| 1 |
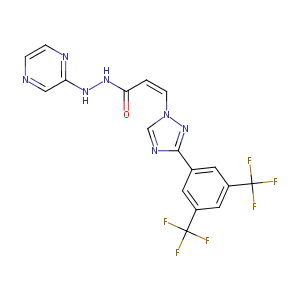
ClinicalTrials.gov (NCT03955783) Venetoclax and Selinexor in Treating Patients With Relapsed or Refractory High Risk Hematologic Malignancies
|
| 2 |
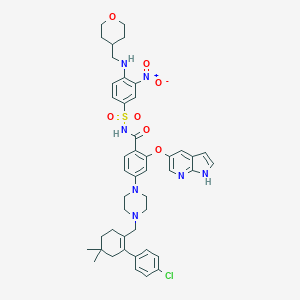
Drugs@FDA. U.S. Food and Drug Administration. U.S. Department of Health & Human Services. 2015
|
| 3 |
Clinical pipeline report, company report or official report of the Pharmaceutical Research and Manufacturers of America (PhRMA)
|
| 4 |
Clinical pipeline report, company report or official report of the Pharmaceutical Research and Manufacturers of America (PhRMA)
|
| 5 |
ClinicalTrials.gov (NCT04349098) Evaluation of Activity and Safety of Oral Selinexor in Participants With Severe COVID-19 Infection. U.S. National Institutes of Health.
|
| 6 |
ClinicalTrials.gov (NCT02025985) Phase II Study of KPT-330 (Selinexor) in Female Patients With Advanced Gynaecologic Malignancies. U.S. National Institutes of Health.
|
| 7 |
Inhibition of CRM1-dependent nuclear export sensitizes malignant cells to cytotoxic and targeted agents. Semin Cancer Biol. 2014 August; 0: 62-73.
|
| 8 |
FDA label of Selinexor. The 2020 official website of the U.S. Food and Drug Administration.
|
| 9 |
The synergy of the XPO1 inhibitors combined with the BET inhibitor INCB057643 in high-grade B-cell lymphoma via downregulation of MYC expression. Sci Rep. 2023 Oct 29;13(1):18554. doi: 10.1038/s41598-023-45721-z.
|
| 10 |
Venclexta FDA label
|
| 11 |
ZCL-082, a boron-containing compound, induces apoptosis of non-Hodgkin's lymphoma via targeting p90 ribosomal S6 kinase 1/NF-B signaling pathway. Chem Biol Interact. 2022 Jan 5;351:109770. doi: 10.1016/j.cbi.2021.109770. Epub 2021 Nov 30.
|
| 12 |
Genomics and drug profiling of fatal TCF3-HLF-positive acute lymphoblastic leukemia identifies recurrent mutation patterns and therapeutic options. Nat Genet. 2015 Sep;47(9):1020-1029. doi: 10.1038/ng.3362. Epub 2015 Jul 27.
|
| 13 |
HSP90 Inhibitor PU-H71 in Combination with BH3-Mimetics in the Treatment of Acute Myeloid Leukemia. Curr Issues Mol Biol. 2023 Aug 23;45(9):7011-7026. doi: 10.3390/cimb45090443.
|
| 14 |
Superior efficacy of cotreatment with BET protein inhibitor and BCL2 or MCL1 inhibitor against AML blast progenitor cells. Blood Cancer J. 2019 Jan 15;9(2):4. doi: 10.1038/s41408-018-0165-5.
|
| 15 |
Prospective Drug Candidates as Human Multidrug Transporter ABCG2 Inhibitors: an In Silico Drug Discovery Study. Cell Biochem Biophys. 2021 Jun;79(2):189-200. doi: 10.1007/s12013-021-00985-y. Epub 2021 May 5.
|
| 16 |
Statin-induced Mitochondrial Priming Sensitizes Multiple Myeloma Cells to BCL2 and MCL-1 Inhibitors. Cancer Res Commun. 2023 Dec 8;3(12):2497-2509. doi: 10.1158/2767-9764.CRC-23-0350.
|
| 17 |
ClinicalTrials.gov (NCT05736978) Adaptive Treatment for Acute Myeloid Leukemia Based on D14 MRD Results
|
| 18 |
ClinicalTrials.gov (NCT05736965) A Study of Selinexor in Combination With Azacitidine and Venetoclax (SAV Regimen) in Treatment Nave Participants With Acute Myeloid Leukemia
|
|
|
|
|
|
|