Details of the Drug
General Information of Drug (ID: DMZPHIK)
| Drug Name |
Beclomethasone
|
||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Synonyms |
Anceron; Beclocort; Beclometason; Beclometasona; Beclometasonum; Becloval; Becodisks; Beconasol; Becotide; Inalone; Viarex; Beclometasondipropionat Mikron; Beclometasone [INN]; Vancenase AQ; B 0385; BMJ 5800; Beclometason (TN); Beclometasona [INN-Spanish]; Beclometasone (INN); Beclometasonum [INN-Latin]; Beconase (TN); Clipper (TN); Propaderm (TN); Vancenase (TN); (11beta,16beta)-9-chloro-11,17,21-trihydroxy-16-methylpregna-1,4-diene-3,20-dione; (8R,9S,10S,11S,13R,14R,16R,17R)-9-chloro-11,17-dihydroxy-17-(2-hydroxyacetyl)-10,13,16-trimethyl-6,7,8,11,12,14,15,16-octahydrocyclopenta[a]phenanthren-3-one; (8S,9R,10S,11S,13S,14S,16S,17R)-9-chloro-11,17-dihydroxy-17-(2-hydroxyacetyl)-10,13,16-trimethyl-6,7,8,11,12,14,15,16-octahydrocyclopenta[a]phenanthren-3-one; 9-Chloro-11beta,17,21-trihydroxy-16beta-methylpregna-1,4-diene-3,20-dione; 9-chloro-11,17-dihydroxy-17-(2-hydroxyacetyl)-10,13,16-trimethyl-6,7,8,11,12,14,15,16-octahydrocyclopenta[a]phenanthren-3-one; 9alpha-Chloro-11beta,17alpha,21-trihydroxy-16beta-methylpregna-1,4-diene-3,20-dione; 9alpha-Chloro-16beta-methyl-1,4-pregnadiene-11beta,17alpha,21-triol-3,20-dione; 9alpha-Chloro-16beta-methylprednisolone
|
||||||||||||||||||||||||||
| Indication |
|
||||||||||||||||||||||||||
| Therapeutic Class |
Antiasthmatic Agents
|
||||||||||||||||||||||||||
| ATC Code |
|
||||||||||||||||||||||||||
| Drug Type |
Small molecular drug
|
||||||||||||||||||||||||||
| Structure |
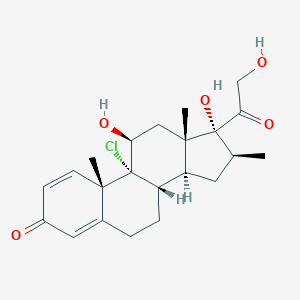 |
||||||||||||||||||||||||||
| 3D MOL | 2D MOL | ||||||||||||||||||||||||||
| #Ro5 Violations (Lipinski): 0 | Molecular Weight (mw) | 408.9 | |||||||||||||||||||||||||
| Logarithm of the Partition Coefficient (xlogp) | 2.2 | ||||||||||||||||||||||||||
| Rotatable Bond Count (rotbonds) | 2 | ||||||||||||||||||||||||||
| Hydrogen Bond Donor Count (hbonddonor) | 3 | ||||||||||||||||||||||||||
| Hydrogen Bond Acceptor Count (hbondacc) | 5 | ||||||||||||||||||||||||||
| Chemical Identifiers |
|
||||||||||||||||||||||||||
| Cross-matching ID | |||||||||||||||||||||||||||
| Repurposed Drugs (RPD) | Click to Jump to the Detailed RPD Information of This Drug | ||||||||||||||||||||||||||
Molecular Interaction Atlas of This Drug
 Drug Therapeutic Target (DTT) |
|
|||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
 Drug Transporter (DTP) |
|
|||||||||||||||||||||||||||||||
 Drug-Metabolizing Enzyme (DME) |
|
|||||||||||||||||||||||||||||||
| Molecular Interaction Atlas (MIA) | ||||||||||||||||||||||||||||||||
References
