Details of the Drug
General Information of Drug (ID: DMB6871)
| Drug Name |
Testosterone enanthate
|
|||||
|---|---|---|---|---|---|---|
| Synonyms |
Testanthate; Testate; Testenate; Testinon; Testoenant; Testonenant; Testosterone 17-enanthate; Testosterone enantate; Testosterone heptanoate; Testosterone heptoate; Testosterone heptylate; Testosterone oenanthate; Testostroval; testosterone enanthate; 17-Hydroxyandrost-4-en-3-one, 17-heptanoate; 315-37-7; Andro L.A. 200; Andropository; Androtardyl; Atlatest; DePatestrye; Delatest; Depo-Testro Med; Ditate; Durathate; Everone; Exten test; Malogen L.A.; Malogen L.A.200; NSC-17591; Orquisteron-E; Primotestone; Reposo TMD; Reposo-TMD
|
|||||
| Affected Organisms |
Humans and other mammals
|
|||||
| Structure |
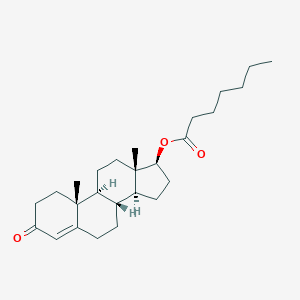 |
|||||
| 3D MOL | 2D MOL | |||||
| #Ro5 Violations (Lipinski): 1 | Molecular Weight (mw) | 400.6 | ||||
| Logarithm of the Partition Coefficient (xlogp) | 6.3 | |||||
| Rotatable Bond Count (rotbonds) | 7 | |||||
| Hydrogen Bond Donor Count (hbonddonor) | 0 | |||||
| Hydrogen Bond Acceptor Count (hbondacc) | 3 | |||||
| ADMET Property |
|
|||||
| Chemical Identifiers |
|
|||||
| Cross-matching ID | ||||||
Molecular Interaction Atlas of This Drug
 Drug-Metabolizing Enzyme (DME) |
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
 Drug Off-Target (DOT) |
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Molecular Interaction Atlas (MIA) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
References
