 receptor antagonists. Hypertension. 1999 Mar;33(3):850-5. doi: 10.1161/01.hyp.33.3.850.
|
| 28 |
[The relationship between the gene polymorphism of TGF-beta1 and early renal injury in patients with essential hypertension, and the effect of the gene polymorphism of TGF- beta1 on the individual treatment with valsartan]. Zhonghua Yi Xue Yi Chuan Xue Za Zhi. 2007 Aug;24(4):428-31.
|
| 29 |
C-reactive protein in heart failure: prognostic value and the effect of valsartan. Circulation. 2005 Sep 6;112(10):1428-34. doi: 10.1161/CIRCULATIONAHA.104.508465. Epub 2005 Aug 29.
|
| 30 |
Is renoprotection by angiotensin receptor blocker dependent on blood pressure?: the Saitama Medical School, Albuminuria Reduction in Diabetics with Valsartan (STAR) study. Hypertens Res. 2007 Jun;30(6):529-33. doi: 10.1291/hypres.30.529.
|
| 31 |
Effects of valsartan on circulating brain natriuretic peptide and norepinephrine in symptomatic chronic heart failure: the Valsartan Heart Failure Trial (Val-HeFT). Circulation. 2002 Nov 5;106(19):2454-8. doi: 10.1161/01.cir.0000036747.68104.ac.
|
| 32 |
Antiviral effect of Bosentan and Valsartan during coxsackievirus B3 infection of human endothelial cells. J Gen Virol. 2010 Aug;91(Pt 8):1959-1970. doi: 10.1099/vir.0.020065-0. Epub 2010 Apr 14.
|
| 33 |
Comparing antihypertensive effect and plasma ciclosporin concentration between amlodipine and valsartan regimens in hypertensive renal transplant patients receiving ciclosporin therapy. Am J Cardiovasc Drugs. 2011 Dec 1;11(6):401-9. doi: 10.2165/11593800-000000000-00000.
|
|
|
|
|
|
|