| 1 |
Recurrent recessive mutation in deoxyguanosine kinase causes idiopathic noncirrhotic portal hypertension.Hepatology. 2016 Jun;63(6):1977-86. doi: 10.1002/hep.28499. Epub 2016 Mar 31.
|
| 2 |
URL: http://www.guidetopharmacology.org Nucleic Acids Res. 2015 Oct 12. pii: gkv1037. The IUPHAR/BPS Guide to PHARMACOLOGY in 2016: towards curated quantitative interactions between 1300 protein targets and 6000 ligands. (Ligand id: 4812).
|
| 3 |
PAN-811 inhibits oxidative stress-induced cell death of human Alzheimer's disease-derived and age-matched olfactory neuroepithelial cells via suppression of intracellular reactive oxygen species. J Alzheimers Dis. 2009;17(3):611-9.
|
| 4 |
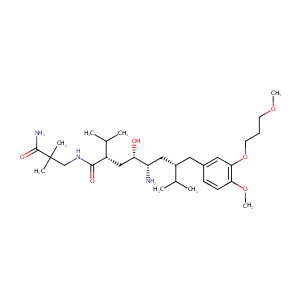
Comparative effects of aliskiren-based and ramipril-based therapy on the renin system during long-term (6 months) treatment and withdrawal in patie... J Renin Angiotensin Aldosterone Syst. 2009 Sep;10(3):157-67.
|
| 5 |
Effects of the inhibition of intestinal P-glycoprotein on aliskiren pharmacokinetics in cynomolgus monkeys. Biopharm Drug Dispos. 2015 Jan;36(1):15-33.
|
| 6 |
Orange and apple juice greatly reduce the plasma concentrations of the OATP2B1 substrate aliskiren. Br J Clin Pharmacol. 2011 May;71(5):718-26.
|
| 7 |
Clinical pharmacokinetics and pharmacodynamics of aliskiren. Clin Pharmacokinet. 2008;47(8):515-31.
|
| 8 |
Association of CYP1A1 and CYP1B1 inhibition in in vitro assays with drug-induced liver injury. J Toxicol Sci. 2021;46(4):167-176. doi: 10.2131/jts.46.167.
|
| 9 |
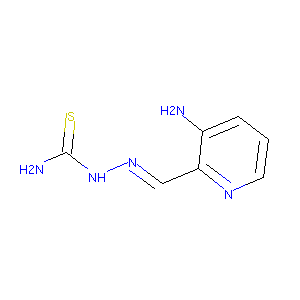
The iron-chelating drug triapine causes pronounced mitochondrial thiol redox stress. Toxicol Lett. 2011 Mar 5;201(2):130-6. doi: 10.1016/j.toxlet.2010.12.017. Epub 2010 Dec 31.
|
| 10 |
Distinct mechanisms of cell-kill by triapine and its terminally dimethylated derivative Dp44mT due to a loss or gain of activity of their copper(II) complexes. Biochem Pharmacol. 2014 Oct 1;91(3):312-22. doi: 10.1016/j.bcp.2014.08.006. Epub 2014 Aug 15.
|
|
|
|
|
|
|