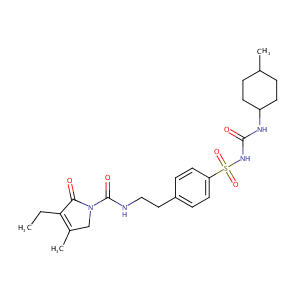
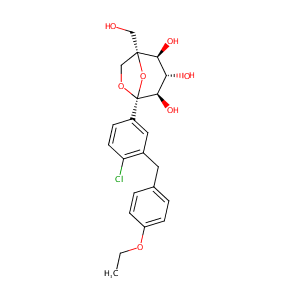
| 1 |
ClinicalTrials.gov (NCT02226003) Efficacy and Safety of Ertugliflozin (MK-8835/PF-04971729) With Sitagliptin in the Treatment of Participants With Type 2 Diabetes Mellitus (T2DM) With Inadequate Glycemic Control on Diet and Exercise (MK-8835-017). U.S. National Institutes of Health.
|
| 2 |
URL: http://www.guidetopharmacology.org Nucleic Acids Res. 2015 Oct 12. pii: gkv1037. The IUPHAR/BPS Guide to PHARMACOLOGY in 2016: towards curated quantitative interactions between 1300 protein targets and 6000 ligands. (Ligand id: 6820).
|
| 3 |
2017 FDA drug approvals.Nat Rev Drug Discov. 2018 Feb;17(2):81-85.
|
| 4 |
Mechanism of disopyramide-induced hypoglycaemia in a patient with Type 2 diabetes. Diabet Med. 2009 Jan;26(1):76-8.
|
| 5 |
Early identification of clinically relevant drug interactions with the human bile salt export pump (BSEP/ABCB11). Toxicol Sci. 2013 Dec;136(2):328-43.
|
| 6 |
Effect of CYP2C9 genetic polymorphisms on the efficacy and pharmacokinetics of glimepiride in subjects with type 2 diabetes. Diabetes Res Clin Pract. 2006 May;72(2):148-54.
|
| 7 |
Interference with bile salt export pump function is a susceptibility factor for human liver injury in drug development. Toxicol Sci. 2010 Dec; 118(2):485-500.
|
| 8 |
Initro inhibition of AKR1Cs by sulphonylureas and the structural basis. Chem Biol Interact. 2015 Oct 5;240:310-5.
|
| 9 |
Screening of a chemical library reveals novel PXR-activating pharmacologic compounds. Toxicol Lett. 2015 Jan 5;232(1):193-202. doi: 10.1016/j.toxlet.2014.10.009. Epub 2014 Oct 16.
|
| 10 |
Effects of prolonged in vitro exposure to sulphonylureas on the function and survival of human islets. J Diabetes Complications. 2005 Jan-Feb;19(1):60-4. doi: 10.1016/j.jdiacomp.2004.05.001.
|
| 11 |
A potential role of calpains in sulfonylureas (SUs) -mediated death of human pancreatic cancer cells (1.2B4). Toxicol In Vitro. 2021 Jun;73:105128. doi: 10.1016/j.tiv.2021.105128. Epub 2021 Feb 27.
|
| 12 |
Systems pharmacological analysis of drugs inducing stevens-johnson syndrome and toxic epidermal necrolysis. Chem Res Toxicol. 2015 May 18;28(5):927-34. doi: 10.1021/tx5005248. Epub 2015 Apr 3.
|
| 13 |
ADReCS-Target: target profiles for aiding drug safety research and application. Nucleic Acids Res. 2018 Jan 4;46(D1):D911-D917. doi: 10.1093/nar/gkx899.
|
| 14 |
Dose-ranging efficacy and safety study of ertugliflozin, a sodium-glucose co-transporter 2 inhibitor, in patients with type 2 diabetes on a background of metformin.Diabetes Obes Metab.2015 Jun;17(6):591-8.
|
| 15 |
The effect of renal impairment on the pharmacokinetics and pharmacodynamics of ertugliflozin in subjects with type 2 diabetes mellitus. J Clin Pharmacol. 2017 Nov;57(11):1432-1443.
|
| 16 |
Discovery of a clinical candidate from the structurally unique dioxa-bicyclo[3.2.1]octane class of sodium-dependent glucose cotransporter 2 inhibitors. J Med Chem. 2011 Apr 28;54(8):2952-60. doi: 10.1021/jm200049r. Epub 2011 Mar 30.
|
| 17 |
ClinicalTrials.gov (NCT02036515) Safety and Efficacy of Ertugliflozin in the Treatment of Participants With Type 2 Diabetes Mellitus Who Have Inadequate Glycemic Control on Metformin and Sitagliptin (MK-8835-006; VERTIS SITA2)
|
|
|
|
|
|
|