| 1 |
Recurrent recessive mutation in deoxyguanosine kinase causes idiopathic noncirrhotic portal hypertension.Hepatology. 2016 Jun;63(6):1977-86. doi: 10.1002/hep.28499. Epub 2016 Mar 31.
|
| 2 |
Drugs@FDA. U.S. Food and Drug Administration. U.S. Department of Health & Human Services. 2015
|
| 3 |
Loperamide FDA Label
|
| 4 |
Screening of an FDA-approved Compound Library Identifies Four Small-Molecule Inhibitors of Middle East Respiratory Syndrome Coronavirus Replication in Cell Culture Antimicrob Agents Chemother. 2014 Aug;58(8):4875-84.
|
| 5 |
Deubiquitylating enzymes and drug discovery: emerging opportunities. Nat Rev Drug Discov. 2018 Jan;17(1):57-78.
|
| 6 |
Loperamide: evidence of interaction with mu and delta opioid receptors. Life Sci. 1983;33 Suppl 1:315-8.
|
| 7 |
In vitro P-glycoprotein assays to predict the in vivo interactions of P-glycoprotein with drugs in the central nervous system. Drug Metab Dispos. 2008 Feb;36(2):268-75.
|
| 8 |
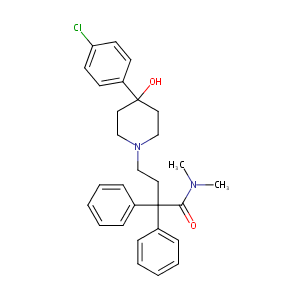
Loperamide: a pharmacological review. Rev Gastroenterol Disord. 2007;7 Suppl 3:S11-8.
|
| 9 |
Identification of cytochrome P450 isoforms involved in the metabolism of loperamide in human liver microsomes. Eur J Clin Pharmacol. 2004 Oct;60(8):575-81.
|
| 10 |
Reduction of the prodrug loperamide oxide to its active drug loperamide in the gut of rats, dogs, and humans. Drug Metab Dispos. 1995 Mar;23(3):354-62.
|
| 11 |
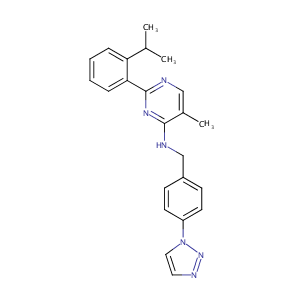
Synthesis and structure-activity relationship studies of N-benzyl-2-phenylpyrimidin-4-amine derivatives as potent USP1/UAF1 deubiquitinase inhibitors with anticancer activity against nonsmall cell lung cancer. J Med Chem. 2014 Oct 9;57(19):8099-110.
|
|
|
|
|
|
|