Details of the Drug
General Information of Drug (ID: DMNP60F)
| Drug Name |
Anastrozole
|
||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Synonyms |
Anastrazole; Anastrole; Anastrozol; Arimidex; Asiolex; Astra brand of anastrozole; AstraZeneca brand of anastrozole; Zeneca brand of anastrozole; ZD 1033; ZD1033; Zeneca ZD 1033; Arimidex (TN); Arimidex (Zeneca); Arimidex, Anastrozole; ZD-1033; Anastrozole [USAN:INN:BAN]; Anastrozole (JAN/USAN/INN); Alpha,alpha,alpha',alpha'-tetramethyl-5(1H-1,2,4-triazol-1-ylmethyl)-m-benzenediacetonitrile; Alpha,alpha,alpha',alpha'-Tetramethyl-5-(1H-1,2,4-triazol-1-ylmethyl)-m-benzenediacetonitrile; 1,3-benzenediacetonitrile, a, a,a', a'-tetramethyl-5-(1H-1,2,4-triazol-1-ylmethyl); 2,2'-(5-(1H-1,2,4-triazol-1-ylmethyl)-1,3-phenylene)bis(2-methylpropionitrile); 2,2'-[5-(1H-1,2,4-triazol-1-ylmethyl)-1,3-phenylene]bis(2-methylpropanenitrile); 2,2'-[5-(1H-1,2,4-triazol-1-ylmethyl)benzene-1,3-diyl]bis(2-methylpropanenitrile); 2-[3-(2-cyanopropan-2-yl)-5-(1,2,4-triazol-1-ylmethyl)phenyl]-2-methylpropanenitrile
|
||||||||||||||||||||||||||||||
| Indication |
|
||||||||||||||||||||||||||||||
| Therapeutic Class |
Anticancer Agents
|
||||||||||||||||||||||||||||||
| Affected Organisms |
Humans and other mammals
|
||||||||||||||||||||||||||||||
| ATC Code | |||||||||||||||||||||||||||||||
| Drug Type |
Small molecular drug
|
||||||||||||||||||||||||||||||
| Structure |
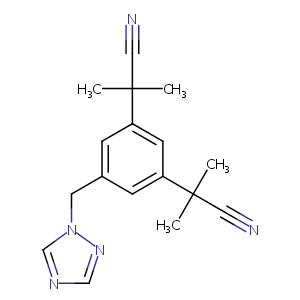 |
||||||||||||||||||||||||||||||
| 3D MOL | 2D MOL | ||||||||||||||||||||||||||||||
| #Ro5 Violations (Lipinski): 0 | Molecular Weight (mw) | 293.4 | |||||||||||||||||||||||||||||
| Logarithm of the Partition Coefficient (xlogp) | 2.1 | ||||||||||||||||||||||||||||||
| Rotatable Bond Count (rotbonds) | 4 | ||||||||||||||||||||||||||||||
| Hydrogen Bond Donor Count (hbonddonor) | 0 | ||||||||||||||||||||||||||||||
| Hydrogen Bond Acceptor Count (hbondacc) | 4 | ||||||||||||||||||||||||||||||
| ADMET Property |
|
||||||||||||||||||||||||||||||
| Adverse Drug Reaction (ADR) |
|
||||||||||||||||||||||||||||||
| Chemical Identifiers |
|
||||||||||||||||||||||||||||||
| Cross-matching ID | |||||||||||||||||||||||||||||||
| Combinatorial Drugs (CBD) | Click to Jump to the Detailed CBD Information of This Drug | ||||||||||||||||||||||||||||||
| Repurposed Drugs (RPD) | Click to Jump to the Detailed RPD Information of This Drug | ||||||||||||||||||||||||||||||
Molecular Interaction Atlas of This Drug
 Drug Therapeutic Target (DTT) |
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
 Drug Transporter (DTP) |
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||
 Drug-Metabolizing Enzyme (DME) |
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||
 Drug Off-Target (DOT) |
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Molecular Interaction Atlas (MIA) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Drug-Drug Interaction (DDI) Information of This Drug
|
Coadministration of a Drug Treating the Same Disease as Anastrozole
Coadministration of a Drug Treating the Disease Different from Anastrozole (Comorbidity)
|
||||||||||||||||||||||||||||||||||||||||||||||||||||
Drug Inactive Ingredient(s) (DIG) and Formulation(s) of This Drug
References
| 1 | URL: http://www.guidetopharmacology.org Nucleic Acids Res. 2015 Oct 12. pii: gkv1037. The IUPHAR/BPS Guide to PHARMACOLOGY in 2016: towards curated quantitative interactions between 1300 protein targets and 6000 ligands. (Ligand id: 5137). | ||||
|---|---|---|---|---|---|
| 2 | Anastrozole FDA Label | ||||
| 3 | FDA Approved Drug Products: Arimidex (anastrozole) oral tablets | ||||
| 4 | BDDCS applied to over 900 drugs | ||||
| 5 | Critical Evaluation of Human Oral Bioavailability for Pharmaceutical Drugs by Using Various Cheminformatics Approaches | ||||
| 6 | Rosenzweig P, Canal M, Patat A, Bergougnan L, Zieleniuk I, Bianchetti G: A review of the pharmacokinetics, tolerability and pharmacodynamics of amisulpride in healthy volunteers. Hum Psychopharmacol. 2002 Jan;17(1):1-13. | ||||
| 7 | Estimating the safe starting dose in phase I clinical trials and no observed effect level based on QSAR modeling of the human maximum recommended daily dose | ||||
| 8 | Miyajima M, Kusuhara H, Takahashi K, Takashima T, Hosoya T, Watanabe Y, Sugiyama Y: Investigation of the effect of active efflux at the blood-brain barrier on the distribution of nonsteroidal aromatase inhibitors in the central nervous system. J Pharm Sci. 2013 Sep;102(9):3309-19. doi: 10.1002/jps.23600. Epub 2013 May 27. | ||||
| 9 | ADReCS-Target: target profiles for aiding drug safety research and application. Nucleic Acids Res. 2018 Jan 4;46(D1):D911-D917. doi: 10.1093/nar/gkx899. | ||||
| 10 | Effective aromatase inhibition by anastrozole in a patient with gonadotropin-independent precocious puberty in McCune-Albright syndrome. J Pediatr Endocrinol Metab. 2002;15 Suppl 3:945-8. | ||||
| 11 | Anastrozole Aromatase Inhibitor Plasma Drug Concentration Genome-Wide Association Study: Functional Epistatic Interaction Between SLC38A7 and ALPPL2. Clin Pharmacol Ther. 2019 Jan 16. | ||||
| 12 | In vitro and in vivo oxidative metabolism and glucuronidation of anastrozole. Br J Clin Pharmacol. 2010 Dec;70(6):854-69. | ||||
| 13 | Aromatase inhibition: translation into a successful therapeutic approach. Clin Cancer Res. 2005 Apr 15;11(8):2809-21. doi: 10.1158/1078-0432.CCR-04-2187. | ||||
| 14 | Inhibition of estrogen receptor reduces connexin 43 expression in breast cancers. Toxicol Appl Pharmacol. 2018 Jan 1;338:182-190. doi: 10.1016/j.taap.2017.11.020. Epub 2017 Nov 24. | ||||
| 15 | Assessment of Five Pesticides as Endocrine-Disrupting Chemicals: Effects on Estrogen Receptors and Aromatase. Int J Environ Res Public Health. 2022 Feb 10;19(4):1959. doi: 10.3390/ijerph19041959. | ||||
| 16 | Aromatase inhibitors: cellular and molecular effects. J Steroid Biochem Mol Biol. 2005 May;95(1-5):83-9. doi: 10.1016/j.jsbmb.2005.04.010. | ||||
| 17 | Biomarker changes during neoadjuvant anastrozole, tamoxifen, or the combination: influence of hormonal status and HER-2 in breast cancer--a study from the IMPACT trialists. J Clin Oncol. 2005 Apr 10;23(11):2477-92. doi: 10.1200/JCO.2005.07.559. Epub 2005 Mar 14. | ||||
| 18 | Product Information. Tukysa (tucatinib). Seattle Genetics Inc, Bothell, WA. | ||||
| 19 | Product Information. Tavalisse (fostamatinib). Rigel Pharmaceuticals, South San Francisco, CA. | ||||
