| 1 |
ClinicalTrials.gov (NCT05677282) Single Dose Antibiotic Treatment of Acute Watery Diarrhea, Rifaximin Versus Azithromycin, With Loperamide Adjunct
|
| 2 |
Drugs@FDA. U.S. Food and Drug Administration. U.S. Department of Health & Human Services. 2015
|
| 3 |
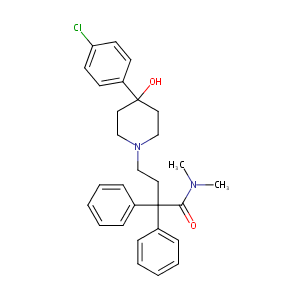
Loperamide FDA Label
|
| 4 |
Screening of an FDA-approved Compound Library Identifies Four Small-Molecule Inhibitors of Middle East Respiratory Syndrome Coronavirus Replication in Cell Culture Antimicrob Agents Chemother. 2014 Aug;58(8):4875-84.
|
| 5 |
ClinicalTrials.gov (NCT01654939) Impact of an Antibiotic (Rifaximin) on Liver Scarring in HIV-Infected Patients With Liver Disease. U.S. National Institutes of Health.
|
| 6 |
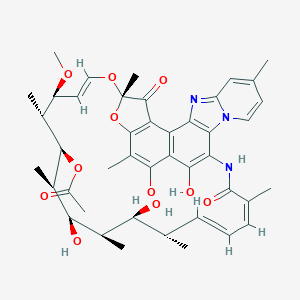
Rifaximin FDA Label
|
| 7 |
Loperamide: evidence of interaction with mu and delta opioid receptors. Life Sci. 1983;33 Suppl 1:315-8.
|
| 8 |
In vitro P-glycoprotein assays to predict the in vivo interactions of P-glycoprotein with drugs in the central nervous system. Drug Metab Dispos. 2008 Feb;36(2):268-75.
|
| 9 |
Loperamide: a pharmacological review. Rev Gastroenterol Disord. 2007;7 Suppl 3:S11-8.
|
| 10 |
Identification of cytochrome P450 isoforms involved in the metabolism of loperamide in human liver microsomes. Eur J Clin Pharmacol. 2004 Oct;60(8):575-81.
|
| 11 |
Reduction of the prodrug loperamide oxide to its active drug loperamide in the gut of rats, dogs, and humans. Drug Metab Dispos. 1995 Mar;23(3):354-62.
|
| 12 |
Advantageous Solubility-Permeability Interplay When Using Amorphous Solid Dispersion (ASD) Formulation for the BCS Class IV P-gp Substrate Rifaximin: Simultaneous Increase of Both the Solubility and the Permeability. AAPS J. 2017 May;19(3):806-813.
|
| 13 |
Probable interaction between warfarin and rifaximin in a patient treated for small intestine bacterial overgrowth. Ann Pharmacother. 2011 May;45(5):e25.
|
| 14 |
Solomonsterols A and B from Theonella swinhoeiThe first example of C-24 and C-23 sulfated sterols from a marine source endowed with a PXR agonistic activity. J Med Chem. 2011 Jan 13;54(1):401-5.
|
| 15 |
Total synthesis and pharmacological characterization of solomonsterol A, a potent marine pregnane-X-receptor agonist endowed with anti-inflammatory activity. J Med Chem. 2011 Jul 14;54(13):4590-9. doi: 10.1021/jm200241s. Epub 2011 Jun 3.
|
|
|
|
|
|
|