Details of the Drug
General Information of Drug (ID: DMCMD93)
| Drug Name |
SSR149415
|
||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Synonyms |
Nelivaptan; SSR 149415; 439687-69-1; UNII-3TY57MQ4OA; SR-149415; SSR-149415; 3TY57MQ4OA; CHEMBL582857; Nelivaptan [INN]; DSSTox_CID_27358; DSSTox_RID_82294; DSSTox_GSID_47358; GTPL2202; DTXSID7047358; SCHEMBL14517029; MolPort-042-665-640; Tox21_300240; BDBM50299343; ZINC42833251; DB12643; SSR-149,415; NCGC00254222-01; NCGC00247960-01; CAS-439687-69-1; (2S,4R)-1-[(3R)-5-chloro-1-(2,4-dimethoxyphenyl)sulfonyl-3-(2-methoxyphenyl)-2-oxoindol-3-yl]-4-hydroxy-N,N-dimethylpyrrolidine-2-carboxamide; Nelivaptan; 1-(5-chloro-1-((2,4-dimethoxyphenyl)sulfonyl)-3-(2-methoxyphenyl)-2-oxo-2,3-dihydro-1H-indol-3-yl)-4-hydroxy-N,N-dimethyl-2-pyrrolidinecarboxamide
|
||||||||||||||||||||||||||
| Indication |
|
||||||||||||||||||||||||||
| Drug Type |
Small molecular drug
|
||||||||||||||||||||||||||
| Structure |
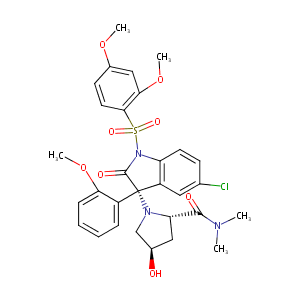 |
||||||||||||||||||||||||||
| 3D MOL | 2D MOL | ||||||||||||||||||||||||||
| #Ro5 Violations (Lipinski): 1 | Molecular Weight (mw) | 630.1 | |||||||||||||||||||||||||
| Logarithm of the Partition Coefficient (xlogp) | 3.4 | ||||||||||||||||||||||||||
| Rotatable Bond Count (rotbonds) | 8 | ||||||||||||||||||||||||||
| Hydrogen Bond Donor Count (hbonddonor) | 1 | ||||||||||||||||||||||||||
| Hydrogen Bond Acceptor Count (hbondacc) | 9 | ||||||||||||||||||||||||||
| Chemical Identifiers |
|
||||||||||||||||||||||||||
| Cross-matching ID | |||||||||||||||||||||||||||
| Repurposed Drugs (RPD) | Click to Jump to the Detailed RPD Information of This Drug | ||||||||||||||||||||||||||
Molecular Interaction Atlas of This Drug
 Drug Therapeutic Target (DTT) |
|
|||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
 Drug Off-Target (DOT) |
|
|||||||||||||||||||||||||||||||||||||||||
| Molecular Interaction Atlas (MIA) | ||||||||||||||||||||||||||||||||||||||||||
Molecular Expression Atlas of This Drug
| The Studied Disease | Anxiety disorder | |||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ICD Disease Classification | 6B00-6B0Z | |||||||||||||||||||||||
|
||||||||||||||||||||||||
| Molecular Expression Atlas (MEA) | ||||||||||||||||||||||||
References
