Details of the Drug
General Information of Drug (ID: DM8JRH0)
| Drug Name |
Cangrelor
|
|||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Synonyms |
Cangrelor; 163706-06-7; Kengreal; UNII-6AQ1Y404U7; AR-C69931XX; CHEMBL334966; 6AQ1Y404U7; kengrexal; 5'-O-[({[dichloro(phosphono)methyl](hydroxy)phosphoryl}oxy)(hydroxy)phosphoryl]-N-[2-(methylsulfanyl)ethyl]-2-[(3,3,3-trifluoropropyl)sulfanyl]adenosine; 5'-Adenylicacid,N-[2-(methylthio)ethyl]-2-[(3,3,3-trifluoropropyl)thio]-,anhydridewithP,P'-(dichloromethylene)bis[phosphonicacid](1:1)
|
|||||||||||||||||||||||||
| Indication |
|
|||||||||||||||||||||||||
| Affected Organisms |
Humans and other mammals
|
|||||||||||||||||||||||||
| ATC Code | ||||||||||||||||||||||||||
| Drug Type |
Small molecular drug
|
|||||||||||||||||||||||||
| Structure |
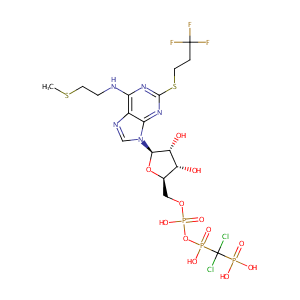 |
|||||||||||||||||||||||||
| 3D MOL | 2D MOL | |||||||||||||||||||||||||
| #Ro5 Violations (Lipinski): 4 | Molecular Weight (mw) | 776.4 | ||||||||||||||||||||||||
| Logarithm of the Partition Coefficient (xlogp) | -1 | |||||||||||||||||||||||||
| Rotatable Bond Count (rotbonds) | 15 | |||||||||||||||||||||||||
| Hydrogen Bond Donor Count (hbonddonor) | 7 | |||||||||||||||||||||||||
| Hydrogen Bond Acceptor Count (hbondacc) | 21 | |||||||||||||||||||||||||
| ADMET Property |
|
|||||||||||||||||||||||||
| Adverse Drug Reaction (ADR) |
|
|||||||||||||||||||||||||
| Chemical Identifiers |
|
|||||||||||||||||||||||||
| Cross-matching ID | ||||||||||||||||||||||||||
| Combinatorial Drugs (CBD) | Click to Jump to the Detailed CBD Information of This Drug | |||||||||||||||||||||||||
Molecular Interaction Atlas of This Drug
 Drug Therapeutic Target (DTT) |
|
||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
 Drug Off-Target (DOT) |
|
||||||||||||||||||||||||||
| Molecular Interaction Atlas (MIA) | |||||||||||||||||||||||||||
Drug-Drug Interaction (DDI) Information of This Drug
|
Coadministration of a Drug Treating the Same Disease as Cangrelor
Coadministration of a Drug Treating the Disease Different from Cangrelor (Comorbidity)
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Drug Inactive Ingredient(s) (DIG) and Formulation(s) of This Drug
References
| 1 | URL: http://www.guidetopharmacology.org Nucleic Acids Res. 2015 Oct 12. pii: gkv1037. The IUPHAR/BPS Guide to PHARMACOLOGY in 2016: towards curated quantitative interactions between 1300 protein targets and 6000 ligands. (Ligand id: 1776). | ||||
|---|---|---|---|---|---|
| 2 | Trend Analysis of a Database of Intravenous Pharmacokinetic Parameters in Humans for 1352 Drug Compounds | ||||
| 3 | ADReCS-Target: target profiles for aiding drug safety research and application. Nucleic Acids Res. 2018 Jan 4;46(D1):D911-D917. doi: 10.1093/nar/gkx899. | ||||
| 4 | URL: http://www.guidetopharmacology.org Nucleic Acids Res. 2015 Oct 12. pii: gkv1037. The IUPHAR/BPS Guide to PHARMACOLOGY in 2016: towards curated quantitative interactions between 1300 protein targets and 6000 ligands. (Target id: 328). | ||||
| 5 | Cerner Multum, Inc. "UK Summary of Product Characteristics.". | ||||
| 6 | Alderman CP, Moritz CK, Ben-Tovim DI "Abnormal platelet aggregation associated with fluoxetine therapy." Ann Pharmacother 26 (1992): 1517-9. [PMID: 1482806] | ||||
| 7 | Product Information. Ofev (nintedanib). Boehringer Ingelheim, Ridgefield, CT. | ||||
| 8 | Bodiford AB, Kessler FO, Fermo JD, Ragucci KR "Elevated international normalized ratio with the consumption of grapefruit and use of warfarin." SAGE Open Med Case Rep 0 (2013): 1-3. [PMID: 27489634] | ||||
| 9 | Heck AM, DeWitt BA, Lukes AL "Potential interactions between alternative therapies and warfarin." Am J Health Syst Pharm 57 (2000): 1221-7 quiz 1228-30. [PMID: 10902065] | ||||
| 10 | Caruso V, Iacoviello L, Di Castelnuovo A, et.al "Thrombotic complications in childhood acute lymphoblastic leukemia: a meta-analysis of 17 prospective studies comprising 1752 pediatric patients." Blood 108 (2006): 2216-22. [PMID: 16804111] | ||||
| 11 | Product Information. Calquence (acalabrutinib). Astra-Zeneca Pharmaceuticals, Wilmington, DE. | ||||
| 12 | Agencia Espaola de Medicamentos y Productos Sanitarios Healthcare "Centro de informacion online de medicamentos de la AEMPS - CIMA.". | ||||
| 13 | Product Information. Iclusig (ponatinib). Ariad Pharmaceuticals Inc, Cambridge, MA. | ||||
| 14 | Cerner Multum, Inc. "Australian Product Information.". | ||||
| 15 | Product Information. Acular (ketorolac). Allergan Inc, Irvine, CA. | ||||
| 16 | Abebe W "Herbal medication: potential for adverse interactions with analgesic drugs." J Clin Pharm Ther 27 (2002): 391-401. [PMID: 12472978] | ||||
| 17 | Product Information. Cometriq (cabozantinib). Exelixis Inc, S San Francisco, CA. | ||||
