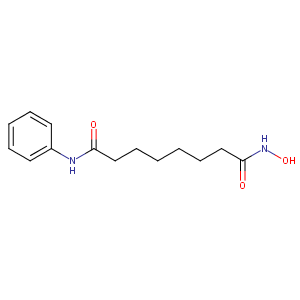
|
Cancer Drug Sensitivity Data Curated from 45 Cell Line(s) in Bone Tissue
| Cell Line |
Original Dataset |
IC50 |
IC90 |
EC50 |
AUC |
|
RPMI-3460/CS1
|
GDSC1; GDSC2
|
0.0551
|
3.0242
|
-0.1666
|
33.8456
|
|
ES8
|
GDSC1; GDSC2
|
0.041
|
1.6088
|
0.0268
|
32.9955
|
|
ES5
|
GDSC1; GDSC2
|
0.2914
|
2.0399
|
0.2569
|
30.5852
|
|
EW-3
|
GDSC1; GDSC2
|
0.3591
|
2.1958
|
0.3121
|
29.9696
|
|
ES1
|
GDSC1; GDSC2
|
0.4381
|
2.0949
|
0.4059
|
29.1025
|
|
EW-1
|
GDSC1; GDSC2
|
0.5351
|
1.8695
|
0.5229
|
28.0277
|
|
EW-7
|
GDSC1; GDSC2
|
0.5443
|
3.3735
|
0.2764
|
29.2391
|
|
U2OS
|
GDSC1; GDSC2; CTRP2; CTRP1
|
0.7243
|
3.4738
|
0.6567
|
29.7797
|
|
EW-12
|
GDSC2
|
0.7449
|
1.8932
|
0.7374
|
25.8983
|
|
EW-18
|
GDSC1; GDSC2
|
0.7648
|
3.0476
|
0.6011
|
26.5061
|
|
TC-71
|
GDSC1; GDSC2
|
0.7725
|
1.7925
|
0.7687
|
25.6024
|
|
MHH-ES-1
|
GDSC1; GDSC2; CTRP2
|
0.7785
|
2.1925
|
0.7768
|
28.4258
|
|
CHSA0108
|
GDSC1; GDSC2
|
0.7923
|
2.9771
|
0.6472
|
26.1338
|
|
EW-22
|
GDSC1; GDSC2
|
0.84
|
2.1628
|
0.8208
|
25.0031
|
|
NOS-1 [Human HNSCC]
|
GDSC1; GDSC2
|
0.9011
|
2.5287
|
0.8468
|
24.5704
|
|
SK-N-MC
|
CTRP2
|
0.9094
|
2.0899
|
0.9089
|
27.5494
|
|
EW-11
|
GDSC1; GDSC2
|
0.936
|
2.0507
|
0.9269
|
23.9886
|
|
CADO-ES1
|
GDSC1; GDSC2
|
1.0618
|
3.5433
|
0.7916
|
24.1047
|
|
SK-ES-1
|
GDSC1; GDSC2; CTRP2
|
1.1597
|
2.8904
|
1.1484
|
25.9181
|
|
EW-13
|
GDSC1; GDSC2
|
1.1743
|
4.0432
|
0.748
|
23.8318
|
|
ES7
|
GDSC1; GDSC2
|
1.1966
|
2.6849
|
1.1402
|
21.6168
|
|
HOS
|
GDSC1; GDSC2; CTRP2
|
1.2464
|
2.5033
|
1.245
|
25.3075
|
|
A-673
|
GDSC1; GDSC2; CTRP2
|
1.2858
|
3.4724
|
1.2412
|
25.1905
|
|
EW-16
|
GDSC1; GDSC2
|
1.2897
|
3.0084
|
1.1812
|
20.9568
|
|
HuO9
|
GDSC1; GDSC2
|
1.3099
|
3.345
|
1.1199
|
21.1913
|
|
SW1353
|
CTRP2
|
1.3376
|
3.021
|
1.3255
|
24.7358
|
|
ES4
|
GDSC1; GDSC2
|
1.431
|
3.2431
|
1.2793
|
19.7716
|
|
EW-24
|
GDSC1; GDSC2
|
1.6461
|
3.8014
|
1.3428
|
18.4538
|
|
SK-PN-DW
|
GDSC1; GDSC2
|
1.7826
|
3.4764
|
1.5974
|
16.4324
|
|
TC71
|
CTRP2
|
1.7844
|
3.3141
|
1.7716
|
21.7612
|
|
ES6
|
GDSC1; GDSC2
|
1.8301
|
3.6429
|
1.5961
|
16.2274
|
|
NY
|
GDSC1; GDSC2
|
2.0381
|
5.1214
|
1.2125
|
17.5977
|
|
CAL-72
|
GDSC1; GDSC2
|
2.1387
|
4.683
|
1.5107
|
15.4409
|
|
SaOS-2
|
GDSC1; GDSC2; CTRP2
|
2.3294
|
3.7458
|
2.3106
|
18.151
|
|
SJSA-1
|
GDSC1; GDSC2
|
2.3401
|
5.0882
|
1.5293
|
14.5596
|
|
HuO-3N1
|
GDSC1; GDSC2
|
2.5219
|
5.3159
|
1.5992
|
13.4706
|
|
CAL-78
|
GDSC1; GDSC2; CTRP2
|
2.5412
|
4.5583
|
2.4283
|
17.0643
|
|
Hs 888.T
|
CTRP2
|
2.7392
|
8.0118
|
1.3754
|
20.4572
|
|
CHSA8926
|
GDSC1; GDSC2
|
2.9
|
3.9108
|
2.6297
|
5.7865
|
|
MG-63
|
GDSC1; GDSC2; CTRP2
|
2.9836
|
6.7449
|
2.1789
|
16.716
|
|
H-EMC-SS
|
GDSC1; GDSC2
|
2.9846
|
6.1442
|
1.6207
|
11.7761
|
|
CHSA0011
|
GDSC1; GDSC2
|
3.3539
|
5.3727
|
2.323
|
6.2322
|
|
G-292 clone A141B1
|
GDSC1; GDSC2; CTRP2
|
3.4006
|
8.3828
|
1.8646
|
16.8764
|
|
U-CH2
|
GDSC2
|
3.4467
|
5.4948
|
2.3377
|
5.878
|
|
Hs 822.T
|
CTRP2
|
5.2213
|
6.6507
|
4.3835
|
2.4549
|
| ----------- |
|
|
|
|
|
| ⏷ Show the Full List of 45 Cancer Drug Sensitivity Data of This Drug |
|
Cancer Drug Sensitivity Data Curated from 219 Cell Line(s) in Haematopoietic And Lymphoid Tissue Tissue
| Cell Line |
Original Dataset |
IC50 |
IC90 |
EC50 |
AUC |
|
MEC1
|
CTRP2
|
0.9045
|
2.4135
|
0.9012
|
27.5915
|
|
JK1
|
CTRP2
|
1.412
|
2.9707
|
1.4036
|
24.2278
|
|
JM1
|
GDSC1; GDSC2; CTRP2
|
-0.3608
|
1.3205
|
-0.3622
|
36.0148
|
|
KMS-26
|
CTRP2
|
-2.0171
|
-0.7672
|
-2.0171
|
47.0446
|
|
Karpas-45
|
GDSC1; GDSC2
|
-1.6759
|
-0.3865
|
-1.6761
|
50.1537
|
|
NB4
|
GDSC1; GDSC2; CTRP2
|
-1.6051
|
-0.5798
|
-1.6051
|
44.3001
|
|
CRO-AP2
|
GDSC1; GDSC2
|
-1.5517
|
0.6495
|
-1.567
|
48.9219
|
|
DoHH2
|
GDSC1; GDSC2; CTRP2
|
-1.5444
|
1.4294
|
-1.5651
|
43.947
|
|
BCP-1
|
CTRP2
|
-1.5331
|
3.3853
|
-1.7584
|
44.2488
|
|
Kasumi-1
|
GDSC1; GDSC2; CTRP2
|
-1.489
|
0.1309
|
-1.4892
|
43.527
|
|
BE-13
|
GDSC1; GDSC2
|
-1.4884
|
0.2322
|
-1.4918
|
48.2783
|
|
SU-DHL-5
|
GDSC1; GDSC2; CTRP1
|
-1.3675
|
0.0296
|
-1.3676
|
55.489
|
|
SUP-B8
|
GDSC1; GDSC2
|
-1.2546
|
2.7642
|
-1.5321
|
46.4311
|
|
SKM-1
|
GDSC1; GDSC2; CTRP2; CTRP1
|
-1.1933
|
0.0858
|
-1.1933
|
42.1924
|
|
ALL-PO
|
GDSC1; GDSC2
|
-1.1843
|
1.3234
|
-1.2275
|
45.3438
|
|
Karpas-1106P
|
GDSC1; GDSC2
|
-1.0301
|
0.5551
|
-1.0335
|
43.6873
|
|
RPMI-8866
|
GDSC1
|
-1.0069
|
1.3846
|
-1.047
|
53.9048
|
|
SU-DHL-16
|
GDSC1; GDSC2
|
-0.9627
|
-0.0816
|
-0.9627
|
42.9964
|
|
Kasumi-2
|
CTRP2
|
-0.9573
|
1.5208
|
-0.9683
|
40.0182
|
|
SEM
|
CTRP2
|
-0.9473
|
0.3791
|
-0.9473
|
39.9176
|
|
EoL-1
|
GDSC1; GDSC2; CTRP2
|
-0.9453
|
0.9223
|
-0.9468
|
39.909
|
|
NU-DUL-1
|
GDSC1; GDSC2; CTRP2; CTRP1
|
-0.9316
|
0.5057
|
-0.9317
|
40.468
|
|
NKM-1
|
GDSC1; GDSC2
|
-0.9261
|
1.202
|
-0.9499
|
42.7228
|
|
MOLM-16
|
GDSC1; GDSC2; CTRP2; CTRP1
|
-0.8937
|
0.5083
|
-0.8937
|
40.2179
|
|
PL21
|
CTRP2; CTRP1
|
-0.8821
|
0.18
|
-0.8821
|
40.1415
|
|
OCI-Ly7
|
GDSC1; GDSC2
|
-0.8772
|
0.4783
|
-0.8786
|
42.1448
|
|
MY-M12
|
GDSC1; GDSC2
|
-0.8737
|
0.9507
|
-0.8842
|
42.1482
|
|
Peer
|
CTRP2
|
-0.8699
|
0.0248
|
-0.8699
|
39.4018
|
|
MOLT-13
|
GDSC1; GDSC2; CTRP2
|
-0.8696
|
1.0537
|
-0.8716
|
39.4062
|
|
RCH-ACV
|
GDSC1; GDSC2; CTRP2
|
-0.8585
|
1.1546
|
-0.8614
|
39.3352
|
|
Karpas-231
|
GDSC1; GDSC2
|
-0.8535
|
0.5966
|
-0.8558
|
41.9112
|
|
KMS-21-BM
|
CTRP2
|
-0.8417
|
1.2926
|
-0.8462
|
39.2285
|
|
CTB-1
|
GDSC1; GDSC2
|
-0.8347
|
2.0021
|
-0.934
|
42.0764
|
|
MV4-11
|
GDSC1; GDSC2; CTRP2
|
-0.809
|
0.4295
|
-0.809
|
38.996
|
|
A3/Kawakami
|
GDSC1; GDSC2; CTRP2
|
-0.7924
|
0.4889
|
-0.7924
|
38.8857
|
|
MOLM-13
|
GDSC1; GDSC2; CTRP2
|
-0.7891
|
1.0923
|
-0.791
|
38.8696
|
|
M-07e
|
CTRP2
|
-0.7438
|
0.7811
|
-0.7441
|
38.5628
|
|
JSC-1
|
GDSC1; GDSC2
|
-0.7281
|
0.7648
|
-0.7316
|
40.6584
|
|
RPMI-8402
|
GDSC1; GDSC2; CTRP2
|
-0.6874
|
0.3773
|
-0.6874
|
38.1863
|
|
BC-3
|
GDSC2
|
-0.6685
|
0.493
|
-0.669
|
40.0468
|
|
ALL-SIL
|
GDSC1; GDSC2
|
-0.6255
|
0.6871
|
-0.6271
|
39.6206
|
|
H9
|
GDSC1; GDSC2
|
-0.6176
|
0.928
|
-0.6228
|
39.558
|
|
TALL-1 [Human adult T-ALL]
|
GDSC2; CTRP2
|
-0.6085
|
0.9195
|
-0.6089
|
37.6617
|
|
SR
|
GDSC1; GDSC2; CTRP2
|
-0.5732
|
1.4107
|
-0.5767
|
37.4367
|
|
KMS-12-BM
|
GDSC1; GDSC2; CTRP2
|
-0.5703
|
0.9197
|
-0.5706
|
37.407
|
|
HuT 78
|
CTRP2
|
-0.5159
|
1.6716
|
-0.5234
|
37.0681
|
|
KMOE-2
|
GDSC1; GDSC2
|
-0.488
|
1.4957
|
-0.5142
|
38.3555
|
|
MHH-PREB-1
|
GDSC1; GDSC2
|
-0.4445
|
2.0068
|
-0.5183
|
38.1295
|
|
MOLT-4
|
GDSC1; GDSC2
|
-0.4173
|
1.2079
|
-0.4266
|
37.5687
|
|
ATN-1
|
GDSC1; GDSC2
|
-0.4171
|
1.0916
|
-0.423
|
37.5508
|
|
DB
|
GDSC1; GDSC2; CTRP2
|
-0.417
|
1.5415
|
-0.4209
|
36.3976
|
|
P12-Ichikawa
|
GDSC1; GDSC2; CTRP2
|
-0.3746
|
0.571
|
-0.3747
|
36.1025
|
|
WSU-NHL
|
GDSC1; GDSC2
|
-0.3718
|
1.3239
|
-0.3846
|
37.1292
|
|
Loucy
|
GDSC1; GDSC2
|
-0.3581
|
2.2085
|
-0.4541
|
37.3672
|
|
WSU-DLCL2
|
GDSC1; GDSC2; CTRP2; CTRP1
|
-0.3527
|
0.9682
|
-0.3528
|
36.6534
|
|
BL-70
|
CTRP2; CTRP1
|
-0.3123
|
0.9618
|
-0.3124
|
36.3871
|
|
KMS-34
|
CTRP2
|
-0.3043
|
1.2675
|
-0.3051
|
35.6363
|
|
RPMI-8226
|
GDSC1; GDSC2; CTRP2; CTRP1
|
-0.2976
|
1.7343
|
-0.3023
|
36.305
|
|
KY821
|
GDSC1; GDSC2
|
-0.2633
|
1.8313
|
-0.3067
|
36.1867
|
|
NALM-6
|
GDSC1; GDSC2; CTRP2
|
-0.2491
|
1.2638
|
-0.2497
|
35.268
|
|
JVM-3
|
GDSC1; GDSC2; CTRP2
|
-0.224
|
2.2224
|
-0.2434
|
35.1626
|
|
OCI-M1
|
GDSC1; GDSC2; CTRP2
|
-0.2058
|
0.7553
|
-0.2058
|
34.9776
|
|
A4/Fukuda
|
GDSC1; GDSC2; CTRP2
|
-0.1973
|
1.1762
|
-0.1976
|
34.9218
|
|
SUP-T1
|
GDSC1; GDSC2; CTRP2
|
-0.1948
|
1.3378
|
-0.1956
|
34.9069
|
|
Ri-1
|
CTRP2
|
-0.1913
|
2.1837
|
-0.2082
|
34.9365
|
|
OCI-AML-3
|
GDSC1; GDSC2; CTRP2; CTRP1
|
-0.1882
|
1.3363
|
-0.1888
|
35.5707
|
|
CCRF-CEM
|
GDSC1; GDSC2
|
-0.1818
|
1.4387
|
-0.1944
|
35.2228
|
|
SUP-M2
|
GDSC1; GDSC2; CTRP2
|
-0.1732
|
1.1535
|
-0.1734
|
34.7611
|
|
QIMR-WIL
|
GDSC1; GDSC2
|
-0.1578
|
2.3337
|
-0.2586
|
35.3994
|
|
Farage
|
GDSC1; GDSC2
|
-0.1578
|
2.0998
|
-0.2251
|
35.2438
|
|
Mino
|
CTRP2
|
-0.1526
|
0.8103
|
-0.1526
|
34.6234
|
|
GA-10
|
GDSC1; GDSC2; CTRP2
|
-0.1507
|
2.4613
|
-0.1804
|
34.7077
|
|
697
|
GDSC1; GDSC2; CTRP2
|
-0.1405
|
0.9456
|
-0.1405
|
34.5426
|
|
KOPN-8
|
GDSC1; GDSC2
|
-0.129
|
1.6203
|
-0.1496
|
34.7326
|
|
CA46
|
GDSC1; GDSC2; CTRP2
|
-0.1055
|
1.4825
|
-0.1066
|
34.313
|
|
KE-37
|
GDSC1; GDSC2; CTRP2
|
-0.1
|
1.0518
|
-0.1001
|
34.273
|
|
P30/OHK
|
GDSC1; GDSC2
|
-0.0954
|
0.8848
|
-0.0958
|
34.2949
|
|
RS4;11
|
GDSC1; GDSC2
|
-0.0889
|
2.1701
|
-0.1608
|
34.5774
|
|
KE-97
|
CTRP2; CTRP1
|
-0.0811
|
1.2303
|
-0.0812
|
34.8634
|
|
Jurkat
|
GDSC1; GDSC2; CTRP2
|
-0.0498
|
1.1565
|
-0.0499
|
33.9384
|
|
OCI-Ly10
|
CTRP2; CTRP1
|
-0.0483
|
2.9576
|
-0.1057
|
34.8315
|
|
SET-2
|
CTRP2
|
-0.0452
|
1.107
|
-0.0453
|
33.9079
|
|
JVM-2
|
GDSC1; GDSC2; CTRP2
|
-0.0386
|
4.3192
|
-0.3229
|
34.7564
|
|
AMO1
|
GDSC1; GDSC2; CTRP2; CTRP1
|
-0.0193
|
1.3984
|
-0.0197
|
34.4569
|
|
BV-173
|
GDSC1; GDSC2; CTRP2
|
-0.0138
|
2.2186
|
-0.0276
|
33.7442
|
|
NU-DHL-1
|
CTRP2
|
-0.0073
|
1.2539
|
-0.0075
|
33.6559
|
|
BC-1
|
GDSC1; GDSC2
|
0.0041
|
0.9834
|
0.0036
|
33.2974
|
|
NOMO-1
|
GDSC1; GDSC2; CTRP2; CTRP1
|
0.0127
|
1.789
|
0.0101
|
34.2535
|
|
KO52
|
CTRP2
|
0.0352
|
1.3148
|
0.035
|
33.3725
|
|
CML-T1
|
GDSC1; GDSC2; CTRP2
|
0.0694
|
1.5078
|
0.0688
|
33.1462
|
|
HEL
|
GDSC1; GDSC2; CTRP2
|
0.0749
|
1.1451
|
0.0749
|
33.1075
|
|
L-1236
|
GDSC1; GDSC2; CTRP2
|
0.1032
|
1.4408
|
0.1028
|
32.9204
|
|
P32/ISH
|
GDSC1; GDSC2
|
0.106
|
2.0232
|
0.0633
|
32.4855
|
|
NK-92MI
|
GDSC2
|
0.1126
|
1.5078
|
0.1046
|
32.2461
|
|
KMS-28BM
|
CTRP2
|
0.1313
|
4.8496
|
-0.2655
|
33.9851
|
|
KG-1
|
GDSC1; GDSC2
|
0.1372
|
1.574
|
0.1272
|
32.0092
|
|
VL51
|
GDSC1; GDSC2
|
0.1513
|
1.7775
|
0.1311
|
31.9187
|
|
MOLT-16
|
GDSC1; GDSC2; CTRP2
|
0.1993
|
1.0804
|
0.1993
|
32.2788
|
|
JJN-3
|
GDSC1; GDSC2; CTRP2
|
0.2133
|
2.2277
|
0.2039
|
32.2169
|
|
MM1.S
|
GDSC1; GDSC2; CTRP2
|
0.2203
|
2.1279
|
0.2136
|
32.1613
|
|
SU-DHL-6
|
GDSC1; GDSC2; CTRP2
|
0.2218
|
1.4171
|
0.2216
|
32.1294
|
|
OCI-AML-5
|
GDSC1; CTRP2
|
0.2261
|
1.5896
|
0.2256
|
32.1019
|
|
ROS-50
|
GDSC1; GDSC2
|
0.2269
|
2.4634
|
0.134
|
31.5237
|
|
P3HR-1
|
CTRP2; CTRP1
|
0.229
|
2.0092
|
0.2256
|
32.8309
|
|
CTV-1
|
GDSC1; GDSC2
|
0.2414
|
1.5387
|
0.235
|
30.9455
|
|
HH [Human lymphoma]
|
GDSC1; GDSC2; CTRP2
|
0.2597
|
1.9349
|
0.2568
|
31.8861
|
|
Ki-JK
|
CTRP2
|
0.2716
|
2.6644
|
0.2447
|
31.8865
|
|
MOLT-3
|
CTRP2
|
0.2789
|
1.007
|
0.2789
|
31.7482
|
|
HL-60
|
GDSC1; GDSC2; CTRP2
|
0.2789
|
1.2189
|
0.2789
|
31.7484
|
|
Ramos.2G6.4C10
|
GDSC1; GDSC2
|
0.2925
|
2.5513
|
0.1898
|
30.9167
|
|
SCC-3
|
GDSC2
|
0.2942
|
1.513
|
0.2895
|
30.4074
|
|
DND-41
|
GDSC1; GDSC2; CTRP2
|
0.2946
|
1.9636
|
0.2917
|
31.6535
|
|
ME1
|
GDSC1; GDSC2
|
0.297
|
2.3602
|
0.225
|
30.7178
|
|
EM-2
|
GDSC1; GDSC2; CTRP2
|
0.3032
|
1.5171
|
0.303
|
31.5873
|
|
EJM
|
GDSC1; GDSC2; CTRP2; CTRP1
|
0.3089
|
2.5502
|
0.2923
|
32.3476
|
|
CMK
|
GDSC1; CTRP2
|
0.3119
|
1.262
|
0.3119
|
31.5288
|
|
OPM-2
|
GDSC1; GDSC2; CTRP2
|
0.3121
|
1.2655
|
0.3121
|
31.5273
|
|
YT
|
GDSC1; GDSC2
|
0.3149
|
2.7632
|
0.1742
|
30.8826
|
|
JURL-MK1
|
GDSC1; CTRP2
|
0.3176
|
1.756
|
0.3167
|
31.4937
|
|
AML-193
|
CTRP2
|
0.3235
|
1.6312
|
0.3231
|
31.4529
|
|
JeKo-1
|
GDSC1; GDSC2; CTRP2
|
0.3252
|
1.8921
|
0.3233
|
31.446
|
|
KMS-11
|
GDSC1; GDSC2; CTRP2; CTRP1
|
0.3265
|
2.0029
|
0.324
|
32.1852
|
|
MOLP-8
|
GDSC1; GDSC2; CTRP2
|
0.3324
|
1.8215
|
0.3311
|
31.3966
|
|
Sc-1
|
GDSC1
|
0.3357
|
1.4806
|
0.3323
|
37.3484
|
|
THP-1
|
GDSC1; GDSC2; CTRP2; CTRP1
|
0.3446
|
1.7264
|
0.344
|
32.0598
|
|
Namalwa
|
GDSC1; GDSC2; CTRP2
|
0.3555
|
1.9893
|
0.3528
|
31.2474
|
|
HEL 92.1.7
|
CTRP2
|
0.3623
|
1.236
|
0.3623
|
31.1926
|
|
PF-382
|
GDSC1; GDSC2; CTRP2
|
0.3817
|
1.7398
|
0.381
|
31.0658
|
|
SK-MM-2
|
GDSC1; GDSC2
|
0.3835
|
1.7076
|
0.3743
|
29.5337
|
|
HAL-01
|
GDSC1; GDSC2
|
0.3898
|
2.3347
|
0.3276
|
29.7387
|
|
ST486
|
GDSC1; GDSC2; CTRP2
|
0.3903
|
1.5293
|
0.3902
|
31.0065
|
|
TK [Human B-cell lymphoma]
|
GDSC1; GDSC2
|
0.4128
|
2.9269
|
0.2457
|
30.0384
|
|
WIL2 NS
|
GDSC1; GDSC2
|
0.4254
|
1.9167
|
0.4067
|
29.1613
|
|
Ku812
|
GDSC1; GDSC2; CTRP2; CTRP1
|
0.4334
|
1.7148
|
0.433
|
31.474
|
|
OCI-Ly3
|
CTRP2
|
0.4457
|
1.9117
|
0.4443
|
30.6417
|
|
DEL
|
GDSC1; GDSC2; CTRP2
|
0.4793
|
2.0325
|
0.4771
|
30.4206
|
|
MHH-CALL-4
|
GDSC2; CTRP2
|
0.4795
|
2.0808
|
0.4768
|
30.421
|
|
HT
|
GDSC1; GDSC2; CTRP2; CTRP1
|
0.5142
|
2.2201
|
0.5106
|
30.9522
|
|
Mono-Mac-6
|
GDSC1; GDSC2; CTRP2
|
0.5314
|
3.5107
|
0.4374
|
30.3809
|
|
U-698-M
|
GDSC1; GDSC2
|
0.5395
|
1.9355
|
0.5237
|
28.0018
|
|
OCI-AML-2
|
GDSC1; GDSC2; CTRP2
|
0.5406
|
1.9215
|
0.5396
|
30.0084
|
|
SUP-B15
|
GDSC1; GDSC2; CTRP2
|
0.5486
|
2.3047
|
0.5428
|
29.9708
|
|
MLMA
|
GDSC1; GDSC2
|
0.5491
|
2.6824
|
0.4434
|
28.365
|
|
P31/FUJ
|
GDSC1; GDSC2; CTRP2
|
0.5493
|
1.9433
|
0.5483
|
29.9504
|
|
Karpas-422
|
GDSC1; GDSC2; CTRP2
|
0.557
|
2.8564
|
0.5286
|
29.9907
|
|
F-36P
|
CTRP2
|
0.5665
|
2.4255
|
0.558
|
29.8608
|
|
EHEB
|
GDSC1; CTRP2
|
0.5819
|
2.9195
|
0.5503
|
29.8356
|
|
KMS-27
|
CTRP2
|
0.5947
|
2.4572
|
0.5859
|
29.674
|
|
BONNA-12
|
GDSC2
|
0.6043
|
2.6779
|
0.5038
|
27.7852
|
|
Karpas-299
|
GDSC1; GDSC2; CTRP2
|
0.6109
|
1.9957
|
0.6098
|
29.5402
|
|
TUR
|
GDSC1; GDSC2
|
0.619
|
2.5764
|
0.5371
|
27.5417
|
|
KHM-1B
|
CTRP2
|
0.629
|
2.6508
|
0.614
|
29.4662
|
|
BALL-1
|
GDSC1; GDSC2
|
0.6404
|
3.5087
|
0.3424
|
28.4406
|
|
HPB-ALL
|
CTRP2
|
0.6458
|
1.6395
|
0.6457
|
29.3046
|
|
LAMA-84
|
GDSC1; GDSC2; CTRP2
|
0.6571
|
1.7631
|
0.657
|
29.2292
|
|
KMS-20
|
CTRP2
|
0.6637
|
3.0253
|
0.6278
|
29.3049
|
|
SUP-HD1
|
GDSC1; GDSC2; CTRP2
|
0.6642
|
3.0844
|
0.6236
|
29.3174
|
|
LP-1
|
GDSC1; GDSC2; CTRP2; CTRP1
|
0.6652
|
1.8898
|
0.6648
|
29.9466
|
|
NCI-H929
|
GDSC1; GDSC2; CTRP2
|
0.6873
|
1.9576
|
0.6867
|
29.0297
|
|
DG-75
|
GDSC1; GDSC2
|
0.7185
|
2.8255
|
0.5993
|
26.738
|
|
MOTN-1
|
CTRP2
|
0.7316
|
1.9459
|
0.7312
|
28.7338
|
|
IM-9
|
GDSC1; GDSC2
|
0.7453
|
2.1976
|
0.719
|
25.9897
|
|
Reh
|
GDSC1; GDSC2; CTRP2
|
0.7518
|
2.1782
|
0.7501
|
28.6034
|
|
Pfeiffer
|
CTRP2
|
0.7583
|
2.0168
|
0.7577
|
28.5567
|
|
Mono-Mac-1
|
CTRP2
|
0.7598
|
3.9057
|
0.6229
|
29.0063
|
|
L-363
|
GDSC1; GDSC2; CTRP2
|
0.762
|
1.7287
|
0.762
|
28.5299
|
|
SU-DHL-4
|
GDSC1; GDSC2; CTRP2; CTRP1
|
0.7945
|
2.1347
|
0.7936
|
29.0962
|
|
KCL-22
|
GDSC1; GDSC2; CTRP2
|
0.804
|
2.0437
|
0.8034
|
28.2521
|
|
VAL
|
GDSC1; GDSC2
|
0.8113
|
3.497
|
0.5323
|
26.6465
|
|
MHH-CALL-2
|
GDSC1; GDSC2
|
0.8342
|
2.0334
|
0.8229
|
25.0217
|
|
RL
|
GDSC1; GDSC2; CTRP2
|
0.8472
|
2.2425
|
0.8455
|
27.9682
|
|
Daudi
|
GDSC1; GDSC2; CTRP2
|
0.8751
|
2.9034
|
0.8551
|
27.8433
|
|
Jiyoye
|
GDSC1; GDSC2
|
0.8759
|
2.3252
|
0.8443
|
24.7064
|
|
ME1
|
CTRP2
|
0.8838
|
3.3984
|
0.825
|
27.9161
|
|
MOLM-6
|
CTRP2
|
0.8875
|
2.3088
|
0.8854
|
27.7009
|
|
EB1
|
CTRP2
|
0.8977
|
2.209
|
0.8966
|
27.6297
|
|
LC4-1
|
GDSC1; GDSC2
|
0.8991
|
3.7849
|
0.5383
|
26.2051
|
|
ML-2
|
GDSC1; GDSC2
|
0.9112
|
2.2742
|
0.8863
|
24.3182
|
|
GDM-1
|
GDSC1; GDSC2; CTRP2; CTRP1
|
0.9148
|
2.7142
|
0.9062
|
28.3291
|
|
HDLM-2
|
GDSC1; GDSC2; CTRP2
|
0.915
|
3.3136
|
0.8664
|
27.6739
|
|
GR-ST
|
GDSC1; GDSC2
|
0.9165
|
4.0282
|
0.4749
|
26.4584
|
|
PL-21
|
GDSC1; GDSC2
|
0.9209
|
3.3741
|
0.6872
|
25.3154
|
|
Raji
|
GDSC1; GDSC2; CTRP2
|
0.9219
|
2.6501
|
0.9136
|
27.4926
|
|
L-428
|
GDSC1; GDSC2; CTRP2
|
0.9384
|
2.4091
|
0.9355
|
27.3644
|
|
RPMI-6666
|
GDSC1; GDSC2; CTRP2
|
0.9426
|
2.6392
|
0.935
|
27.3523
|
|
Hs 445
|
GDSC2
|
0.956
|
3.6015
|
0.6585
|
25.3052
|
|
MN-60
|
GDSC1; GDSC2
|
0.9696
|
4.1456
|
0.4906
|
26.133
|
|
NCO2
|
CTRP2
|
0.975
|
2.6121
|
0.9687
|
27.1323
|
|
SU-DHL-1
|
GDSC2; CTRP2
|
1.0511
|
2.7038
|
1.0437
|
26.6287
|
|
TF-1
|
CTRP2
|
1.0709
|
1.8852
|
1.0709
|
26.472
|
|
U266B1
|
GDSC1; GDSC2; CTRP2
|
1.1191
|
2.9995
|
1.1019
|
26.2084
|
|
Granta-519
|
GDSC1; GDSC2; CTRP2
|
1.1373
|
3.1851
|
1.1095
|
26.1228
|
|
OCI-Ly19
|
GDSC1; GDSC2; CTRP2
|
1.1506
|
2.9435
|
1.1369
|
25.9871
|
|
SUP-T11
|
CTRP2
|
1.1679
|
2.8903
|
1.1568
|
25.8626
|
|
SU-DHL-8
|
GDSC2; CTRP2
|
1.1682
|
2.2795
|
1.1677
|
25.8252
|
|
KYO-1
|
CTRP2
|
1.1695
|
2.163
|
1.1693
|
25.8157
|
|
HC-1
|
GDSC1; GDSC2
|
1.191
|
3.8511
|
0.8331
|
23.2933
|
|
SU-DHL-10
|
GDSC2; CTRP2
|
1.2195
|
1.9303
|
1.2195
|
25.4818
|
|
L-540
|
GDSC1; GDSC2; CTRP2
|
1.2368
|
2.5488
|
1.2348
|
25.3735
|
|
MEG-01
|
GDSC1; GDSC2
|
1.2401
|
2.672
|
1.1887
|
21.1542
|
|
U-937
|
CTRP2; CTRP1
|
1.2482
|
2.2304
|
1.248
|
26.1041
|
|
EB2
|
GDSC1; GDSC2
|
1.278
|
2.3627
|
1.2626
|
20.5887
|
|
SIG-M5
|
GDSC2; CTRP2
|
1.2847
|
2.3184
|
1.2843
|
25.049
|
|
MC116
|
GDSC1; GDSC2; CTRP2; CTRP1
|
1.3001
|
2.5912
|
1.2986
|
25.7663
|
|
KMS-18
|
CTRP2
|
1.3124
|
2.5502
|
1.3109
|
24.868
|
|
K-562
|
GDSC1; GDSC2; CTRP2
|
1.3906
|
2.8819
|
1.3843
|
24.3631
|
|
RC-K8
|
GDSC1; GDSC2
|
1.4888
|
3.1029
|
1.3766
|
18.9799
|
|
CESS
|
GDSC1; GDSC2
|
1.5944
|
3.4196
|
1.4091
|
18.317
|
|
Hs 611.T
|
CTRP2
|
1.675
|
7.3225
|
0.5665
|
26.3511
|
|
MHH-CALL-3
|
CTRP2
|
1.8102
|
9.3559
|
-0.1698
|
28.35
|
|
ARH-77
|
GDSC1; GDSC2
|
1.8161
|
3.0627
|
1.7414
|
15.4979
|
|
KM-H2
|
GDSC1; GDSC2; CTRP2
|
1.8851
|
3.7248
|
1.8475
|
21.1741
|
|
MOLP-2
|
CTRP2
|
2.1113
|
3.5205
|
2.0982
|
19.5843
|
|
HD-MY-Z
|
CTRP2
|
2.3827
|
4.5215
|
2.2638
|
18.1406
|
|
Karpas-620
|
GDSC1; GDSC2; CTRP2
|
5.9675
|
8.1997
|
4.1536
|
2.2924
|
|
Toledo
|
CTRP2
|
6.4511
|
10.6655
|
3.3021
|
5.0095
|
|
BL-41
|
GDSC1; GDSC2; CTRP2
|
6.9072
|
8.3
|
4.5885
|
0.2178
|
|
REC-1
|
CTRP2
|
7.2375
|
9.69
|
4.1951
|
0.9755
|
|
KMM-1
|
CTRP2
|
7.5687
|
10.8417
|
3.881
|
1.6732
|
| ----------- |
|
|
|
|
|
| ⏷ Show the Full List of 219 Cancer Drug Sensitivity Data of This Drug |
|
Cancer Drug Sensitivity Data Curated from 27 Cell Line(s) in Oesophagus Tissue
| Cell Line |
Original Dataset |
IC50 |
IC90 |
EC50 |
AUC |
|
TE15
|
CTRP2
|
2.2589
|
4.0075
|
2.2123
|
18.715
|
|
TE-4
|
GDSC1; GDSC2
|
-0.4038
|
0.9883
|
-0.4073
|
37.4052
|
|
TE-1
|
GDSC1; GDSC2; CTRP2
|
0.6273
|
2.3317
|
0.6221
|
29.4448
|
|
TE-15
|
GDSC1
|
0.9019
|
2.4607
|
0.8565
|
30.5355
|
|
T.T
|
GDSC1; GDSC2
|
0.997
|
2.7045
|
0.9227
|
23.712
|
|
TE-8
|
GDSC1; GDSC2
|
1.0652
|
2.8518
|
0.9697
|
23.1389
|
|
KYSE-410
|
GDSC1; GDSC2; CTRP2; CTRP1
|
1.8236
|
3.7288
|
1.7893
|
22.4259
|
|
OE19
|
GDSC1; GDSC2; CTRP2
|
1.869
|
4.6127
|
1.6866
|
21.7842
|
|
OE33
|
GDSC1; GDSC2; CTRP2
|
1.9522
|
3.9512
|
1.8934
|
20.7997
|
|
TE-6
|
GDSC1; GDSC2
|
1.9587
|
5.5587
|
0.9328
|
19.5342
|
|
KYSE-70
|
GDSC1; GDSC2; CTRP2
|
2.0048
|
4.0889
|
1.9308
|
20.5016
|
|
KYSE-270
|
GDSC1; GDSC2
|
2.2669
|
4.7593
|
1.6071
|
14.3685
|
|
TE-5
|
GDSC1; GDSC2; CTRP2
|
2.3284
|
5.1404
|
2.0535
|
19.0585
|
|
COLO 680N
|
GDSC1; GDSC2; CTRP2
|
2.3845
|
5.0445
|
2.1416
|
18.5699
|
|
KYSE-140
|
GDSC1; GDSC2; CTRP2; CTRP1
|
2.4011
|
6.6828
|
1.646
|
21.2112
|
|
TE-14
|
CTRP2
|
2.4878
|
4.3707
|
2.4052
|
17.3141
|
|
OE21
|
GDSC1; GDSC2; CTRP2
|
2.6281
|
4.5693
|
2.5203
|
16.4675
|
|
KYSE-450
|
GDSC1; GDSC2; CTRP2; CTRP1
|
2.6645
|
5.1909
|
2.4371
|
17.5565
|
|
KYSE-180
|
GDSC1; GDSC2; CTRP2
|
2.6841
|
5.6001
|
2.2979
|
17.1039
|
|
EC-GI-10
|
GDSC1; GDSC2; CTRP2
|
2.7352
|
4.4941
|
2.6504
|
15.6744
|
|
TE-9
|
GDSC1; GDSC2; CTRP2
|
2.7677
|
4.3411
|
2.7104
|
15.3623
|
|
KYSE-510
|
GDSC1; GDSC2; CTRP2
|
2.7685
|
4.792
|
2.6247
|
15.6601
|
|
KYSE-30
|
CTRP2
|
2.7964
|
4.8843
|
2.6328
|
15.5448
|
|
TE-10
|
GDSC1; GDSC2; CTRP2
|
2.8498
|
4.9349
|
2.6784
|
15.2173
|
|
KYSE-150
|
GDSC1; GDSC2; CTRP2
|
2.9004
|
4.2974
|
2.8582
|
14.4263
|
|
TE-11
|
GDSC1; GDSC2; CTRP2
|
3.442
|
5.6682
|
3.0929
|
11.9377
|
|
KYSE-520
|
GDSC1; GDSC2; CTRP2
|
4.552
|
9.4456
|
2.421
|
11.9938
|
| ----------- |
|
|
|
|
|
| ⏷ Show the Full List of 27 Cancer Drug Sensitivity Data of This Drug |
|
Cancer Drug Sensitivity Data Curated from 39 Cell Line(s) in Pancreas Tissue
| Cell Line |
Original Dataset |
IC50 |
IC90 |
EC50 |
AUC |
|
N2a-PK1
|
CTRP2
|
3.579
|
8.2868
|
2.0839
|
15.5691
|
|
HuP-T4
|
GDSC1; GDSC2; CTRP2; CTRP1
|
1.3118
|
4.0214
|
1.2114
|
26.0211
|
|
MIA PaCa-2
|
GDSC1; GDSC2; CTRP2
|
1.3416
|
2.7603
|
1.3375
|
24.6822
|
|
PSN1
|
GDSC1; GDSC2; CTRP2
|
1.698
|
3.8412
|
1.6368
|
22.5013
|
|
PaTu 8988s
|
CTRP2
|
1.7016
|
5.4158
|
1.2885
|
23.7187
|
|
PaTu 8902
|
GDSC1; GDSC2; CTRP2; CTRP1
|
1.9391
|
3.5466
|
1.9225
|
21.6055
|
|
L3.3
|
CTRP2
|
1.9958
|
4.2322
|
1.8988
|
20.6408
|
|
Hs 766T
|
GDSC1; GDSC2; CTRP2
|
2.0262
|
5.5559
|
1.5979
|
21.6233
|
|
PL4
|
GDSC1; GDSC2
|
2.0854
|
3.9524
|
1.76
|
14.1963
|
|
DAN-G
|
GDSC1; GDSC2; CTRP2
|
2.1336
|
4.0934
|
2.0678
|
19.6153
|
|
QGP-1
|
GDSC1; GDSC2; CTRP2; CTRP1
|
2.1499
|
5.0774
|
1.9056
|
21.0024
|
|
BxPC-3
|
GDSC1; GDSC2; CTRP2
|
2.1799
|
4.7836
|
1.9854
|
19.7577
|
|
TCC-Pan2
|
CTRP2
|
2.2377
|
3.6566
|
2.2212
|
18.7537
|
|
Panc 08.13
|
GDSC1; GDSC2; CTRP2
|
2.2456
|
4.5351
|
2.1124
|
19.1038
|
|
KP-3
|
GDSC1; GDSC2; CTRP2; CTRP1
|
2.2946
|
5.1915
|
2.0332
|
20.1109
|
|
SUIT-2
|
GDSC1; GDSC2; CTRP2; CTRP1
|
2.3487
|
4.6755
|
2.2153
|
19.305
|
|
PK-59
|
CTRP2
|
2.3534
|
3.7896
|
2.3324
|
17.9982
|
|
KP-4
|
GDSC1; GDSC2; CTRP2
|
2.499
|
3.8687
|
2.4783
|
17.0274
|
|
SNU-213
|
CTRP2
|
2.5567
|
6.2017
|
1.9444
|
18.7945
|
|
Capan-1
|
GDSC1; GDSC2; CTRP2
|
2.6097
|
4.4264
|
2.527
|
16.5026
|
|
Panc 05.04
|
CTRP2
|
2.6256
|
4.3933
|
2.5497
|
16.3732
|
|
Panc 10.05
|
GDSC1; GDSC2; CTRP2; CTRP1
|
2.6644
|
4.6324
|
2.5655
|
17.1058
|
|
HuP-T3
|
GDSC1; GDSC2; CTRP2
|
2.7683
|
6.6899
|
1.9761
|
18.0837
|
|
Panc 04.03
|
GDSC1; GDSC2; CTRP2
|
2.7884
|
5.1716
|
2.5464
|
15.8807
|
|
HPAC
|
GDSC1; GDSC2; CTRP2; CTRP1
|
2.8029
|
5.2215
|
2.5799
|
16.6303
|
|
AsPC-1
|
GDSC1; GDSC2; CTRP2
|
2.8236
|
5.5702
|
2.4592
|
16.0967
|
|
Panc 03.27
|
GDSC1; GDSC2; CTRP2; CTRP1
|
2.9799
|
4.9674
|
2.8378
|
15.1785
|
|
CFPAC-1
|
GDSC1; GDSC2; CTRP2; CTRP1
|
3.0424
|
6.3406
|
2.4619
|
16.3749
|
|
KP-1N
|
GDSC1; GDSC2
|
3.1847
|
5.1841
|
2.2742
|
7.0435
|
|
SU.86.86
|
GDSC1; GDSC2
|
3.2373
|
6.3751
|
1.7243
|
10.3502
|
|
KP-2
|
GDSC1; GDSC2; CTRP2
|
3.511
|
6.1265
|
2.9881
|
12.1469
|
|
Capan-2
|
GDSC1; GDSC2; CTRP2; CTRP1
|
3.5881
|
6.2682
|
3.0699
|
12.5717
|
|
MZ-PC-1
|
GDSC1; GDSC2
|
3.6281
|
6.7698
|
1.846
|
8.4808
|
|
PK-45H
|
CTRP2
|
3.6707
|
5.9867
|
3.2133
|
10.8395
|
|
YAPC
|
GDSC1; GDSC2; CTRP2
|
3.7283
|
6.3796
|
3.1081
|
11.0963
|
|
HPAF-II
|
GDSC1; GDSC2; CTRP2
|
3.8086
|
7.6306
|
2.6179
|
12.8611
|
|
SW1990
|
GDSC1; GDSC2; CTRP2
|
3.8346
|
5.7249
|
3.4914
|
9.3162
|
|
Panc 02.03
|
GDSC1; GDSC2; CTRP2
|
3.9889
|
6.69
|
3.2343
|
9.9254
|
|
PaTu 8988t
|
GDSC1; GDSC2; CTRP2
|
4.9825
|
6.8346
|
4.0972
|
4.1026
|
| ----------- |
|
|
|
|
|
| ⏷ Show the Full List of 39 Cancer Drug Sensitivity Data of This Drug |
|
Cancer Drug Sensitivity Data Curated from 76 Cell Line(s) in Skin Tissue
| Cell Line |
Original Dataset |
IC50 |
IC90 |
EC50 |
AUC |
|
SH4
|
CTRP2
|
3.12
|
11.5653
|
0.0681
|
24.1171
|
|
CHL-1
|
GDSC1; GDSC2
|
-0.162
|
2.5268
|
-0.2963
|
35.5927
|
|
SK-MEL-5
|
GDSC1; GDSC2; CTRP2; CTRP1
|
0.0646
|
1.9876
|
0.0597
|
33.9189
|
|
G-mel
|
GDSC1; GDSC2
|
0.1016
|
1.8156
|
0.0769
|
32.4396
|
|
G-361
|
GDSC1; GDSC2; CTRP2; CTRP1
|
0.2163
|
3.4226
|
0.1239
|
33.2036
|
|
451Lu
|
GDSC1; GDSC2
|
0.4198
|
2.7608
|
0.2885
|
29.788
|
|
COLO 741
|
CTRP2; CTRP1
|
0.5923
|
11.9916
|
-2.5184
|
37.99
|
|
Mel Ho
|
GDSC1; GDSC2; CTRP2
|
0.7477
|
2.7422
|
0.7319
|
28.678
|
|
IGR-1
|
GDSC1; GDSC2; CTRP2
|
1.05
|
2.9793
|
1.0316
|
26.6729
|
|
SK-MEL-2
|
GDSC1; GDSC2; CTRP2
|
1.0675
|
3.2028
|
1.0355
|
26.6021
|
|
LOX-IMVI
|
GDSC1; GDSC2; CTRP2
|
1.1775
|
3.553
|
1.1186
|
25.9603
|
|
A-375
|
GDSC1; GDSC2; CTRP2; CTRP1
|
1.2274
|
3.0489
|
1.2139
|
26.2851
|
|
Hs 938.T
|
GDSC1; GDSC2
|
1.2604
|
3.146
|
1.1177
|
21.4325
|
|
COLO 783
|
GDSC1; GDSC2
|
1.2829
|
3.2198
|
1.1237
|
21.2954
|
|
WM35
|
GDSC1; GDSC2
|
1.284
|
3.0752
|
1.16
|
21.096
|
|
A-431
|
GDSC1; GDSC2
|
1.3511
|
3.3759
|
1.1561
|
20.8058
|
|
Hs 936.T
|
CTRP2; CTRP1
|
1.3527
|
4.1192
|
1.2402
|
25.7926
|
|
MZ-MEL-2
|
GDSC1; GDSC2
|
1.4012
|
3.8371
|
1.0655
|
21.0782
|
|
IGR-37
|
GDSC1; GDSC2; CTRP2
|
1.4479
|
3.5779
|
1.4015
|
24.1168
|
|
M14
|
GDSC1; GDSC2
|
1.5497
|
3.7007
|
1.2725
|
19.2711
|
|
MeWo
|
GDSC1; GDSC2; CTRP2
|
1.6311
|
4.1518
|
1.5196
|
23.12
|
|
RPMI-7951
|
GDSC1; GDSC2; CTRP2
|
1.6395
|
3.0804
|
1.6323
|
22.708
|
|
HMV-II
|
GDSC1; GDSC2
|
1.7072
|
5.0166
|
0.9217
|
20.594
|
|
DJM-1
|
GDSC1; GDSC2
|
1.7187
|
5.0606
|
0.9143
|
20.5899
|
|
A101D
|
GDSC1; GDSC2; CTRP2
|
1.7924
|
3.2215
|
1.7836
|
21.6947
|
|
WM88
|
CTRP2
|
1.8235
|
4.3108
|
1.699
|
21.8839
|
|
Hs 852.T
|
CTRP2; CTRP1
|
1.8707
|
5.8447
|
1.3744
|
23.7316
|
|
A-388
|
GDSC1; GDSC2
|
1.8948
|
4.3239
|
1.4103
|
17.0117
|
|
GAK
|
GDSC1; GDSC2
|
1.9129
|
3.2543
|
1.8021
|
14.7208
|
|
IPC-298
|
GDSC1; GDSC2; CTRP2
|
1.9183
|
4.7773
|
1.7018
|
21.5768
|
|
Hs 944.T
|
CTRP2; CTRP1
|
1.9386
|
4.122
|
1.8673
|
21.7931
|
|
SK-MEL-30
|
GDSC1; GDSC2; CTRP2
|
1.9573
|
4.1672
|
1.8679
|
20.8711
|
|
LB2518-MEL
|
GDSC1; GDSC2
|
1.9661
|
4.5949
|
1.3713
|
16.9487
|
|
UACC-257
|
GDSC1; GDSC2; CTRP2
|
2.0063
|
5.0353
|
1.7314
|
21.2
|
|
HT-144
|
GDSC1; GDSC2; CTRP2
|
2.093
|
3.8331
|
2.0558
|
19.7879
|
|
IST-MEL1
|
GDSC1; GDSC2
|
2.1147
|
3.8114
|
1.8445
|
13.5885
|
|
COLO 829
|
GDSC1; GDSC2; CTRP2
|
2.137
|
5.674
|
1.6797
|
20.9947
|
|
UACC-62
|
GDSC1; GDSC2; CTRP2
|
2.1421
|
3.4537
|
2.1329
|
19.3661
|
|
LB373-MEL-D
|
GDSC1; GDSC2
|
2.1527
|
4.0006
|
1.8128
|
13.6095
|
|
CP66-MEL
|
GDSC1; GDSC2
|
2.1624
|
4.0975
|
1.7844
|
13.7344
|
|
Hs 940.T
|
GDSC1; GDSC2
|
2.1951
|
4.1406
|
1.8013
|
13.5014
|
|
SK-MEL-1
|
GDSC1; GDSC2; CTRP2; CTRP1
|
2.2145
|
4.3233
|
2.1333
|
20.0091
|
|
WM266-4
|
CTRP2
|
2.2631
|
3.8128
|
2.2363
|
18.6195
|
|
SK-MEL-3
|
GDSC1; GDSC2; CTRP2
|
2.3435
|
4.8932
|
2.1367
|
18.713
|
|
Mel JuSo
|
GDSC1; GDSC2; CTRP2
|
2.3606
|
4.6814
|
2.206
|
18.4138
|
|
VMRC-MELG
|
GDSC1; GDSC2
|
2.37
|
4.3007
|
1.9157
|
12.1232
|
|
Hs 294T
|
CTRP2
|
2.3889
|
4.4514
|
2.2841
|
18.0503
|
|
A2058
|
GDSC1; GDSC2; CTRP2
|
2.4248
|
3.9316
|
2.3954
|
17.5511
|
|
K029AX
|
CTRP2
|
2.4865
|
4.0864
|
2.4444
|
17.1832
|
|
COLO 792
|
GDSC1; GDSC2; CTRP2
|
2.5735
|
6.3522
|
1.9046
|
18.897
|
|
COLO 800
|
GDSC1; GDSC2; CTRP2
|
2.619
|
4.4888
|
2.5259
|
16.4768
|
|
MDA-MB-435S
|
CTRP2
|
2.644
|
4.6724
|
2.5162
|
16.4323
|
|
COLO 679
|
GDSC1; GDSC2
|
2.6613
|
5.0078
|
1.8806
|
11.2455
|
|
SK-MEL-31
|
GDSC1; GDSC2; CTRP2
|
2.6696
|
4.3781
|
2.5996
|
16.0596
|
|
MZ-MEL-7
|
GDSC1; GDSC2
|
2.6861
|
7.6355
|
0.6147
|
18.7268
|
|
WM278
|
GDSC1; GDSC2
|
2.6983
|
4.0276
|
2.3725
|
8.11
|
|
SH-4
|
GDSC1; GDSC2
|
2.7661
|
5.1866
|
1.8937
|
10.7979
|
|
MMAc-SF
|
GDSC1; GDSC2
|
2.7781
|
5.9782
|
1.5172
|
13.0896
|
|
CP50-MEL-B
|
GDSC1; GDSC2
|
2.8433
|
5.7473
|
1.6904
|
11.8008
|
|
WM115
|
GDSC1; GDSC2; CTRP2
|
2.8462
|
4.1873
|
2.8138
|
14.754
|
|
WM1552C
|
GDSC1; GDSC2
|
2.9331
|
4.5659
|
2.3522
|
7.3719
|
|
RVH-421
|
GDSC1; GDSC2; CTRP2
|
2.9628
|
5.0145
|
2.7808
|
14.5037
|
|
WM983B
|
CTRP2
|
3.0412
|
6.9014
|
2.1726
|
16.5858
|
|
WM793
|
GDSC1; GDSC2; CTRP2
|
3.0424
|
4.6474
|
2.9537
|
13.6419
|
|
WM1799
|
CTRP2
|
3.1371
|
5.6275
|
2.7788
|
13.994
|
|
HMCB
|
CTRP2
|
3.264
|
5.987
|
2.782
|
13.6218
|
|
SK-MEL-24
|
GDSC1; GDSC2; CTRP2
|
3.642
|
5.8392
|
3.2419
|
10.808
|
|
SK-MEL-28
|
GDSC1; GDSC2; CTRP2; CTRP1
|
3.8907
|
6.2502
|
3.3956
|
10.5081
|
|
Hs 895.T
|
CTRP2
|
4.3357
|
9.5202
|
2.1915
|
13.3971
|
|
WT2-iPS
|
GDSC1; GDSC2; CTRP2
|
4.5504
|
8.2947
|
2.9912
|
9.6313
|
|
CJM [Human melanoma]
|
CTRP2
|
5.0885
|
6.767
|
4.2195
|
3.3832
|
|
IGR-39
|
CTRP2
|
5.2458
|
8.4963
|
3.4833
|
6.1831
|
|
Hs 934.T
|
CTRP2
|
5.5003
|
10.0155
|
2.9279
|
8.0472
|
|
Hs 839.T
|
CTRP2
|
5.6568
|
7.5842
|
4.2415
|
2.3598
|
|
Hs 688(A).T
|
CTRP2
|
5.8253
|
7.351
|
4.4586
|
1.3013
|
|
EquiPSC Line K2
|
GDSC1; GDSC2
|
1.6548
|
5.2772
|
0.7535
|
21.7551
|
| ----------- |
|
|
|
|
|
| ⏷ Show the Full List of 76 Cancer Drug Sensitivity Data of This Drug |
|
Cancer Drug Sensitivity Data Curated from 11 Cell Line(s) in Aero Dig Tract Tissue
| Cell Line |
Original Dataset |
IC50 |
IC90 |
EC50 |
AUC |
|
KYAE-1
|
GDSC1; GDSC2
|
0.0608
|
1.4789
|
0.0526
|
32.767
|
|
ESO-51
|
GDSC1; GDSC2
|
0.1022
|
3.8524
|
-0.3499
|
34.4023
|
|
OACP4 C
|
GDSC1; GDSC2
|
0.5589
|
2.0652
|
0.5349
|
27.8487
|
|
ESO-26
|
GDSC1; GDSC2
|
0.7514
|
2.1999
|
0.7252
|
25.9281
|
|
FLO-1
|
GDSC1; GDSC2
|
0.9874
|
3.1739
|
0.813
|
24.336
|
|
SK-GT-4
|
GDSC1; GDSC2
|
1.4437
|
4.0415
|
1.0387
|
21.0407
|
|
TE-12
|
GDSC1; GDSC2
|
1.6493
|
5.9761
|
0.4322
|
23.5299
|
|
KYSE-50
|
GDSC1; GDSC2
|
1.8097
|
3.1178
|
1.7225
|
15.6286
|
|
KYSE-220
|
GDSC1; GDSC2
|
1.9816
|
3.6933
|
1.7432
|
14.7377
|
|
HCE-4
|
GDSC1; GDSC2
|
2.0554
|
5.9478
|
0.8467
|
19.6586
|
|
OACM5.1 C
|
GDSC1; GDSC2
|
2.7377
|
4.6767
|
2.1122
|
9.5463
|
| ----------- |
|
|
|
|
|
| ⏷ Show the Full List of 11 Cancer Drug Sensitivity Data of This Drug |
|
Cancer Drug Sensitivity Data Curated from 1 Cell Line(s) in Ascites Tissue
| Cell Line |
Original Dataset |
IC50 |
IC90 |
EC50 |
AUC |
|
COLO 684
|
GDSC1; GDSC2; CTRP2
|
1.8933
|
4.7786
|
1.6742
|
21.7523
|
| ----------- |
|
|
|
|
|
Cancer Drug Sensitivity Data Curated from 36 Cell Line(s) in Autonomic Ganglia Tissue
| Cell Line |
Original Dataset |
IC50 |
IC90 |
EC50 |
AUC |
|
IMR-32
|
CTRP2
|
-1.7308
|
2.5885
|
-1.8522
|
45.3507
|
|
NB69
|
GDSC1; GDSC2
|
-1.0245
|
1.1649
|
-1.0496
|
43.7084
|
|
SJNB-14
|
GDSC1; GDSC2
|
-0.8626
|
1.1702
|
-0.8825
|
42.0744
|
|
CHP-212
|
GDSC1; GDSC2; CTRP2
|
-0.5807
|
1.802
|
-0.5928
|
37.5144
|
|
TGW
|
GDSC1; GDSC2
|
-0.1251
|
1.0301
|
-0.1266
|
34.5984
|
|
GOTO
|
GDSC1; GDSC2
|
0.0728
|
2.5455
|
-0.0461
|
33.1888
|
|
SJNB-7
|
GDSC1; GDSC2
|
0.1202
|
4.5235
|
-0.5609
|
35.1595
|
|
Kelly
|
GDSC1; GDSC2; CTRP2
|
0.2111
|
1.3231
|
0.2111
|
32.2002
|
|
CHP-134
|
GDSC2
|
0.3306
|
2.2141
|
0.2798
|
30.2743
|
|
MHH-NB-11
|
GDSC1; GDSC2; CTRP2
|
0.4137
|
2.4638
|
0.4007
|
30.8938
|
|
SK-N-DZ
|
GDSC1; GDSC2; CTRP2
|
0.4234
|
1.8754
|
0.4222
|
30.7897
|
|
CHP-126
|
GDSC1; GDSC2; CTRP2
|
0.4615
|
1.6742
|
0.4612
|
30.5327
|
|
SiMa
|
GDSC1; GDSC2; CTRP2
|
0.4776
|
2.8399
|
0.4472
|
30.5262
|
|
SK-N-MC-IXC
|
GDSC1; GDSC2
|
0.5245
|
1.451
|
0.5234
|
28.0782
|
|
SJNB-5
|
GDSC1; GDSC2
|
0.5248
|
2.4796
|
0.4512
|
28.4437
|
|
SJNB-17
|
GDSC1; GDSC2
|
0.5392
|
1.6257
|
0.5357
|
27.9424
|
|
SJNB-10
|
GDSC1; GDSC2
|
0.6759
|
2.3742
|
0.6271
|
26.8016
|
|
NH-12
|
GDSC1; GDSC2
|
0.692
|
3.2341
|
0.4753
|
27.5097
|
|
SK-N-BE(2)
|
CTRP2
|
0.8499
|
3.9932
|
0.705
|
28.4343
|
|
NH-6
|
CTRP2
|
0.9263
|
2.7861
|
0.9134
|
27.4784
|
|
SJNB-6
|
GDSC1; GDSC2
|
0.936
|
2.1147
|
0.9236
|
24.0058
|
|
SJNB-13
|
GDSC1; GDSC2
|
0.9745
|
1.7322
|
0.9737
|
23.5606
|
|
KP-N-RT-BM-1
|
GDSC2
|
0.9784
|
4.0684
|
0.5276
|
25.8975
|
|
KP-N-SI9s
|
CTRP2
|
1.0098
|
2.6578
|
1.0029
|
26.9022
|
|
SK-N-BE(2)-M17
|
GDSC1; GDSC2
|
1.0458
|
1.8886
|
1.0438
|
22.8509
|
|
SJNB-12
|
GDSC1; GDSC2
|
1.1251
|
4.6564
|
0.463
|
25.5799
|
|
IMR-5
|
GDSC1; GDSC2
|
1.1391
|
2.3894
|
1.1151
|
22.0266
|
|
KP-N-YS
|
GDSC1; GDSC2
|
1.1659
|
3.111
|
1.023
|
22.3807
|
|
SK-N-AS
|
GDSC1; GDSC2; CTRP2
|
1.2468
|
4.796
|
0.9651
|
26.269
|
|
SK-N-SH
|
GDSC1; GDSC2; CTRP2
|
1.3051
|
4.4616
|
1.1067
|
25.5919
|
|
NB(TU)1
|
GDSC1; GDSC2
|
1.3523
|
2.6247
|
1.3149
|
19.956
|
|
SK-N-FI
|
GDSC1; GDSC2; CTRP2
|
1.4266
|
3.0213
|
1.4168
|
24.1349
|
|
NB1
|
GDSC1; GDSC2; CTRP2
|
1.5156
|
3.4235
|
1.4862
|
23.6081
|
|
GI-ME-N
|
GDSC1; GDSC2
|
1.9391
|
5.9567
|
0.7285
|
20.7713
|
|
LA-N-6
|
GDSC1; GDSC2
|
2.3404
|
5.0003
|
1.5704
|
14.3105
|
|
KP-N-YN
|
GDSC1; GDSC2; CTRP2
|
5.8817
|
11.3292
|
2.5696
|
8.8666
|
| ----------- |
|
|
|
|
|
| ⏷ Show the Full List of 36 Cancer Drug Sensitivity Data of This Drug |
|
Cancer Drug Sensitivity Data Curated from 8 Cell Line(s) in Biliary Tract Tissue
| Cell Line |
Original Dataset |
IC50 |
IC90 |
EC50 |
AUC |
|
SNU-1079
|
CTRP2
|
-2.3102
|
2.1449
|
-2.4153
|
49.046
|
|
SNU-245
|
CTRP2
|
1.2818
|
4.6906
|
1.0291
|
25.9358
|
|
SNU-478
|
CTRP2
|
1.3334
|
3.0791
|
1.3186
|
24.7726
|
|
HuCC-T1
|
GDSC1; GDSC2; CTRP2
|
1.7518
|
4.7416
|
1.529
|
22.7065
|
|
HuH-28
|
CTRP2
|
1.8703
|
4.0514
|
1.7924
|
21.4108
|
|
SNU-1196
|
CTRP2
|
2.0259
|
3.7049
|
1.9971
|
20.2065
|
|
SNU-308
|
CTRP2
|
3.4436
|
5.7184
|
3.0761
|
11.9967
|
|
SNU-869
|
CTRP2
|
3.5904
|
5.9015
|
3.1625
|
11.256
|
| ----------- |
|
|
|
|
|
| ⏷ Show the Full List of 8 Cancer Drug Sensitivity Data of This Drug |
|
Cancer Drug Sensitivity Data Curated from 90 Cell Line(s) in Breast Tissue
| Cell Line |
Original Dataset |
IC50 |
IC90 |
EC50 |
AUC |
|
HCC2157
|
GDSC1; GDSC2
|
-0.9541
|
0.6019
|
-0.9575
|
38.8883
|
|
HCC1599
|
GDSC2
|
0.2964
|
2.644
|
0.1774
|
28.0556
|
|
MDA-MB-453
|
GDSC1; GDSC2; CTRP2
|
0.3388
|
1.7492
|
0.338
|
31.3519
|
|
DU4475
|
GDSC1; GDSC2; CTRP2
|
0.3651
|
2.4052
|
0.3532
|
31.2137
|
|
HCC1428
|
GDSC1; GDSC2; CTRP2
|
0.3722
|
2.2517
|
0.3649
|
31.1512
|
|
CAL-148
|
GDSC2; CTRP2
|
0.7639
|
2.2533
|
0.7615
|
28.5251
|
|
EFM-19
|
GDSC1; GDSC2
|
0.7639
|
2.9463
|
0.6231
|
23.9154
|
|
EFM-192A
|
GDSC1; GDSC2; CTRP2
|
0.8991
|
4.1817
|
0.7235
|
28.2119
|
|
HCC2218
|
GDSC1; GDSC2; CTRP2
|
0.9409
|
3.5851
|
0.8639
|
27.5975
|
|
Evsa-T
|
GDSC1; GDSC2
|
0.9425
|
2.5487
|
0.8881
|
21.8831
|
|
MDA-MB-361
|
GDSC1; GDSC2; CTRP2
|
0.9912
|
3.7066
|
0.9015
|
27.3063
|
|
HCC1187
|
GDSC1; GDSC2
|
1.0874
|
9.4304
|
-1.7484
|
32.2851
|
|
MCF-7
|
GDSC1; GDSC2; CTRP2; CTRP1
|
1.0876
|
4.468
|
0.8898
|
27.8267
|
|
SK-BR-3
|
CTRP2
|
1.1699
|
2.5481
|
1.1673
|
25.8209
|
|
MDA-MB-330
|
GDSC1; GDSC2
|
1.1821
|
4.0856
|
0.7412
|
21.6086
|
|
AU565
|
GDSC1; GDSC2; CTRP2
|
1.1832
|
2.6772
|
1.1786
|
25.7392
|
|
OCUB-M
|
GDSC1; GDSC2
|
1.495
|
3.3716
|
1.3144
|
17.4723
|
|
HCC1500
|
GDSC1; GDSC2; CTRP2
|
1.6149
|
4.3585
|
1.4641
|
23.3645
|
|
CAMA-1
|
GDSC1; GDSC2; CTRP2
|
1.6726
|
3.8321
|
1.6109
|
22.6721
|
|
CAL-51
|
GDSC1; GDSC2; CTRP2
|
1.7243
|
4.0375
|
1.6381
|
22.4118
|
|
BT-474
|
GDSC1; GDSC2; CTRP2
|
1.7366
|
3.6149
|
1.7016
|
22.1548
|
|
HCC1569
|
GDSC1; GDSC2
|
1.7471
|
4.2011
|
1.303
|
16.5325
|
|
Hs 578T
|
GDSC1; GDSC2; CTRP2
|
1.7756
|
4.2475
|
1.6588
|
22.1763
|
|
HCC1143
|
GDSC1; GDSC2; CTRP2
|
1.8104
|
4.0775
|
1.7243
|
21.8383
|
|
MDA-MB-175-VII
|
GDSC1; GDSC2
|
1.8175
|
4.09
|
1.4212
|
15.6487
|
|
UACC-812
|
GDSC2
|
1.836
|
4.4426
|
1.2997
|
16.2162
|
|
MDA-MB-231
|
GDSC1; GDSC2; CTRP2
|
1.906
|
3.7097
|
1.8709
|
21.0266
|
|
MFM-223
|
GDSC1; GDSC2
|
1.9097
|
3.1717
|
1.8189
|
13.2674
|
|
COLO 824
|
GDSC1; GDSC2
|
1.9341
|
4.5863
|
1.3418
|
15.6317
|
|
T-47D
|
GDSC1; GDSC2; CTRP2
|
1.9764
|
3.8637
|
1.9294
|
20.5982
|
|
HCC70
|
GDSC1; GDSC2
|
2.0483
|
4.0912
|
1.6662
|
13.492
|
|
CAL-120
|
GDSC1; GDSC2; CTRP2
|
2.1329
|
4.2883
|
2.0369
|
19.7243
|
|
HCC1806
|
GDSC1; GDSC2; CTRP2
|
2.1632
|
4.827
|
1.9567
|
19.9116
|
|
MDA-MB-157
|
GDSC1; GDSC2; CTRP2
|
2.1787
|
4.9203
|
1.95
|
19.8876
|
|
HCC1419
|
GDSC1; GDSC2; CTRP2
|
2.2265
|
4.3298
|
2.1306
|
19.1006
|
|
HMC-1-8
|
CTRP2
|
2.2916
|
3.4654
|
2.2854
|
18.3601
|
|
CAL-85-1
|
GDSC1; GDSC2; CTRP2
|
2.4688
|
5.462
|
2.1094
|
18.4338
|
|
JIMT-1
|
GDSC1; GDSC2; CTRP2
|
2.5233
|
3.9481
|
2.4973
|
16.8836
|
|
UACC-893
|
GDSC1; GDSC2
|
2.5496
|
5.2199
|
1.6717
|
11.7148
|
|
MDA-MB-415
|
GDSC1; GDSC2
|
2.5848
|
4.7875
|
1.9109
|
10.2694
|
|
MDA-MB-436
|
GDSC1; GDSC2
|
2.7328
|
5.5748
|
1.6734
|
11.1268
|
|
HDQ-P1
|
GDSC1; GDSC2
|
2.7467
|
4.9276
|
2.0007
|
9.2426
|
|
HCC202
|
GDSC1; GDSC2; CTRP2
|
2.8033
|
5.0461
|
2.5982
|
15.6479
|
|
BT-483
|
GDSC1; GDSC2
|
2.8723
|
6.192
|
1.4969
|
11.7028
|
|
HCC1954
|
GDSC1; GDSC2; CTRP2
|
2.9094
|
5.0202
|
2.721
|
14.8817
|
|
MDA-MB-468
|
GDSC1; GDSC2; CTRP2
|
2.9975
|
5.7645
|
2.5794
|
15.1442
|
|
ZR-75-30
|
GDSC1; GDSC2; CTRP2
|
3.0457
|
5.8507
|
2.5999
|
14.9294
|
|
MRK-nu-1
|
GDSC1; GDSC2
|
3.2203
|
6.6754
|
1.5598
|
10.3641
|
|
BT-20
|
GDSC1; GDSC2; CTRP2
|
3.3686
|
6.0075
|
2.8852
|
12.9356
|
|
HCC1395
|
GDSC1; GDSC2; CTRP2
|
3.4122
|
5.8307
|
3.0002
|
12.3739
|
|
BT-549
|
GDSC1; GDSC2; CTRP2
|
3.4784
|
6.4601
|
2.8148
|
12.9105
|
|
ZR-75-1
|
CTRP2
|
3.5322
|
6.3127
|
2.9332
|
12.3037
|
|
HCC38
|
GDSC1; GDSC2; CTRP2
|
3.7003
|
5.8833
|
3.2862
|
10.4767
|
|
HCC1937
|
GDSC1; GDSC2; CTRP2
|
4.0713
|
7.924
|
2.7306
|
11.7788
|
|
YMB-1-E
|
GDSC2
|
4.0885
|
7.973
|
1.6002
|
8.824
|
|
Hs 578Bst
|
CTRP2
|
5.1822
|
6.8738
|
4.2418
|
3.1167
|
|
BT 428
|
CTRP2
|
0.2829
|
6.4172
|
-0.5825
|
34.3596
|
|
BT 416
|
CTRP2
|
0.936
|
2.7973
|
0.9228
|
27.4149
|
|
BT 245
|
CTRP2
|
1.9665
|
4.3768
|
1.8413
|
20.9345
|
|
BT 145
|
CTRP2
|
2.1811
|
4.655
|
2.0167
|
19.643
|
|
BT 216
|
CTRP2
|
2.3903
|
8.9276
|
0.6093
|
24.1804
|
|
BT 187
|
CTRP2
|
2.854
|
7.6393
|
1.6583
|
19.0754
|
|
BT 333
|
CTRP2
|
3.0312
|
5.5615
|
2.6871
|
14.6436
|
|
BT 179
|
CTRP2
|
3.1822
|
4.6808
|
3.0986
|
12.6933
|
|
BT 172
|
CTRP2
|
3.2458
|
7.5462
|
2.0977
|
16.3448
|
|
BT 224
|
CTRP2
|
3.3517
|
5.9039
|
2.9076
|
12.8967
|
|
BT 286
|
CTRP2
|
3.44
|
9.8989
|
1.1779
|
19.4356
|
|
BT 330
|
CTRP2
|
3.5272
|
8.6963
|
1.8391
|
16.6685
|
|
BT 159
|
CTRP2
|
3.5844
|
7.4853
|
2.4648
|
14.0288
|
|
BT164
|
CTRP2
|
3.7195
|
6.5059
|
3.0439
|
11.3733
|
|
BT 139
|
CTRP2
|
3.9326
|
7.4362
|
2.8306
|
11.6979
|
|
BT 320
|
CTRP2
|
4.0012
|
5.7308
|
3.6657
|
8.1855
|
|
BT 112
|
CTRP2
|
4.0454
|
7.7582
|
2.7858
|
11.6132
|
|
BT 359
|
CTRP2
|
4.0455
|
8.2104
|
2.5675
|
12.5066
|
|
BT 231
|
CTRP2
|
4.2129
|
11.4586
|
1.1204
|
17.8761
|
|
BT 248
|
CTRP2
|
4.2493
|
8.9953
|
2.3727
|
12.8453
|
|
BT 504
|
CTRP2
|
4.2557
|
7.1683
|
3.2703
|
9.1333
|
|
BT 232
|
CTRP2
|
4.3675
|
6.9661
|
3.4741
|
8.0347
|
|
BT147
|
CTRP2
|
4.5069
|
7.2552
|
3.4644
|
7.7571
|
|
BT 239
|
CTRP2
|
4.577
|
10.3303
|
2.0025
|
13.6504
|
|
BT 131
|
CTRP2
|
4.7964
|
8.7318
|
2.9901
|
9.1225
|
|
BT 340
|
CTRP2
|
5.099
|
7.6645
|
3.7795
|
5.2122
|
|
BT 444
|
CTRP2
|
5.1459
|
8.0836
|
3.6086
|
5.842
|
|
BT 498
|
CTRP2
|
5.1852
|
7.2512
|
4.056
|
3.8905
|
|
BT 422
|
CTRP2
|
5.2599
|
15.1602
|
0.1649
|
19.0922
|
|
BT 271
|
CTRP2
|
5.5197
|
9.7261
|
3.0869
|
7.3514
|
|
BT 228
|
CTRP2
|
5.6526
|
13.0249
|
1.549
|
13.3484
|
|
BT 440
|
CTRP2
|
5.7779
|
7.7014
|
4.2661
|
2.0969
|
|
BT 328
|
CTRP2
|
5.9739
|
9.6581
|
3.4588
|
5.0694
|
|
BT 482
|
CTRP2
|
6.7225
|
21.2367
|
-1.6993
|
21.8835
|
| ----------- |
|
|
|
|
|
| ⏷ Show the Full List of 90 Cancer Drug Sensitivity Data of This Drug |
|
Cancer Drug Sensitivity Data Curated from 1 Cell Line(s) in Tongue Tissue
| Cell Line |
Original Dataset |
IC50 |
IC90 |
EC50 |
AUC |
|
YD-38
|
CTRP2
|
2.5584
|
4.0083
|
2.5285
|
16.6628
|
| ----------- |
|
|
|
|
|
Cancer Drug Sensitivity Data Curated from 67 Cell Line(s) in Central Nervous System Tissue
| Cell Line |
Original Dataset |
IC50 |
IC90 |
EC50 |
AUC |
|
D283 Med
|
GDSC1; GDSC2; CTRP2
|
-0.3388
|
0.9401
|
-0.3389
|
35.8639
|
|
D-423MG
|
GDSC1; GDSC2
|
-0.0364
|
2.1829
|
-0.1066
|
34.0448
|
|
D-566MG
|
GDSC1; GDSC2
|
0
|
1.7393
|
-0.0234
|
33.4524
|
|
PFSK-1
|
GDSC1; GDSC2
|
0.5098
|
2.5335
|
0.4267
|
28.6426
|
|
MOG-G-UVW
|
GDSC1; GDSC2
|
0.8445
|
3.1684
|
0.6586
|
25.8263
|
|
H4
|
GDSC1; GDSC2
|
0.9023
|
3.0558
|
0.749
|
25.0758
|
|
Daoy
|
GDSC1; GDSC2; CTRP2
|
1.2356
|
3.4194
|
1.1934
|
25.5168
|
|
SNU-201
|
CTRP2
|
1.4149
|
4.9644
|
1.105
|
25.2525
|
|
GI-1
|
GDSC1; GDSC2
|
1.4823
|
3.2006
|
1.3461
|
19.1735
|
|
AM-38
|
GDSC1; GDSC2
|
1.4837
|
2.7569
|
1.4371
|
18.6851
|
|
GB-1
|
GDSC1; GDSC2; CTRP2
|
1.5199
|
3.7627
|
1.4573
|
23.6923
|
|
SK-MG-1
|
GDSC1; GDSC2
|
1.5336
|
3.5429
|
1.3057
|
19.1593
|
|
SNU-466
|
CTRP2
|
1.6379
|
2.3587
|
1.6379
|
22.6943
|
|
SF268
|
GDSC1; GDSC2; CTRP2
|
1.6821
|
3.3666
|
1.6632
|
22.4634
|
|
LNZTA3WT4
|
GDSC1
|
1.7291
|
3.5582
|
1.5138
|
21.3384
|
|
MOG-G-CCM
|
GDSC1; GDSC2
|
1.7586
|
3.6192
|
1.5263
|
16.9334
|
|
D-245MG
|
GDSC2
|
1.7906
|
5.1856
|
0.9327
|
20.1952
|
|
ONS-76
|
GDSC1; GDSC2; CTRP2
|
1.8474
|
4.2885
|
1.729
|
21.7035
|
|
SF295
|
GDSC1; GDSC2; CTRP2; CTRP1
|
1.9443
|
3.5293
|
1.9288
|
21.5677
|
|
M059K
|
CTRP2
|
1.9642
|
4.2669
|
1.8588
|
20.8807
|
|
D-263MG
|
GDSC1; GDSC2
|
1.9916
|
4.2931
|
1.5247
|
15.949
|
|
LN-18
|
GDSC1; GDSC2; CTRP2
|
2.2107
|
3.6878
|
2.1911
|
18.9441
|
|
A-172
|
GDSC1; GDSC2; CTRP2
|
2.2502
|
4.6214
|
2.0992
|
19.1359
|
|
SF126
|
GDSC1; GDSC2; CTRP2
|
2.2865
|
3.9928
|
2.2432
|
18.5197
|
|
M059J
|
GDSC1; GDSC2
|
2.3725
|
4.5406
|
1.8124
|
12.7305
|
|
KS-1 [Human Krukenberg tumour]
|
GDSC1; GDSC2
|
2.4365
|
4.7535
|
1.7799
|
12.6825
|
|
SNU-1105
|
CTRP2
|
2.4671
|
4.3766
|
2.3818
|
17.4615
|
|
KG-1-C
|
CTRP2
|
2.4923
|
4.4246
|
2.4007
|
17.3156
|
|
SF539
|
GDSC1; GDSC2; CTRP2
|
2.5439
|
6.1951
|
1.9332
|
18.8726
|
|
YKG-1
|
GDSC1; GDSC2; CTRP2
|
2.5573
|
5.2076
|
2.2839
|
17.5315
|
|
SNU-738
|
CTRP2
|
2.5806
|
5.4439
|
2.2368
|
17.6342
|
|
Becker
|
GDSC1; GDSC2; CTRP2
|
2.5876
|
4.4598
|
2.4972
|
16.6766
|
|
U-251MG
|
GDSC1; GDSC2; CTRP2
|
2.6188
|
4.7248
|
2.4772
|
16.6487
|
|
Hs 683
|
GDSC1; GDSC2; CTRP2
|
2.6307
|
4.8461
|
2.4617
|
16.6669
|
|
42-MG-BA
|
GDSC1; GDSC2; CTRP2
|
2.6777
|
4.6922
|
2.5485
|
16.2127
|
|
GaMG
|
GDSC1; GDSC2; CTRP2; CTRP1
|
2.6829
|
4.8929
|
2.5317
|
17.1659
|
|
D-502MG
|
GDSC1; GDSC2
|
2.6948
|
5.8114
|
1.5225
|
13.3381
|
|
U-118MG
|
GDSC1; GDSC2; CTRP2
|
2.6957
|
5.9772
|
2.1773
|
17.521
|
|
TM-31
|
CTRP2
|
2.6962
|
4.4498
|
2.6162
|
15.9173
|
|
KINGS-1
|
GDSC1; GDSC2
|
2.7074
|
4.9128
|
1.9703
|
10.5286
|
|
SNU-626
|
CTRP2
|
2.7484
|
4.9558
|
2.5623
|
15.9448
|
|
YH-13
|
GDSC1; GDSC2; CTRP2
|
2.7647
|
5.5505
|
2.4019
|
16.4819
|
|
D-247MG
|
GDSC1; GDSC2
|
2.7859
|
5.5719
|
1.7241
|
11.7837
|
|
SNU-489
|
CTRP2
|
2.9291
|
5.9769
|
2.4279
|
15.9111
|
|
CAS-1
|
GDSC1; GDSC2
|
2.9484
|
4.5872
|
2.3563
|
7.2942
|
|
LN-229
|
GDSC1; GDSC2; CTRP2; CTRP1
|
2.9544
|
6.3552
|
2.3625
|
16.9932
|
|
Onda 10
|
GDSC1; GDSC2
|
3.009
|
4.9834
|
2.2179
|
7.9563
|
|
LN-405
|
GDSC1; GDSC2
|
3.0189
|
5.5688
|
1.9369
|
9.6889
|
|
DK-MG
|
GDSC1; GDSC2; CTRP2
|
3.0666
|
5.9046
|
2.6023
|
14.8611
|
|
D-392MG
|
GDSC1; GDSC2
|
3.0683
|
4.7574
|
2.3832
|
6.7244
|
|
D-336MG
|
GDSC1; GDSC2
|
3.1314
|
4.5317
|
2.5518
|
5.4601
|
|
SW1088
|
GDSC1; GDSC2; CTRP2
|
3.1577
|
5.2702
|
2.9196
|
13.4115
|
|
U-87MG ATCC
|
GDSC1; GDSC2; CTRP2
|
3.2423
|
5.626
|
2.893
|
13.2627
|
|
T98G
|
GDSC1; GDSC2; CTRP2
|
3.2483
|
5.1333
|
3.0593
|
12.6302
|
|
KNS-81-FD
|
GDSC1; GDSC2
|
3.4285
|
5.9633
|
2.0889
|
7.5006
|
|
DBTRG-05MG
|
GDSC1; GDSC2; CTRP2
|
3.4318
|
5.9872
|
2.9604
|
12.4715
|
|
KALS-1
|
GDSC1; GDSC2; CTRP2
|
3.4846
|
7.3495
|
2.4274
|
14.4291
|
|
GOS-3
|
CTRP2
|
3.6166
|
6.7149
|
2.8465
|
12.4226
|
|
Onda 11
|
GDSC1; GDSC2
|
3.6292
|
7.3555
|
1.5551
|
10.2848
|
|
SNB-75
|
GDSC1; GDSC2; CTRP2
|
3.7166
|
7.5916
|
2.5461
|
13.3755
|
|
D-542MG
|
GDSC1; GDSC2
|
4.0447
|
8.1636
|
1.4739
|
9.6973
|
|
NMC-G1
|
GDSC1; GDSC2; CTRP2
|
4.0836
|
6.5722
|
3.383
|
9.0897
|
|
GMS-10
|
CTRP2
|
4.1027
|
6.3876
|
3.4864
|
8.6265
|
|
8-MG-BA
|
GDSC1; GDSC2; CTRP2
|
4.1935
|
5.9952
|
3.7543
|
7.3256
|
|
SW1783
|
GDSC1; GDSC2; CTRP2
|
4.6861
|
9.1137
|
2.7039
|
10.5428
|
|
KNS-42
|
GDSC1; GDSC2; CTRP2
|
4.9958
|
7.8733
|
3.5893
|
6.2107
|
|
CCF-STTG1
|
CTRP2
|
5.502
|
7.4643
|
4.1913
|
2.7931
|
| ----------- |
|
|
|
|
|
| ⏷ Show the Full List of 67 Cancer Drug Sensitivity Data of This Drug |
|
Cancer Drug Sensitivity Data Curated from 3 Cell Line(s) in Digestive System Tissue
| Cell Line |
Original Dataset |
IC50 |
IC90 |
EC50 |
AUC |
|
TGBC24TKB
|
GDSC1; GDSC2
|
1.6818
|
3.1583
|
1.5739
|
17.0216
|
|
ETK-1
|
GDSC1; GDSC2
|
2.1766
|
5.7069
|
1.0787
|
17.8593
|
|
TGBC1TKB
|
GDSC1; GDSC2
|
2.7761
|
4.3785
|
2.2905
|
8.3125
|
| ----------- |
|
|
|
|
|
Cancer Drug Sensitivity Data Curated from 23 Cell Line(s) in Endometrium Tissue
| Cell Line |
Original Dataset |
IC50 |
IC90 |
EC50 |
AUC |
|
HEC-6
|
CTRP2; CTRP1
|
-0.1549
|
2.2597
|
-0.1715
|
35.4032
|
|
MFE-319
|
GDSC1; GDSC2; CTRP2; CTRP1
|
0.4043
|
2.6086
|
0.3877
|
31.7192
|
|
JHUEM-2
|
CTRP2; CTRP1
|
0.5755
|
6.0886
|
-0.1125
|
32.7123
|
|
RL95-2
|
GDSC1; GDSC2; CTRP2
|
1.1096
|
2.6462
|
1.1046
|
26.231
|
|
HEC-59
|
CTRP2; CTRP1
|
1.3494
|
3.7981
|
1.2817
|
25.6626
|
|
HEC-1-A
|
CTRP2
|
1.5078
|
3.8665
|
1.431
|
23.8214
|
|
HEC-151
|
CTRP2; CTRP1
|
1.9295
|
3.7759
|
1.8963
|
21.7242
|
|
KLE
|
GDSC1; GDSC2; CTRP2; CTRP1
|
2.0846
|
5.3977
|
1.746
|
21.7665
|
|
HEC-1-B
|
CTRP2
|
2.0961
|
3.8153
|
2.0608
|
19.7609
|
|
HEC-265
|
CTRP2; CTRP1
|
2.1285
|
3.6592
|
2.112
|
20.3571
|
|
JHUEM-3
|
CTRP2
|
2.1773
|
4.0253
|
2.1234
|
19.2834
|
|
ESS-1
|
GDSC1; GDSC2; CTRP2
|
2.3868
|
4.8722
|
2.1892
|
18.3923
|
|
HEC-108
|
CTRP2; CTRP1
|
2.3945
|
4.5135
|
2.2956
|
18.8837
|
|
HEC-251
|
CTRP2; CTRP1
|
2.419
|
5.7669
|
1.9955
|
19.8772
|
|
MFE-280
|
GDSC1; GDSC2
|
2.4333
|
4.397
|
1.9383
|
11.7405
|
|
EN
|
GDSC1; GDSC2; CTRP2
|
2.8141
|
5.813
|
2.3643
|
16.479
|
|
JHUEM-1
|
CTRP2
|
2.8188
|
4.3602
|
2.7624
|
15.0187
|
|
MFE-296
|
GDSC1; GDSC2; CTRP2
|
2.8245
|
6.1782
|
2.2395
|
16.9211
|
|
AN3-CA
|
GDSC1; GDSC2; CTRP2
|
2.8438
|
5.1561
|
2.6114
|
15.4773
|
|
EFE-184
|
CTRP2
|
3.1316
|
5.6717
|
2.7573
|
14.0903
|
|
JHUEM-7
|
CTRP2
|
3.3472
|
5.5125
|
3.0461
|
12.3865
|
|
HEC-50B
|
CTRP2
|
4.6937
|
7.3418
|
3.5918
|
6.8182
|
|
Ishikawa (Heraklio) 02 ER-
|
CTRP2; CTRP1
|
-0.09
|
1.8326
|
-0.0941
|
34.9353
|
| ----------- |
|
|
|
|
|
| ⏷ Show the Full List of 23 Cancer Drug Sensitivity Data of This Drug |
|
Cancer Drug Sensitivity Data Curated from 40 Cell Line(s) in Kidney Tissue
| Cell Line |
Original Dataset |
IC50 |
IC90 |
EC50 |
AUC |
|
LB1047-RCC
|
GDSC1; GDSC2
|
0.6081
|
2.0224
|
0.5893
|
27.3284
|
|
CAL-54
|
GDSC1; GDSC2; CTRP2
|
0.9089
|
5.2059
|
0.4861
|
28.9856
|
|
KTCTL-21
|
GDSC1; GDSC2
|
1.19
|
7.0804
|
-0.5349
|
30.0071
|
|
KTCTL-140
|
GDSC1; GDSC2
|
1.3746
|
3.4163
|
1.1702
|
20.6221
|
|
LB2241-RCC
|
GDSC1; GDSC2
|
1.4875
|
3.8949
|
1.1386
|
20.2916
|
|
SW13
|
GDSC1; GDSC2
|
1.5942
|
2.8985
|
1.5328
|
17.654
|
|
KTCTL-195
|
GDSC1; GDSC2
|
1.6322
|
4.2272
|
1.1706
|
19.4871
|
|
TUHR14TKB
|
CTRP2
|
1.67
|
5.4593
|
1.2407
|
23.9859
|
|
KTCTL-13
|
GDSC1; GDSC2
|
1.7147
|
3.3428
|
1.5622
|
16.9327
|
|
TUHR4TKB
|
CTRP2
|
1.7372
|
4.5791
|
1.5508
|
22.6754
|
|
A-498
|
GDSC1; GDSC2
|
1.7616
|
3.8251
|
1.4593
|
17.2948
|
|
RXF 393L
|
GDSC1; GDSC2
|
1.8309
|
4.0508
|
1.4505
|
17.0474
|
|
BB65-RCC
|
GDSC1; GDSC2
|
1.8411
|
3.9699
|
1.492
|
16.7674
|
|
NCC021
|
GDSC1; GDSC2
|
1.8561
|
4.2413
|
1.4028
|
17.2157
|
|
TK-10
|
GDSC1; GDSC2
|
2.0655
|
4.1549
|
1.6588
|
14.8643
|
|
SN12C
|
GDSC1; GDSC2
|
2.0674
|
4.4994
|
1.5172
|
15.6852
|
|
KTCTL-1M
|
GDSC1; GDSC2
|
2.0796
|
3.9639
|
1.7494
|
14.282
|
|
VMRC-RCW
|
GDSC1; GDSC2; CTRP2; CTRP1
|
2.1281
|
4.461
|
2.0174
|
20.6793
|
|
SW156
|
GDSC1; GDSC2
|
2.1432
|
4.8094
|
1.4592
|
15.729
|
|
KTCTL-26A
|
GDSC1; GDSC2
|
2.1798
|
3.595
|
1.9914
|
12.4755
|
|
769-P
|
GDSC1; GDSC2; CTRP2
|
2.2084
|
4.0438
|
2.1542
|
19.0773
|
|
BFTC-909
|
GDSC1; GDSC2; CTRP2
|
2.2449
|
4.3826
|
2.1411
|
19.0057
|
|
A-704
|
GDSC1; GDSC2
|
2.274
|
4.4136
|
1.7677
|
13.382
|
|
NCC010
|
GDSC1; GDSC2
|
2.353
|
4.9679
|
1.598
|
14.0965
|
|
UO-31
|
GDSC1; GDSC2; CTRP2
|
2.5199
|
4.8985
|
2.3277
|
17.4871
|
|
VMRC-RCZ
|
GDSC1; GDSC2; CTRP2
|
2.5203
|
4.0383
|
2.4854
|
16.9338
|
|
Caki-1
|
GDSC1; GDSC2; CTRP2
|
2.6012
|
5.5053
|
2.2393
|
17.5641
|
|
KMRC-20
|
GDSC1; GDSC2; CTRP2
|
2.6527
|
4.9597
|
2.4569
|
16.6162
|
|
HA7-RCC
|
GDSC1; GDSC2
|
2.6666
|
4.6286
|
2.0657
|
10.0889
|
|
ACHN
|
GDSC1; GDSC2; CTRP2
|
2.7685
|
4.7293
|
2.6391
|
15.6089
|
|
LB996-RCC
|
GDSC2
|
2.7996
|
4.8971
|
2.0657
|
9.6148
|
|
SNU-1272
|
CTRP2
|
2.8039
|
7.2698
|
1.7686
|
18.7843
|
|
786-O
|
GDSC1; GDSC2; CTRP2
|
2.8521
|
5.0433
|
2.6522
|
15.3044
|
|
OS-RC-2
|
GDSC1; GDSC2; CTRP2
|
3.0117
|
6.4453
|
2.3323
|
16.0466
|
|
KMRC-3
|
CTRP2
|
3.1114
|
7.3739
|
2.0387
|
16.9239
|
|
Caki-2
|
CTRP2
|
3.1147
|
5.7021
|
2.7282
|
14.2485
|
|
RCC10RGB
|
GDSC1; GDSC2; CTRP2; CTRP1
|
3.1183
|
5.9362
|
2.6945
|
15.2909
|
|
TUHR10TKB
|
CTRP2
|
3.3115
|
4.1983
|
3.3006
|
11.5815
|
|
KMRC-1
|
GDSC1; GDSC2; CTRP2
|
3.446
|
5.4785
|
3.1648
|
11.6563
|
|
KMRC-2
|
CTRP2
|
4.0572
|
8.9283
|
2.2288
|
13.8626
|
| ----------- |
|
|
|
|
|
| ⏷ Show the Full List of 40 Cancer Drug Sensitivity Data of This Drug |
|
Cancer Drug Sensitivity Data Curated from 63 Cell Line(s) in Large Intestine Tissue
| Cell Line |
Original Dataset |
IC50 |
IC90 |
EC50 |
AUC |
|
COLO 320HSR
|
GDSC1; GDSC2
|
0.0884
|
1.7893
|
0.065
|
32.5655
|
|
DiFi
|
GDSC1; GDSC2
|
0.1206
|
1.8077
|
0.0972
|
32.2426
|
|
NCI-H508
|
GDSC1; GDSC2; CTRP2; CTRP1
|
0.1292
|
2.2538
|
0.1192
|
33.5101
|
|
RCM-1 [Human ESC]
|
GDSC1; GDSC2; CTRP2; CTRP1
|
0.3034
|
1.5851
|
0.3032
|
32.3301
|
|
HCT 116
|
GDSC1; GDSC2; CTRP2; CTRP1
|
0.3383
|
2.5334
|
0.3232
|
32.1491
|
|
CL-34
|
CTRP1
|
0.366
|
2.4964
|
0.353
|
67.8628
|
|
LoVo
|
GDSC1; GDSC2; CTRP2; CTRP1
|
0.4675
|
2.4713
|
0.4575
|
31.281
|
|
COLO 320
|
CTRP2; CTRP1
|
0.6394
|
5.1011
|
0.2215
|
30.7374
|
|
COLO205
|
GDSC1; GDSC2; CTRP2; CTRP1
|
0.6892
|
1.832
|
0.689
|
29.7877
|
|
CCK-81
|
GDSC1; GDSC2; CTRP2
|
0.7726
|
2.7693
|
0.7562
|
28.5141
|
|
SW948
|
GDSC1; GDSC2; CTRP2; CTRP1
|
0.8392
|
2.3716
|
0.8366
|
28.8071
|
|
SNU-175
|
GDSC2; CTRP2; CTRP1
|
0.9402
|
2.7028
|
0.9324
|
28.1587
|
|
HT115
|
GDSC1; GDSC2; CTRP2; CTRP1
|
0.9483
|
2.4726
|
0.9453
|
28.0897
|
|
SW626
|
GDSC1; GDSC2
|
0.9563
|
2.9484
|
0.8318
|
24.3831
|
|
LS411N
|
GDSC1; GDSC2; CTRP2; CTRP1
|
1.0129
|
2.8075
|
1.0034
|
27.6853
|
|
CW-2
|
GDSC1; GDSC2; CTRP2; CTRP1
|
1.0232
|
3.5671
|
0.9626
|
27.7878
|
|
C2BBe1
|
GDSC1; GDSC2; CTRP2; CTRP1
|
1.0912
|
2.7645
|
1.0844
|
27.1606
|
|
HT-55
|
GDSC1; GDSC2; CTRP2; CTRP1
|
1.13
|
2.6539
|
1.1261
|
26.8951
|
|
SNU-C5
|
GDSC1; GDSC2; CTRP2; CTRP1
|
1.1435
|
3.9433
|
1.0428
|
27.1303
|
|
SW48
|
GDSC1; GDSC2; CTRP2; CTRP1
|
1.1903
|
2.7129
|
1.1861
|
26.4987
|
|
GP2d
|
CTRP2; CTRP1
|
1.2515
|
2.9654
|
1.2418
|
26.114
|
|
HCT 8
|
CTRP2; CTRP1
|
1.4137
|
2.9332
|
1.408
|
25.0314
|
|
CL-11
|
GDSC1; GDSC2; CTRP2
|
1.4375
|
3.2738
|
1.4155
|
24.1036
|
|
OUMS-23
|
CTRP2
|
1.46
|
2.3832
|
1.4598
|
23.8806
|
|
CaR-1
|
GDSC1; GDSC2
|
1.4766
|
3.54
|
1.2445
|
19.7528
|
|
MDST8
|
GDSC1; GDSC2; CTRP2; CTRP1
|
1.4985
|
2.8392
|
1.4957
|
24.463
|
|
SNU-1033
|
CTRP2
|
1.5456
|
3.303
|
1.5257
|
23.3763
|
|
LS513
|
GDSC1; CTRP2; CTRP1
|
1.6363
|
3.6091
|
1.6033
|
23.6557
|
|
SW1463
|
GDSC1; GDSC2; CTRP2
|
1.6389
|
3.7901
|
1.5803
|
22.8859
|
|
SNU-C1
|
GDSC1; GDSC2; CTRP2
|
1.6491
|
3.0287
|
1.6434
|
22.639
|
|
LS123
|
GDSC1; GDSC2; CTRP2; CTRP1
|
1.7328
|
4.0915
|
1.6518
|
23.182
|
|
T84
|
GDSC1; GDSC2; CTRP2
|
1.7383
|
3.6524
|
1.6999
|
22.155
|
|
SNU-1040
|
CTRP1
|
1.7503
|
3.9511
|
1.6888
|
49.4513
|
|
LS180
|
GDSC1; GDSC2; CTRP2; CTRP1
|
1.7552
|
3.272
|
1.746
|
22.7926
|
|
NCI-H747
|
GDSC1; GDSC2; CTRP2
|
1.7623
|
3.6373
|
1.7266
|
21.9862
|
|
COLO201
|
CTRP2
|
1.9021
|
2.5626
|
1.9021
|
20.9345
|
|
SNU-61
|
GDSC1; GDSC2; CTRP2; CTRP1
|
1.9074
|
3.8484
|
1.8663
|
21.8963
|
|
SW620
|
GDSC1; GDSC2; CTRP2; CTRP1
|
1.909
|
3.6767
|
1.8828
|
21.8361
|
|
SNU-C2A
|
CTRP2; CTRP1
|
1.9433
|
5.3068
|
1.6179
|
22.6478
|
|
DLD-1
|
CTRP2; CTRP1
|
1.9787
|
3.9014
|
1.9361
|
21.4317
|
|
SW480
|
CTRP1
|
1.9998
|
4.4406
|
1.8827
|
46.1956
|
|
SW1116
|
GDSC1; GDSC2
|
2.0362
|
3.4734
|
1.8747
|
13.7628
|
|
HT-29
|
GDSC1; GDSC2; CTRP2; CTRP1
|
2.0587
|
4.8039
|
1.8726
|
21.3982
|
|
SNU-407
|
GDSC1; GDSC2; CTRP2
|
2.0678
|
4.5963
|
1.9064
|
20.3866
|
|
SK-CO-1
|
GDSC1; GDSC2; CTRP2; CTRP1
|
2.0685
|
3.9717
|
2.0236
|
20.8475
|
|
GP5d
|
GDSC1; GDSC2
|
2.1018
|
3.6209
|
1.8982
|
13.3402
|
|
SNU-C2B
|
GDSC1; GDSC2
|
2.1684
|
5.1323
|
1.3382
|
16.3523
|
|
SW837
|
GDSC1; GDSC2
|
2.1828
|
3.869
|
1.8959
|
13.0044
|
|
KM12
|
GDSC1; GDSC2; CTRP2; CTRP1
|
2.2117
|
4.4418
|
2.111
|
20.0941
|
|
SNU-503
|
CTRP2
|
2.2897
|
5.3028
|
1.9638
|
19.5009
|
|
SNU-81
|
GDSC1; GDSC2; CTRP1
|
2.3061
|
4.3568
|
2.2264
|
24.8388
|
|
HCC-56
|
CTRP2
|
2.4167
|
3.536
|
2.4109
|
17.5254
|
|
RKO
|
GDSC1; GDSC2; CTRP2; CTRP1
|
2.5493
|
5.4312
|
2.2429
|
18.5973
|
|
SNU-283
|
GDSC2
|
2.5906
|
3.8285
|
2.3451
|
8.7037
|
|
HCT 15
|
GDSC1; GDSC2; CTRP2
|
2.5927
|
4.5068
|
2.4941
|
16.6712
|
|
SW1417
|
GDSC1; GDSC2; CTRP2
|
2.6782
|
3.9151
|
2.6616
|
15.8195
|
|
CL-40
|
CTRP1
|
2.68
|
4.5737
|
2.5929
|
36.7044
|
|
HCC2998
|
GDSC1; GDSC2
|
2.7123
|
5.1896
|
1.8416
|
11.3065
|
|
COLO 678
|
GDSC1; GDSC2; CTRP2; CTRP1
|
2.9143
|
5.3768
|
2.6571
|
16.0203
|
|
NCI-H716
|
GDSC1; GDSC2; CTRP2
|
2.9354
|
6.3976
|
2.271
|
16.4923
|
|
SNU-C4
|
CTRP2; CTRP1
|
3.0837
|
4.7198
|
3.0026
|
14.2826
|
|
LS1034
|
GDSC1; GDSC2; CTRP2
|
3.1007
|
4.268
|
3.074
|
13.0394
|
|
SW403
|
CTRP1
|
3.6803
|
6.6073
|
3.0257
|
26.7779
|
| ----------- |
|
|
|
|
|
| ⏷ Show the Full List of 63 Cancer Drug Sensitivity Data of This Drug |
|
Cancer Drug Sensitivity Data Curated from 1 Cell Line(s) in Leukemia Tissue
| Cell Line |
Original Dataset |
IC50 |
IC90 |
EC50 |
AUC |
|
MO-T
|
GDSC1; GDSC2
|
0.3977
|
3.3039
|
0.1355
|
30.6609
|
| ----------- |
|
|
|
|
|
Cancer Drug Sensitivity Data Curated from 24 Cell Line(s) in Liver Tissue
| Cell Line |
Original Dataset |
IC50 |
IC90 |
EC50 |
AUC |
|
JHH-1
|
GDSC1; GDSC2; CTRP2
|
-0.2367
|
0.9293
|
-0.2367
|
35.1835
|
|
SNU-182
|
CTRP2
|
0.0728
|
3.8252
|
-0.1036
|
33.6927
|
|
Hep-G2
|
CTRP2; CTRP1
|
0.5503
|
3.5712
|
0.4605
|
30.9989
|
|
HuH-6
|
CTRP2
|
0.7299
|
3.2854
|
0.6743
|
60.5529
|
|
Huh-7
|
GDSC1; GDSC2
|
1.345
|
3.048
|
1.2326
|
20.423
|
|
Hep 3B2.1-7
|
GDSC1; GDSC2; CTRP2
|
1.4517
|
3.4599
|
1.4165
|
24.0537
|
|
HLE
|
GDSC1; GDSC2
|
1.5382
|
6.348
|
0.15
|
25.4345
|
|
Li-7
|
CTRP2
|
1.9117
|
5.9542
|
1.3401
|
22.9027
|
|
JHH-5
|
CTRP2
|
1.932
|
4.7561
|
1.7219
|
21.4629
|
|
SNU-398
|
GDSC1; GDSC2; CTRP1
|
2.0301
|
5.4753
|
1.6636
|
28.4083
|
|
JHH-7
|
GDSC1; GDSC2
|
2.0855
|
3.5626
|
1.9
|
13.4024
|
|
JHH-2
|
GDSC1; GDSC2; CTRP2
|
2.1022
|
4.3546
|
1.9922
|
19.9775
|
|
HuH-1
|
GDSC1; GDSC2; CTRP2
|
2.139
|
3.8926
|
2.0982
|
19.494
|
|
JHH-4
|
GDSC1; GDSC2; CTRP2
|
2.1721
|
3.7322
|
2.1476
|
19.2179
|
|
SNU-886
|
CTRP2
|
2.2375
|
3.9752
|
2.1933
|
18.849
|
|
SNU-423
|
GDSC1; GDSC2; CTRP2
|
2.4159
|
4.4524
|
2.3131
|
17.8633
|
|
Hep-G2/C3A
|
GDSC1; GDSC2
|
2.5143
|
3.9875
|
2.1993
|
9.869
|
|
SNU-449
|
GDSC1; GDSC2; CTRP2; CTRP1
|
2.6272
|
5.2393
|
2.3832
|
17.8611
|
|
SNU-387
|
GDSC1; GDSC2; CTRP2
|
2.6447
|
4.7653
|
2.4962
|
16.5008
|
|
PLC/PRF/5
|
CTRP2
|
2.7594
|
4.183
|
2.7224
|
15.3479
|
|
SK-HEP-1
|
GDSC1; GDSC2; CTRP2
|
2.9087
|
5.1222
|
2.6919
|
14.9892
|
|
HLF
|
CTRP2
|
2.9956
|
4.2418
|
2.9658
|
13.7497
|
|
JHH-6
|
GDSC1; GDSC2; CTRP2; CTRP1
|
5.1287
|
7.6954
|
3.8986
|
5.6061
|
|
SNU-475
|
CTRP2
|
5.5813
|
8.4043
|
3.7882
|
4.3298
|
| ----------- |
|
|
|
|
|
| ⏷ Show the Full List of 24 Cancer Drug Sensitivity Data of This Drug |
|
Cancer Drug Sensitivity Data Curated from 226 Cell Line(s) in Lung Tissue
| Cell Line |
Original Dataset |
IC50 |
IC90 |
EC50 |
AUC |
|
COR-L51
|
CTRP1
|
-3.1625
|
-0.1682
|
-3.1684
|
93.9782
|
|
SCLC-21H
|
CTRP1
|
-2.579
|
0.2422
|
-2.5849
|
92.1086
|
|
DMS 454
|
CTRP2
|
-1.3276
|
4.8106
|
-5.5703
|
15.9442
|
|
HCC33
|
GDSC2; CTRP1
|
-0.6318
|
0.7709
|
-0.6319
|
49.2829
|
|
NCI-H290
|
GDSC1; GDSC2
|
-0.6002
|
1.5691
|
-0.6367
|
39.5221
|
|
LC-1/sq
|
GDSC1; GDSC2
|
-0.42
|
2.053
|
-0.4982
|
37.9051
|
|
LB647-SCLC
|
GDSC1; GDSC2
|
-0.3096
|
1.8138
|
-0.3537
|
36.6529
|
|
COR-L321
|
GDSC1; GDSC2
|
-0.3045
|
0.9908
|
-0.307
|
36.4036
|
|
NCI-H524
|
GDSC1; GDSC2
|
-0.2876
|
1.1252
|
-0.2923
|
36.2454
|
|
NCI-H1105
|
GDSC1; GDSC2; CTRP2; CTRP1
|
-0.2816
|
0.9918
|
-0.2817
|
36.1849
|
|
NCI-H209
|
GDSC1; GDSC2
|
-0.264
|
1.586
|
-0.2875
|
36.0992
|
|
NCI-H146
|
GDSC1; GDSC2; CTRP2
|
-0.1701
|
1.1534
|
-0.1704
|
34.7407
|
|
MOR/CPR
|
CTRP2
|
-0.166
|
0.9297
|
-0.166
|
34.7123
|
|
NCI-H2171
|
GDSC2; CTRP2
|
-0.1494
|
1.0184
|
-0.1494
|
34.6019
|
|
SBC-3
|
GDSC1; GDSC2
|
-0.1152
|
2.2894
|
-0.206
|
34.929
|
|
Lu-139
|
GDSC1
|
-0.0209
|
1.8364
|
-0.0527
|
41.9434
|
|
NCI-H2009
|
GDSC1; GDSC2; CTRP2; CTRP1
|
0.0128
|
2.1718
|
0.003
|
34.2764
|
|
NCI-H250
|
GDSC2
|
0.0452
|
1.6969
|
0.0262
|
32.9772
|
|
EMC-BAC-1
|
GDSC1; GDSC2
|
0.0512
|
2.8424
|
-0.1289
|
33.6922
|
|
COR-L303
|
GDSC2
|
0.0709
|
1.6336
|
0.0563
|
32.6978
|
|
NCI-H64
|
GDSC1; GDSC2
|
0.0941
|
1.1819
|
0.0926
|
32.399
|
|
NCI-H1963
|
GDSC1; GDSC2; CTRP1
|
0.1114
|
1.4542
|
0.1111
|
43.0133
|
|
NCI-H1694
|
GDSC1; GDSC2; CTRP2; CTRP1
|
0.1346
|
1.5297
|
0.1341
|
33.4433
|
|
A-427
|
GDSC1; GDSC2
|
0.1677
|
2.9888
|
-0.0354
|
32.6487
|
|
NCI-H211
|
GDSC1; GDSC2; CTRP2
|
0.1806
|
1.1391
|
0.1806
|
32.4033
|
|
NCI-H810
|
GDSC1; GDSC2; CTRP2; CTRP1
|
0.1908
|
1.6149
|
0.1903
|
33.073
|
|
Ms-1
|
GDSC1; GDSC2
|
0.2225
|
1.6933
|
0.2096
|
31.1681
|
|
Sq-1
|
CTRP1
|
0.2959
|
2.5354
|
0.2796
|
68.5427
|
|
NCI-H1341
|
GDSC1; GDSC2; CTRP2
|
0.3278
|
1.5496
|
0.3276
|
31.4234
|
|
IST-SL2
|
GDSC1; GDSC2
|
0.3443
|
2.4176
|
0.2671
|
30.2704
|
|
NCI-H2106
|
CTRP2
|
0.3558
|
2.1925
|
0.3497
|
31.2566
|
|
NCI-H847
|
GDSC1; GDSC2
|
0.3744
|
1.1792
|
0.3741
|
29.5801
|
|
NCI-H187
|
GDSC1
|
0.3979
|
1.248
|
0.3975
|
36.5525
|
|
NCI-H2227
|
GDSC1; GDSC2
|
0.4006
|
1.9773
|
0.3765
|
29.4377
|
|
COR-L88
|
GDSC1; GDSC2; CTRP1
|
0.4405
|
1.9092
|
0.4394
|
40.24
|
|
CPC-N
|
GDSC1; GDSC2
|
0.4616
|
1.5427
|
0.4587
|
28.7183
|
|
NCI-H1876
|
GDSC1; GDSC2; CTRP1
|
0.4633
|
2.4611
|
0.4535
|
40.0814
|
|
NCI-H2126
|
CTRP2; CTRP1
|
0.4678
|
2.2054
|
0.4639
|
31.2587
|
|
IST-SL1
|
GDSC1; GDSC2
|
0.5102
|
1.7309
|
0.5032
|
28.2515
|
|
COR-L311
|
GDSC2
|
0.5143
|
1.8209
|
0.5038
|
28.2276
|
|
DMS 273
|
GDSC1; GDSC2; CTRP2; CTRP1
|
0.5522
|
4.5792
|
0.284
|
31.567
|
|
SBC-1
|
GDSC1; GDSC2
|
0.5698
|
1.6642
|
0.5659
|
27.6374
|
|
NCI-H835
|
GDSC1; GDSC2
|
0.5918
|
1.6243
|
0.589
|
27.4113
|
|
NCI-H748
|
GDSC2
|
0.6254
|
1.9534
|
0.6116
|
27.1299
|
|
NCI-H510A
|
GDSC1; GDSC2
|
0.627
|
2.3743
|
0.5751
|
27.3078
|
|
VMRC-LCD
|
GDSC1; GDSC2; CTRP2; CTRP1
|
0.652
|
2.5843
|
0.6422
|
30.0646
|
|
NCI-H1770
|
GDSC1; GDSC2
|
0.6558
|
2.164
|
0.6281
|
26.8949
|
|
NCI-H526
|
GDSC1; GDSC2; CTRP2
|
0.6584
|
1.9303
|
0.6578
|
29.222
|
|
COR-L279
|
GDSC1; GDSC2; CTRP1
|
0.6688
|
2.2
|
0.6668
|
38.3177
|
|
NCI-H2081
|
GDSC1; GDSC2; CTRP2; CTRP1
|
0.6726
|
2.5308
|
0.6648
|
29.9221
|
|
NCI-H720
|
GDSC1; GDSC2
|
0.6952
|
3.5468
|
0.3909
|
27.9303
|
|
NCI-H1930
|
CTRP1
|
0.6987
|
1.3049
|
0.6987
|
64.4362
|
|
NCI-H345
|
GDSC1; GDSC2
|
0.7001
|
2.8105
|
0.5823
|
26.9149
|
|
NCI-H82
|
GDSC1; GDSC2; CTRP1
|
0.7554
|
2.6749
|
0.7448
|
37.6224
|
|
COR-L23
|
GDSC1; GDSC2; CTRP1
|
0.8013
|
2.8435
|
0.7853
|
37.2567
|
|
NCI-H69
|
GDSC1; GDSC2; CTRP2
|
0.8107
|
2.1621
|
0.8095
|
28.2094
|
|
COR-L32
|
GDSC2
|
0.8443
|
3.0239
|
0.6932
|
25.6453
|
|
NCI-H1734
|
GDSC1; GDSC2; CTRP2
|
0.8533
|
3.6454
|
0.7627
|
28.2273
|
|
NCI-H889
|
CTRP2
|
0.8638
|
2.56
|
0.857
|
27.8743
|
|
NCI-H1184
|
CTRP2
|
0.8738
|
2.8435
|
0.8568
|
27.842
|
|
HCC78
|
GDSC1; GDSC2; CTRP2; CTRP1
|
0.8841
|
3.2223
|
0.8482
|
28.622
|
|
LXF 289
|
GDSC1; GDSC2; CTRP2; CTRP1
|
0.9139
|
3.1888
|
0.8818
|
28.4128
|
|
Lu-134-A
|
GDSC1; GDSC2
|
0.9212
|
2.6745
|
0.8461
|
24.4765
|
|
NCI-H446
|
GDSC1; GDSC2; CTRP2
|
0.9383
|
3.052
|
0.9117
|
27.4446
|
|
HCC1833
|
CTRP2; CTRP1
|
0.9954
|
4.6879
|
0.7417
|
28.6202
|
|
NCI-H1435
|
GDSC1; GDSC2; CTRP2; CTRP1
|
1.0105
|
2.7675
|
1.0022
|
27.6972
|
|
NCI-H23
|
GDSC1; GDSC2; CTRP2; CTRP1
|
1.0111
|
3.2653
|
0.9774
|
27.7776
|
|
HCC15
|
GDSC1; GDSC2; CTRP2; CTRP1
|
1.0304
|
3.7392
|
0.95
|
27.8068
|
|
NCI-H1437
|
GDSC1; GDSC2; CTRP1
|
1.0647
|
4.4894
|
0.8605
|
35.7655
|
|
EBC-1
|
GDSC1; GDSC2; CTRP2; CTRP1
|
1.0917
|
3.208
|
1.0653
|
27.2223
|
|
IA-LM
|
GDSC1; GDSC2
|
1.0932
|
2.9912
|
0.9713
|
22.9979
|
|
SBC-5
|
GDSC1; GDSC2; CTRP2
|
1.0977
|
1.9684
|
1.0976
|
26.2939
|
|
A-549
|
GDSC1; GDSC2; CTRP2; CTRP1
|
1.1293
|
3.0895
|
1.1111
|
26.9471
|
|
HCC44
|
GDSC1; GDSC2; CTRP2; CTRP1
|
1.1566
|
4.1535
|
1.0236
|
27.1532
|
|
NCI-H1581
|
GDSC1; GDSC2; CTRP2
|
1.1743
|
1.7277
|
1.1743
|
25.7829
|
|
NCI-H1385
|
CTRP2
|
1.1818
|
4.5274
|
0.9581
|
26.4989
|
|
NCI-H2795
|
GDSC1; GDSC2
|
1.1974
|
3.4689
|
0.9616
|
22.5652
|
|
NCI-H1688
|
GDSC1; GDSC2
|
1.2046
|
3.2292
|
1.0359
|
22.1306
|
|
NCI-H1568
|
GDSC1; GDSC2; CTRP2; CTRP1
|
1.2517
|
4.3003
|
1.1002
|
26.5902
|
|
NCI-H2818
|
GDSC1; GDSC2
|
1.2617
|
3.3408
|
1.0683
|
21.692
|
|
NCI-H1436
|
GDSC1; GDSC2
|
1.2772
|
2.9714
|
1.1755
|
21.0458
|
|
EMC-BAC-2
|
GDSC1; GDSC2
|
1.2778
|
3.3432
|
1.0852
|
21.5268
|
|
NCI-H1836
|
GDSC1; GDSC2; CTRP1
|
1.289
|
3.5717
|
1.2423
|
33.2718
|
|
NCI-H1975
|
GDSC1; GDSC2; CTRP1
|
1.3227
|
3.2255
|
1.3034
|
32.8735
|
|
COLO 699
|
CTRP2; CTRP1
|
1.3504
|
4.9026
|
1.0776
|
26.3571
|
|
NCI-H1666
|
GDSC1; GDSC2; CTRP2; CTRP1
|
1.3599
|
5.0751
|
1.0442
|
26.4425
|
|
NCI-H740
|
GDSC2
|
1.3838
|
3.4554
|
1.1688
|
20.5875
|
|
NCI-H2369
|
GDSC1; GDSC2
|
1.4084
|
3.3693
|
1.2206
|
20.1931
|
|
NCI-H661
|
GDSC1; GDSC2; CTRP2; CTRP1
|
1.4119
|
2.8912
|
1.4072
|
25.0402
|
|
HOP-92
|
GDSC1; GDSC2; CTRP2
|
1.4514
|
4.3476
|
1.2891
|
24.4927
|
|
NCI-H2373
|
GDSC1; GDSC2
|
1.4557
|
3.1101
|
1.339
|
19.336
|
|
HCC827
|
GDSC1; GDSC2; CTRP2; CTRP1
|
1.4558
|
4.6187
|
1.2578
|
25.4069
|
|
Lu-135
|
GDSC1; GDSC2
|
1.4674
|
3.3148
|
1.3
|
19.4912
|
|
DV-90
|
CTRP2; CTRP1
|
1.496
|
3.9272
|
1.4214
|
24.7202
|
|
NCI-H1651
|
GDSC1; GDSC2
|
1.4998
|
3.1216
|
1.3843
|
18.8869
|
|
UMC-11
|
GDSC1; GDSC2
|
1.5159
|
2.4464
|
1.5041
|
18.1832
|
|
NCI-H1155
|
GDSC1; GDSC2; CTRP2
|
1.5215
|
3.035
|
1.5133
|
23.4976
|
|
NCI-H2073
|
CTRP1
|
1.5442
|
4.491
|
1.3802
|
52.037
|
|
NCI-H1092
|
GDSC1; GDSC2; CTRP2
|
1.5497
|
5.1118
|
1.2119
|
24.4572
|
|
RERF-LC-A1
|
CTRP2
|
1.6115
|
4.1385
|
1.5007
|
23.2482
|
|
LK-2
|
GDSC1; GDSC2
|
1.6177
|
2.4742
|
1.608
|
17.1508
|
|
Calu-3
|
GDSC1; GDSC2; CTRP2; CTRP1
|
1.6188
|
4.5893
|
1.4411
|
24.2645
|
|
NCI-H1048
|
GDSC1; GDSC2; CTRP1
|
1.6321
|
4.2569
|
1.5181
|
30.6645
|
|
NCI-H3122
|
GDSC1; GDSC2
|
1.641
|
3.8447
|
1.3222
|
18.592
|
|
NCI-H2122
|
GDSC1; GDSC2; CTRP2; CTRP1
|
1.6452
|
4.3257
|
1.5204
|
23.9085
|
|
NCI-H1915
|
GDSC1; GDSC2; CTRP2; CTRP1
|
1.6613
|
3.9198
|
1.5982
|
23.5923
|
|
ABC-1
|
GDSC1; GDSC2
|
1.6904
|
4.4575
|
1.1405
|
19.414
|
|
NCI-H1755
|
GDSC1; GDSC2; CTRP2; CTRP1
|
1.6956
|
4.0993
|
1.6109
|
23.4393
|
|
COLO 668
|
GDSC1; GDSC2; CTRP2; CTRP1
|
1.709
|
4.4102
|
1.5741
|
23.5229
|
|
HCC1588
|
CTRP2
|
1.7257
|
3.7459
|
1.6773
|
22.2729
|
|
LCLC-103H
|
GDSC1; GDSC2; CTRP2; CTRP1
|
1.7286
|
3.1192
|
1.7234
|
22.955
|
|
NCI-H2804
|
GDSC1; GDSC2
|
1.734
|
4.3404
|
1.2339
|
18.6964
|
|
DMS 79
|
GDSC1; GDSC2
|
1.7485
|
2.8092
|
1.7127
|
15.9725
|
|
DMS 53
|
GDSC1; GDSC2; CTRP2
|
1.778
|
3.6968
|
1.7375
|
21.8979
|
|
NCI-H2170
|
GDSC1; GDSC2; CTRP2
|
1.8117
|
3.7077
|
1.7719
|
21.6706
|
|
PC-14
|
GDSC1; GDSC2
|
1.8209
|
3.3693
|
1.6706
|
15.8575
|
|
Lu-99
|
CTRP2; CTRP1
|
1.8316
|
5.5039
|
1.4387
|
23.6202
|
|
LCLC-97TM1
|
GDSC1; GDSC2; CTRP2; CTRP1
|
1.8438
|
4.0381
|
1.7774
|
22.4007
|
|
SW1573
|
GDSC1; GDSC2
|
1.8462
|
3.3654
|
1.6994
|
15.5847
|
|
LC-2/ad
|
GDSC1; GDSC2
|
1.8483
|
3.1147
|
1.7652
|
15.2201
|
|
HCC2108
|
CTRP2; CTRP1
|
1.8501
|
3.912
|
1.7991
|
22.3073
|
|
NCI-H358
|
GDSC1; GDSC2; CTRP2; CTRP1
|
1.8538
|
3.9669
|
1.7969
|
22.3029
|
|
HOP-62
|
GDSC1; GDSC2; CTRP2
|
1.8547
|
5.2217
|
1.5134
|
22.4458
|
|
NCI-H1648
|
GDSC1; GDSC2
|
1.9205
|
4.3513
|
1.426
|
16.8147
|
|
LK2
|
CTRP2
|
1.9425
|
2.4938
|
1.9425
|
20.6651
|
|
NCI-H1944
|
GDSC1; GDSC2; CTRP2; CTRP1
|
1.9643
|
3.6241
|
1.944
|
21.4518
|
|
RERF-LC-KJ
|
GDSC1; GDSC2; CTRP1
|
1.9799
|
3.9218
|
1.9353
|
27.4376
|
|
NCI-H292
|
GDSC1; GDSC2; CTRP2
|
1.9943
|
4.6819
|
1.8072
|
20.9666
|
|
NCI-H522
|
GDSC2; CTRP2; CTRP1
|
2.0074
|
3.9943
|
1.9564
|
21.271
|
|
HCC1195
|
CTRP2; CTRP1
|
2.0178
|
3.9235
|
1.9751
|
21.1744
|
|
NCI-H196
|
GDSC1; GDSC2; CTRP2
|
2.037
|
3.1139
|
2.0349
|
20.043
|
|
NCI-H841
|
GDSC1; GDSC2; CTRP2; CTRP1
|
2.0387
|
3.9638
|
1.993
|
21.0469
|
|
NCI-H2731
|
GDSC1; GDSC2
|
2.0411
|
3.4097
|
1.8992
|
13.6058
|
|
RERF-LC-Sq1
|
GDSC1; GDSC2
|
2.0568
|
6.238
|
0.7106
|
20.4247
|
|
HCC2279
|
CTRP2
|
2.0612
|
3.6682
|
2.0368
|
19.9565
|
|
CAL-12T
|
GDSC1; GDSC2; CTRP2; CTRP1
|
2.0762
|
5.1604
|
1.8033
|
21.588
|
|
SHP-77
|
GDSC1; GDSC2; CTRP2; CTRP1
|
2.0862
|
4.4833
|
1.9682
|
20.9805
|
|
DMS 114
|
GDSC1; GDSC2; CTRP2
|
2.0885
|
4.0385
|
2.0272
|
19.9003
|
|
NCI-H2110
|
GDSC1; GDSC2; CTRP2; CTRP1
|
2.1349
|
4.9398
|
1.9237
|
20.9845
|
|
NCI-H2029
|
GDSC1; GDSC2; CTRP2; CTRP1
|
2.1479
|
4.1201
|
2.0905
|
20.3667
|
|
HCC366
|
GDSC1; GDSC2; CTRP2
|
2.1655
|
3.9677
|
2.1178
|
19.3409
|
|
BEN
|
GDSC1; GDSC2; CTRP2; CTRP1
|
2.1741
|
3.9081
|
2.141
|
20.1122
|
|
KNS-62
|
GDSC1; GDSC2; CTRP2; CTRP1
|
2.1848
|
5.4545
|
1.8385
|
21.1358
|
|
SW1271
|
GDSC1; GDSC2
|
2.1949
|
4.4863
|
1.6547
|
14.365
|
|
EPLC-272H
|
GDSC1; GDSC2; CTRP2; CTRP1
|
2.2272
|
4.8436
|
2.0469
|
20.2685
|
|
PaCa-3
|
GDSC1; GDSC2
|
2.2371
|
3.6433
|
2.0369
|
11.9691
|
|
HCC1359
|
CTRP1
|
2.2597
|
4.9628
|
2.0542
|
43.0269
|
|
NCI-H2286
|
CTRP2; CTRP1
|
2.267
|
4.6236
|
2.1372
|
19.8303
|
|
NCI-H2141
|
GDSC1; GDSC2; CTRP2; CTRP1
|
2.2945
|
4.8727
|
2.1135
|
19.8278
|
|
LOU-NH91
|
GDSC1; GDSC2; CTRP2
|
2.3073
|
4.2066
|
2.2373
|
18.4727
|
|
NCI-H650
|
GDSC1; GDSC2; CTRP2; CTRP1
|
2.3309
|
5.6301
|
1.9438
|
20.3232
|
|
NCI-H1838
|
GDSC1; GDSC2; CTRP2
|
2.3812
|
5.0783
|
2.1287
|
18.6265
|
|
NCI-H2444
|
GDSC1; GDSC2
|
2.3977
|
4.7335
|
1.7504
|
13.007
|
|
Calu-6
|
GDSC1; GDSC2; CTRP2; CTRP1
|
2.4175
|
4.8932
|
2.2426
|
18.9966
|
|
HCC1438
|
CTRP2
|
2.4556
|
4.7599
|
2.2915
|
17.8147
|
|
RERF-LC-MS
|
GDSC1; GDSC2; CTRP2
|
2.4663
|
4.0255
|
2.4297
|
17.2991
|
|
NCI-H596
|
GDSC1; GDSC2; CTRP1
|
2.4765
|
4.4026
|
2.4022
|
23.3777
|
|
COR-L95
|
GDSC1; CTRP2
|
2.5012
|
3.7696
|
2.4873
|
16.9898
|
|
COR-L105
|
GDSC1; GDSC2; CTRP2
|
2.5111
|
5.8686
|
2.0177
|
18.6497
|
|
SK-MES-1
|
GDSC1; GDSC2; CTRP2
|
2.5168
|
4.746
|
2.3613
|
17.377
|
|
NCI-H1623
|
GDSC1; GDSC2; CTRP2
|
2.5316
|
4.325
|
2.4598
|
16.9851
|
|
NCI-H322M
|
GDSC1; GDSC2
|
2.5476
|
7.2039
|
0.7031
|
18.7027
|
|
LC-1/sq-SF
|
CTRP2; CTRP1
|
2.5955
|
3.7304
|
2.589
|
17.2467
|
|
NCI-H1792
|
GDSC1; GDSC2; CTRP2; CTRP1
|
2.6277
|
5.7684
|
2.2216
|
18.4441
|
|
NCI-H2087
|
GDSC1; GDSC2; CTRP2
|
2.6315
|
5.2077
|
2.3647
|
17.014
|
|
NCI-H520
|
GDSC1; GDSC2; CTRP2
|
2.639
|
4.8927
|
2.4592
|
16.6503
|
|
NCI-H838
|
GDSC1; GDSC2; CTRP2
|
2.6436
|
5.2548
|
2.364
|
16.9803
|
|
HCC95
|
CTRP2
|
2.6747
|
4.0834
|
2.6437
|
15.8917
|
|
NCI-H2595
|
GDSC1; GDSC2
|
2.6785
|
4.9154
|
1.9412
|
10.8106
|
|
NCI-H2030
|
GDSC1; GDSC2; CTRP2; CTRP1
|
2.691
|
4.6301
|
2.5947
|
16.9219
|
|
HCC4006
|
CTRP2; CTRP1
|
2.7087
|
5.3253
|
2.4479
|
17.3853
|
|
Lu-99A
|
GDSC1; GDSC2
|
2.7176
|
7.0374
|
0.9393
|
16.7717
|
|
NCI-H1373
|
CTRP2; CTRP1
|
2.7232
|
5.3316
|
2.4618
|
17.2918
|
|
NCI-H1693
|
GDSC1; GDSC2; CTRP2
|
2.7263
|
4.5953
|
2.6215
|
15.8034
|
|
NCI-H2066
|
GDSC1; GDSC2
|
2.7265
|
4.5299
|
2.1708
|
9.2268
|
|
NCI-H1573
|
GDSC1; GDSC2; CTRP2; CTRP1
|
2.7442
|
5.0367
|
2.5642
|
16.8637
|
|
HCC2935
|
CTRP2; CTRP1
|
2.7478
|
5.114
|
2.5485
|
16.9083
|
|
HARA [Human squamous cell lung carcinoma]
|
GDSC1; GDSC2; CTRP2; CTRP1
|
2.762
|
7.1559
|
1.837
|
19.5024
|
|
T3M-10
|
CTRP2; CTRP1
|
2.7647
|
6.2093
|
2.2144
|
18.0783
|
|
NCI-H647
|
GDSC1; GDSC2; CTRP2
|
2.7798
|
4.5974
|
2.679
|
15.433
|
|
ChaGo-K-1
|
GDSC1; GDSC2; CTRP2; CTRP1
|
2.7806
|
5.3667
|
2.5142
|
16.9325
|
|
NCI-H1339
|
CTRP2
|
2.8245
|
4.4981
|
2.746
|
15.0575
|
|
NCI-H2461
|
GDSC1; GDSC2
|
2.8431
|
5.5428
|
1.791
|
11.1759
|
|
NCI-H2810
|
GDSC1; GDSC2
|
2.8611
|
5.3725
|
1.8911
|
10.4916
|
|
NCI-H2196
|
GDSC2; CTRP2
|
2.8744
|
5.4815
|
2.5439
|
15.6337
|
|
NCI-H460
|
GDSC1; GDSC2; CTRP2; CTRP1
|
2.9179
|
5.2477
|
2.698
|
15.8627
|
|
NCI-H322
|
CTRP2
|
2.9203
|
5.0927
|
2.7129
|
14.8779
|
|
NCI-H1650
|
GDSC1; GDSC2; CTRP2
|
2.9232
|
5.3594
|
2.6363
|
15.1491
|
|
NCI-H1299
|
GDSC1; GDSC2; CTRP2; CTRP1
|
2.9513
|
5.5669
|
2.6389
|
15.9771
|
|
NCI-H2405
|
GDSC1; GDSC2; CTRP2; CTRP1
|
3.0107
|
5.9718
|
2.5657
|
16.0752
|
|
NCI-H1869
|
GDSC1; GDSC2; CTRP2
|
3.0331
|
4.6782
|
2.9374
|
13.7284
|
|
NCI-H2803
|
GDSC1; GDSC2
|
3.0697
|
5.3681
|
2.0817
|
8.6168
|
|
LUDLU-1
|
CTRP2
|
3.0758
|
4.6003
|
2.999
|
13.3781
|
|
NCI-H2085
|
GDSC1; GDSC2; CTRP2
|
3.0787
|
6.8138
|
2.2489
|
16.1886
|
|
NCI-H1703
|
GDSC1; GDSC2; CTRP2
|
3.116
|
4.8027
|
3.0006
|
13.2458
|
|
NCI-H2347
|
GDSC1; GDSC2
|
3.1522
|
5.2292
|
2.2235
|
7.4642
|
|
NCI-H2291
|
GDSC1; GDSC2; CTRP2
|
3.1574
|
5.8493
|
2.7203
|
14.1565
|
|
NCI-H2591
|
GDSC1; GDSC2
|
3.1876
|
6.1434
|
1.7978
|
10.0388
|
|
NCI-H513
|
GDSC1; GDSC2
|
3.2051
|
6.0316
|
1.8686
|
9.5411
|
|
NCI-H441
|
GDSC1; GDSC2; CTRP2; CTRP1
|
3.2228
|
5.5945
|
2.9252
|
14.1395
|
|
NCI-H2342
|
GDSC1; GDSC2; CTRP2; CTRP1
|
3.2541
|
5.2519
|
3.0638
|
13.545
|
|
NCI-H1781
|
GDSC1; GDSC2; CTRP1
|
3.2586
|
4.4603
|
3.2286
|
16.5878
|
|
NCI-H1563
|
GDSC1; GDSC2; CTRP2
|
3.2633
|
7.2206
|
2.2616
|
15.653
|
|
HCC1171
|
CTRP2
|
3.2931
|
5.9104
|
2.8425
|
13.3095
|
|
NCI-H1304
|
GDSC1
|
3.3834
|
6.2976
|
1.885
|
11.0743
|
|
NCI-H2722
|
GDSC1; GDSC2
|
3.3863
|
5.3124
|
2.3798
|
5.7801
|
|
NCI-H2869
|
GDSC1; GDSC2
|
3.4011
|
5.5708
|
2.2628
|
6.4792
|
|
NCI-H1355
|
GDSC1; GDSC2; CTRP2; CTRP1
|
3.4195
|
7.4734
|
2.3785
|
15.6942
|
|
NCI-H727
|
GDSC1; GDSC2; CTRP2; CTRP1
|
3.4233
|
7.5012
|
2.37
|
15.7175
|
|
NCI-H2023
|
GDSC1; GDSC2; CTRP2; CTRP1
|
3.4261
|
5.8258
|
3.0659
|
13.0359
|
|
Lu-165
|
GDSC1; GDSC2
|
3.4694
|
5.4216
|
2.3923
|
5.4709
|
|
SK-LU-1
|
GDSC1; GDSC2; CTRP2; CTRP1
|
3.5254
|
6.0679
|
3.0822
|
12.6967
|
|
NCI-H1993
|
GDSC1; GDSC2
|
3.5566
|
5.2557
|
2.543
|
4.29
|
|
NCI-H2172
|
GDSC1; GDSC2; CTRP2; CTRP1
|
3.5733
|
6.2815
|
3.0489
|
12.6918
|
|
NCI-H3255
|
GDSC1; GDSC2; CTRP1
|
3.5779
|
5.3253
|
3.396
|
14.5767
|
|
NCI-H2228
|
GDSC1; GDSC2
|
3.7068
|
7.9799
|
1.3054
|
11.5965
|
|
NCI-H1793
|
GDSC1; GDSC2; CTRP2; CTRP1
|
3.7646
|
6.4518
|
3.1793
|
11.6779
|
|
Lu-65
|
GDSC1; GDSC2; CTRP2; CTRP1
|
3.7788
|
7.0902
|
2.9149
|
12.6754
|
|
NCI-H2135
|
GDSC1; GDSC2
|
3.8213
|
11.5113
|
-0.3644
|
20.2049
|
|
Calu-1
|
CTRP2
|
3.8929
|
8.4511
|
2.3068
|
13.9238
|
|
NCI-H226
|
GDSC1; GDSC2
|
3.9487
|
8.2591
|
1.3542
|
10.6621
|
|
EKVX
|
GDSC1; GDSC2; CTRP2
|
4.0734
|
7.7631
|
2.8102
|
11.4473
|
|
RERF-LC-Ad2
|
CTRP2
|
4.5381
|
7.4572
|
3.3938
|
7.981
|
|
NCI-H1395
|
CTRP2
|
4.6321
|
7.8953
|
3.2623
|
8.3259
|
|
RERF-LC-Ad1
|
CTRP2
|
5.025
|
7.1498
|
3.9755
|
4.5306
|
|
Hs 888.Lu
|
CTRP2
|
3.4842
|
7.4114
|
2.3988
|
14.544
|
| ----------- |
|
|
|
|
|
| ⏷ Show the Full List of 226 Cancer Drug Sensitivity Data of This Drug |
|
Cancer Drug Sensitivity Data Curated from 1 Cell Line(s) in Lymphoma Tissue
| Cell Line |
Original Dataset |
IC50 |
IC90 |
EC50 |
AUC |
|
CBTCi002-A
|
GDSC1; GDSC2
|
1.2934
|
3.151
|
1.1524
|
21.0929
|
| ----------- |
|
|
|
|
|
Cancer Drug Sensitivity Data Curated from 3 Cell Line(s) in Thigh Tissue
| Cell Line |
Original Dataset |
IC50 |
IC90 |
EC50 |
AUC |
|
SYO-1
|
CTRP2
|
1.0225
|
2.7244
|
1.014
|
26.8228
|
|
P-STS
|
CTRP2
|
1.1982
|
2.1737
|
1.1981
|
25.6243
|
|
H-STS
|
CTRP2
|
1.4098
|
2.4131
|
1.4095
|
24.2153
|
| ----------- |
|
|
|
|
|
Cancer Drug Sensitivity Data Curated from 31 Cell Line(s) in Soft Tissue Tissue
| Cell Line |
Original Dataset |
IC50 |
IC90 |
EC50 |
AUC |
|
Yamato-SS
|
CTRP2
|
1.4134
|
2.9682
|
1.4051
|
24.2179
|
|
Aska-SS
|
CTRP2
|
1.7556
|
2.3317
|
1.7556
|
21.9101
|
|
Rh30
|
GDSC1; GDSC2
|
-1.1489
|
1.1121
|
-1.1753
|
44.9491
|
|
Rh41
|
GDSC1; GDSC2; CTRP2
|
0.2441
|
1.9066
|
0.2415
|
31.9888
|
|
Hs 633.T
|
GDSC1; GDSC2
|
0.4959
|
2.4622
|
0.4229
|
28.7303
|
|
VA-ES-BJ
|
GDSC1; GDSC2
|
0.7628
|
4.5918
|
0.1027
|
29.0572
|
|
EW-8
|
GDSC1; GDSC2
|
0.7695
|
1.8703
|
0.7634
|
25.6443
|
|
G-401
|
GDSC1; GDSC2; CTRP2; CTRP1
|
1.1677
|
3.4093
|
1.1295
|
26.7608
|
|
KYM-1
|
GDSC1; GDSC2; CTRP2
|
1.1865
|
3.0218
|
1.1701
|
25.7567
|
|
Rh18
|
GDSC1; CTRP2
|
1.2574
|
3.4675
|
1.2118
|
25.383
|
|
MFH-ino
|
GDSC1; GDSC2
|
1.695
|
6.1945
|
0.3767
|
23.6406
|
|
STS-0421
|
GDSC1; GDSC2
|
1.7098
|
3.7166
|
1.4409
|
17.6252
|
|
RD
|
GDSC1; GDSC2; CTRP2
|
1.7509
|
4.2665
|
1.6287
|
22.3593
|
|
HT-1080
|
GDSC1; GDSC2; CTRP2; CTRP1
|
1.8434
|
4.4644
|
1.7093
|
22.6352
|
|
SW872
|
GDSC1; GDSC2
|
1.9286
|
3.1472
|
1.8452
|
14.4164
|
|
TE 617.T
|
CTRP2
|
2.0097
|
4.3381
|
1.8951
|
20.6095
|
|
GCT
|
GDSC1; GDSC2; CTRP2
|
2.0874
|
5.3341
|
1.7338
|
20.9451
|
|
SW982
|
GDSC1; GDSC2; CTRP2
|
2.1428
|
3.8198
|
2.1094
|
19.4435
|
|
G-402
|
GDSC1; GDSC2; CTRP2
|
2.1528
|
6.1317
|
1.537
|
21.4742
|
|
A-204
|
GDSC1; GDSC2; CTRP2
|
2.1607
|
5.5758
|
1.7377
|
20.7119
|
|
TE 441.T
|
GDSC2; CTRP2
|
2.248
|
4.4547
|
2.131
|
19.0312
|
|
Tm87-16
|
CTRP2
|
2.3208
|
6.3481
|
1.6365
|
20.6207
|
|
TTC-709
|
CTRP2
|
2.5098
|
5.715
|
2.0699
|
18.4595
|
|
RKN
|
GDSC1; GDSC2; CTRP2; CTRP1
|
2.6371
|
4.4837
|
2.5619
|
17.2044
|
|
MES-SA
|
GDSC1; GDSC2; CTRP2
|
2.7592
|
4.5534
|
2.6653
|
15.5459
|
|
BT-12
|
CTRP2
|
2.8254
|
5.0933
|
2.6092
|
15.5411
|
|
SK-LMS-1
|
GDSC1; GDSC2; CTRP2
|
2.9355
|
5.4046
|
2.6354
|
15.1158
|
|
SK-UT-1
|
GDSC1; GDSC2; CTRP2
|
3.5422
|
8.3111
|
2.0367
|
15.8467
|
|
SW684
|
GDSC1; GDSC2
|
3.5535
|
6.059
|
2.1413
|
6.8271
|
|
Hs 729.T
|
CTRP2
|
3.7158
|
9.0552
|
1.847
|
16.1868
|
|
BT-16
|
CTRP2
|
4.0692
|
6.5807
|
3.3647
|
9.1998
|
| ----------- |
|
|
|
|
|
| ⏷ Show the Full List of 31 Cancer Drug Sensitivity Data of This Drug |
|
Cancer Drug Sensitivity Data Curated from 48 Cell Line(s) in Ovary Tissue
| Cell Line |
Original Dataset |
IC50 |
IC90 |
EC50 |
AUC |
|
2004
|
CTRP2
|
2.7703
|
5.0009
|
2.5743
|
15.8346
|
|
OVK18
|
GDSC1; GDSC2; CTRP2; CTRP1
|
-0.3851
|
2.7984
|
-0.4449
|
37.0526
|
|
JHOC-5
|
CTRP2; CTRP1
|
0.1248
|
2.6997
|
0.0946
|
33.6051
|
|
RMUG-S
|
CTRP1
|
0.195
|
2.6448
|
0.1705
|
69.3308
|
|
OVCAR-5
|
GDSC1; GDSC2; CTRP2
|
0.5379
|
3.5321
|
0.4416
|
30.3454
|
|
COV434
|
CTRP2; CTRP1
|
0.5643
|
3.1827
|
0.5169
|
30.7669
|
|
A2780
|
GDSC1; GDSC2; CTRP2; CTRP1
|
0.7357
|
3.5406
|
0.6612
|
29.7275
|
|
EFO-27
|
GDSC1; GDSC2; CTRP2; CTRP1
|
0.8272
|
3.6004
|
0.7511
|
29.1306
|
|
OVCAR-8
|
GDSC1; GDSC2; CTRP2; CTRP1
|
0.8467
|
3.047
|
0.8214
|
28.8329
|
|
ONCO-DG-1
|
CTRP2
|
0.9379
|
3.5511
|
0.8649
|
27.604
|
|
OV-90
|
GDSC1; GDSC2; CTRP2; CTRP1
|
1.0151
|
4.0561
|
0.8875
|
28.066
|
|
TYK-nu
|
GDSC1; GDSC2; CTRP2; CTRP1
|
1.0293
|
3.7492
|
0.9475
|
27.8189
|
|
COV644
|
CTRP2; CTRP1
|
1.1443
|
4.289
|
0.9861
|
27.3197
|
|
OC 314
|
GDSC1; GDSC2; CTRP2
|
1.3595
|
2.5628
|
1.3582
|
24.5534
|
|
RMG-I
|
GDSC1; GDSC2; CTRP2; CTRP1
|
1.5048
|
3.8774
|
1.4371
|
24.6388
|
|
SNU-8
|
CTRP2
|
1.5772
|
2.9857
|
1.5715
|
23.1177
|
|
IGROV-1
|
GDSC1; GDSC2; CTRP2; CTRP1
|
1.688
|
3.774
|
1.6426
|
23.3564
|
|
OAW28
|
GDSC1; GDSC2; CTRP2
|
1.7749
|
4.0853
|
1.685
|
22.0877
|
|
ES-2
|
GDSC1; GDSC2
|
1.7832
|
2.9115
|
1.7343
|
15.6927
|
|
TOV-112D
|
GDSC1; GDSC2; CTRP2
|
1.8931
|
4.6554
|
1.7032
|
21.65
|
|
SNU-840
|
CTRP2; CTRP1
|
1.9537
|
5.3595
|
1.6143
|
22.6289
|
|
HEY A8
|
CTRP2; CTRP1
|
2.0806
|
4.4243
|
1.9727
|
20.9828
|
|
ES2
|
CTRP2
|
2.1032
|
3.631
|
2.0833
|
19.6613
|
|
OVCAR-4
|
GDSC1; GDSC2; CTRP2; CTRP1
|
2.1217
|
5.99
|
1.5983
|
22.1876
|
|
MCAS
|
CTRP2
|
2.2064
|
4.1204
|
2.1418
|
19.1263
|
|
SK-OV-3
|
GDSC1; GDSC2; CTRP2; CTRP1
|
2.3924
|
4.4029
|
2.3118
|
18.8351
|
|
FU-OV-1
|
GDSC1; GDSC2; CTRP2
|
2.4509
|
5.3857
|
2.1139
|
18.4705
|
|
Caov-4
|
GDSC1; GDSC2
|
2.5334
|
4.4259
|
2.0269
|
10.8227
|
|
OVTOKO
|
GDSC1; GDSC2; CTRP2
|
2.634
|
4.6949
|
2.5004
|
16.5192
|
|
DOV13
|
GDSC1; GDSC2; CTRP2
|
2.6656
|
5.3873
|
2.3472
|
16.9762
|
|
OVKATE
|
GDSC1; GDSC2; CTRP1
|
2.7091
|
3.7429
|
2.7045
|
21.1149
|
|
JHOM-1
|
CTRP2; CTRP1
|
2.825
|
4.2675
|
2.7911
|
15.8259
|
|
OVCAR-3
|
GDSC1; GDSC2
|
2.8583
|
4.5986
|
2.2654
|
8.1715
|
|
OVISE
|
GDSC1; GDSC2; CTRP2
|
2.8614
|
5.3828
|
2.5617
|
15.6068
|
|
Kuramochi
|
GDSC1; GDSC2; CTRP2
|
2.9847
|
5.7718
|
2.5631
|
15.2423
|
|
OVMANA
|
CTRP1
|
3.1894
|
6.6785
|
2.4828
|
33.4957
|
|
OAW42
|
GDSC1; GDSC2; CTRP2; CTRP1
|
3.1949
|
5.2346
|
3.004
|
13.9364
|
|
OV7
|
CTRP2; CTRP1
|
3.3331
|
8.578
|
1.7868
|
18.2107
|
|
Caov-3
|
GDSC2; CTRP2; CTRP1
|
3.563
|
7.7452
|
2.4002
|
15.2471
|
|
TOV-21G
|
GDSC1; GDSC2; CTRP2
|
3.6163
|
6.8107
|
2.8036
|
12.594
|
|
JHOS-2
|
GDSC1; GDSC2; CTRP2
|
3.7356
|
7.2165
|
2.7393
|
12.5498
|
|
OVSAHO
|
CTRP2
|
3.7624
|
6.5657
|
3.0615
|
11.1919
|
|
COV318
|
CTRP1
|
3.9071
|
7.6936
|
2.7657
|
27.1964
|
|
JHOS-4
|
GDSC1; GDSC2; CTRP1
|
3.957
|
11.1906
|
1.1203
|
23.9472
|
|
EFO-21
|
GDSC1; GDSC2; CTRP2; CTRP1
|
4.1463
|
6.6161
|
3.5002
|
9.4363
|
|
SNU-119
|
CTRP2
|
4.1899
|
8.9173
|
2.3567
|
13.0429
|
|
COV362
|
CTRP2; CTRP1
|
4.9004
|
7.4719
|
3.8114
|
6.4307
|
|
OV56
|
GDSC1; GDSC2; CTRP2
|
4.971
|
6.3456
|
4.3317
|
3.1405
|
| ----------- |
|
|
|
|
|
| ⏷ Show the Full List of 48 Cancer Drug Sensitivity Data of This Drug |
|
Cancer Drug Sensitivity Data Curated from 1 Cell Line(s) in Clivus Tissue
| Cell Line |
Original Dataset |
IC50 |
IC90 |
EC50 |
AUC |
|
U-CH1
|
CTRP2
|
3.7345
|
5.3954
|
3.5056
|
9.5467
|
| ----------- |
|
|
|
|
|
Cancer Drug Sensitivity Data Curated from 8 Cell Line(s) in Pleura Tissue
| Cell Line |
Original Dataset |
IC50 |
IC90 |
EC50 |
AUC |
|
MSTO-211H
|
GDSC1; GDSC2; CTRP2; CTRP1
|
1.4423
|
5.1756
|
1.1074
|
25.9684
|
|
NCI-H28
|
GDSC1; GDSC2; CTRP2; CTRP1
|
1.8439
|
4.0515
|
1.7757
|
22.4065
|
|
MPP 89
|
GDSC1; GDSC2; CTRP2
|
2.5593
|
5.4376
|
2.2156
|
17.7753
|
|
NCI-H2052
|
GDSC1; GDSC2; CTRP2; CTRP1
|
2.6535
|
5.6493
|
2.2887
|
18.1242
|
|
NCI-H2452
|
GDSC1; GDSC2
|
2.8199
|
5.0176
|
2.0264
|
9.7879
|
|
IST-Mes1
|
GDSC1; GDSC2; CTRP2
|
3.1226
|
5.8475
|
2.6837
|
14.3938
|
|
JL-1
|
CTRP2
|
3.3551
|
5.0971
|
3.1861
|
11.8472
|
|
ACC-MESO-1
|
CTRP2
|
3.8708
|
5.5265
|
3.6058
|
8.7786
|
| ----------- |
|
|
|
|
|
| ⏷ Show the Full List of 8 Cancer Drug Sensitivity Data of This Drug |
|
Cancer Drug Sensitivity Data Curated from 7 Cell Line(s) in Prostate Tissue
| Cell Line |
Original Dataset |
IC50 |
IC90 |
EC50 |
AUC |
|
NCI-H660
|
GDSC2; CTRP2
|
0.412
|
3.0461
|
0.3627
|
31.0261
|
|
22Rv1
|
GDSC1; GDSC2; CTRP2
|
1.0869
|
3.411
|
1.0378
|
26.5307
|
|
VCaP
|
GDSC1; GDSC2; CTRP2
|
1.5191
|
3.3723
|
1.4937
|
23.5711
|
|
LNCaP clone FGC
|
GDSC1; GDSC2; CTRP2
|
2.0113
|
4.4856
|
1.8684
|
20.6981
|
|
PC-3
|
GDSC1; GDSC2
|
2.5578
|
6.4592
|
1.0784
|
16.4948
|
|
DU145
|
GDSC1; GDSC2; CTRP2
|
2.6242
|
4.8842
|
2.4451
|
16.746
|
|
PaCa-3
|
CTRP2; CTRP1
|
4.3668
|
6.2435
|
3.8909
|
7.3365
|
| ----------- |
|
|
|
|
|
| ⏷ Show the Full List of 7 Cancer Drug Sensitivity Data of This Drug |
|
Cancer Drug Sensitivity Data Curated from 2 Cell Line(s) in Salivary Gland Tissue
| Cell Line |
Original Dataset |
IC50 |
IC90 |
EC50 |
AUC |
|
YD-15
|
CTRP2
|
2.2869
|
4.27
|
2.2056
|
18.6479
|
|
A-253
|
GDSC1; GDSC2; CTRP2
|
2.3869
|
4.786
|
2.2104
|
18.316
|
| ----------- |
|
|
|
|
|
Cancer Drug Sensitivity Data Curated from 1 Cell Line(s) in Small Intestine Tissue
| Cell Line |
Original Dataset |
IC50 |
IC90 |
EC50 |
AUC |
|
HuTu 80
|
GDSC1; GDSC2
|
-0.871
|
2.2792
|
-1.0172
|
42.5743
|
| ----------- |
|
|
|
|
|
Cancer Drug Sensitivity Data Curated from 44 Cell Line(s) in Stomach Tissue
| Cell Line |
Original Dataset |
IC50 |
IC90 |
EC50 |
AUC |
|
ECC10
|
GDSC1; GDSC2; CTRP2
|
-0.3814
|
1.3184
|
-0.3828
|
36.1521
|
|
AGS
|
GDSC1; GDSC2; CTRP2; CTRP1
|
-0.3057
|
1.3251
|
-0.3065
|
36.3457
|
|
NUGC-4
|
GDSC1; GDSC2; CTRP2; CTRP1
|
-0.2711
|
1.7781
|
-0.2763
|
36.1319
|
|
TMK-1
|
GDSC1; GDSC2
|
-0.0484
|
2.3262
|
-0.14
|
34.2668
|
|
GSS
|
CTRP2
|
0.0591
|
1.641
|
0.0577
|
33.2173
|
|
HSC-39
|
GDSC1; GDSC2
|
0.324
|
2.1728
|
0.2776
|
30.3185
|
|
Fu97
|
GDSC1; GDSC2; CTRP1
|
0.3314
|
3.1784
|
0.2727
|
41.3565
|
|
NCC-StC-K140
|
CTRP2
|
0.4657
|
13.4576
|
-8.5058
|
24.2266
|
|
SK-GT-2
|
GDSC1; GDSC2
|
0.542
|
2.9938
|
0.371
|
28.7703
|
|
OCUM-1
|
GDSC1; GDSC2
|
0.5999
|
2.3132
|
0.5538
|
27.5498
|
|
SNU-16
|
GDSC1; GDSC2; CTRP2
|
0.8166
|
2.504
|
0.8104
|
28.187
|
|
HGC-27
|
GDSC1; GDSC2; CTRP2
|
0.8216
|
5.9607
|
0.1567
|
30.3506
|
|
SNU-620
|
CTRP2
|
0.878
|
2.053
|
0.8776
|
27.7586
|
|
SNU-216
|
CTRP2
|
1.0658
|
3.7347
|
0.9773
|
26.8051
|
|
RF-48
|
GDSC1; GDSC2
|
1.1068
|
3.1688
|
0.9446
|
23.0758
|
|
RERF-GC-1B
|
GDSC1; GDSC2
|
1.2255
|
4.3425
|
0.6923
|
23.9077
|
|
23132/87
|
GDSC1; GDSC2; CTRP2
|
1.2648
|
2.9492
|
1.2538
|
25.2167
|
|
SCH
|
GDSC1; GDSC2
|
1.3143
|
2.7484
|
1.2567
|
20.4419
|
|
TGBC11TKB
|
GDSC1; GDSC2
|
1.3948
|
2.8455
|
1.3271
|
19.6874
|
|
MKN28
|
GDSC1; GDSC2
|
1.6134
|
4.5217
|
1.0332
|
20.347
|
|
KE-39
|
CTRP2
|
1.6412
|
4.5312
|
1.4569
|
23.3065
|
|
NCI-N87
|
GDSC1; GDSC2; CTRP2
|
1.6557
|
3.0746
|
1.649
|
22.5984
|
|
SNU-719
|
CTRP2
|
1.7494
|
7.2749
|
0.6644
|
25.795
|
|
Hs 746.T
|
GDSC1; GDSC2; CTRP2
|
1.7563
|
3.6711
|
1.7172
|
22.0374
|
|
SNU-1
|
GDSC1; GDSC2; CTRP2
|
1.7991
|
4.3015
|
1.6743
|
22.0474
|
|
GSU
|
CTRP2
|
1.8551
|
2.9283
|
1.8536
|
21.2525
|
|
GCIY
|
GDSC1; GDSC2
|
1.8778
|
3.5336
|
1.6834
|
15.5299
|
|
SNU-601
|
CTRP2
|
1.9985
|
4.0771
|
1.9257
|
20.5392
|
|
NUGC-3
|
GDSC1; GDSC2; CTRP2
|
2.0218
|
4.5947
|
1.8568
|
20.7058
|
|
SNU-668
|
CTRP2
|
2.0892
|
4.563
|
1.9368
|
20.2128
|
|
IM95
|
GDSC1; GDSC2; CTRP2
|
2.0932
|
4.4206
|
1.97
|
20.0836
|
|
KATO III
|
GDSC1; GDSC2; CTRP2
|
2.1283
|
4.024
|
2.0715
|
19.6199
|
|
SNU-5
|
GDSC1; GDSC2; CTRP2
|
2.1425
|
3.961
|
2.0943
|
19.4958
|
|
ECC12
|
GDSC2; CTRP2
|
2.4172
|
4.0896
|
2.3704
|
17.6609
|
|
MKN74
|
CTRP2; CTRP1
|
2.4355
|
4.56
|
2.3316
|
18.6311
|
|
SH-10-TC
|
CTRP2; CTRP1
|
2.9136
|
4.3516
|
2.8755
|
15.2561
|
|
MKN7
|
GDSC1; GDSC2; CTRP2; CTRP1
|
2.9469
|
5.3305
|
2.7062
|
15.7463
|
|
MKN1
|
GDSC1; GDSC2
|
3.0635
|
5.7371
|
1.8928
|
9.8249
|
|
HuG1-N
|
CTRP2
|
3.5391
|
6.4184
|
2.8956
|
12.4322
|
|
LMSU
|
CTRP2
|
5.2595
|
7.9185
|
3.7824
|
4.9037
|
|
MKN45
|
GDSC1; GDSC2; CTRP2
|
6.0638
|
9.8388
|
3.4345
|
5.0335
|
|
SNU-1077
|
CTRP2
|
1.8254
|
3.9034
|
1.7645
|
21.6516
|
|
SNU-685
|
CTRP2
|
2.3587
|
5.2785
|
2.0463
|
18.9932
|
|
SNG-M
|
GDSC1; GDSC2; CTRP2; CTRP1
|
2.4074
|
4.3718
|
2.3327
|
18.7158
|
| ----------- |
|
|
|
|
|
| ⏷ Show the Full List of 44 Cancer Drug Sensitivity Data of This Drug |
|
Cancer Drug Sensitivity Data Curated from 18 Cell Line(s) in Thyroid Tissue
| Cell Line |
Original Dataset |
IC50 |
IC90 |
EC50 |
AUC |
|
IHH-4
|
GDSC1; GDSC2
|
0.7433
|
1.8696
|
0.7367
|
25.9102
|
|
ML-1 [Human leukemia]
|
GDSC1; GDSC2
|
1.177
|
5.081
|
0.344
|
25.9722
|
|
8505C
|
GDSC1; GDSC2; CTRP2
|
1.3195
|
3.3588
|
1.2865
|
24.9265
|
|
BHT-101
|
GDSC1; GDSC2; CTRP2
|
1.3203
|
3.7904
|
1.2413
|
25.0778
|
|
B-CPAP
|
GDSC1; GDSC2; CTRP2
|
1.3447
|
3.0823
|
1.3302
|
24.6964
|
|
K5
|
GDSC1; GDSC2
|
1.3696
|
3.5819
|
1.115
|
20.946
|
|
TT
|
GDSC1; GDSC2; CTRP2
|
1.5654
|
5.2115
|
1.2007
|
24.4483
|
|
CAL-62
|
GDSC1; GDSC2; CTRP2
|
1.6888
|
3.6095
|
1.6519
|
22.4798
|
|
CGTH-W-1
|
GDSC1; GDSC2; CTRP2
|
1.7355
|
3.6001
|
1.7018
|
22.1575
|
|
KMH-2
|
GDSC1; GDSC2
|
1.7587
|
2.7925
|
1.7259
|
15.8546
|
|
8305C
|
GDSC1; GDSC2; CTRP2
|
1.801
|
3.4875
|
1.7789
|
21.6822
|
|
HTC-C3
|
GDSC1; GDSC2
|
2.0107
|
4.5124
|
1.4528
|
16.2915
|
|
SW579
|
CTRP2
|
2.0306
|
4.4041
|
1.9055
|
20.5069
|
|
ASH-3
|
GDSC1; GDSC2
|
2.2275
|
3.8474
|
1.9519
|
12.4944
|
|
TT2609-C02
|
GDSC1; GDSC2; CTRP2
|
2.4898
|
5.338
|
2.1711
|
18.145
|
|
FTC-238
|
CTRP2
|
2.5175
|
4.3466
|
2.4411
|
17.095
|
|
FTC-133
|
GDSC1; GDSC2; CTRP2
|
2.5752
|
4.6842
|
2.4388
|
16.9205
|
|
WRO
|
GDSC1; GDSC2
|
3.3528
|
6.6491
|
1.684
|
10.2617
|
| ----------- |
|
|
|
|
|
| ⏷ Show the Full List of 18 Cancer Drug Sensitivity Data of This Drug |
|
Cancer Drug Sensitivity Data Curated from 55 Cell Line(s) in Upper Aerodigestive Tract Tissue
| Cell Line |
Original Dataset |
IC50 |
IC90 |
EC50 |
AUC |
|
PCI-06A
|
GDSC1; GDSC2
|
-0.7176
|
1.2392
|
-0.7365
|
40.621
|
|
BICR 78
|
GDSC1; GDSC2
|
-0.6066
|
1.514
|
-0.639
|
39.5686
|
|
NCI-H3118
|
GDSC1; GDSC2
|
-0.549
|
2.0538
|
-0.636
|
39.2206
|
|
SKN-3
|
GDSC1; GDSC2
|
-0.3047
|
2.3706
|
-0.4227
|
36.933
|
|
PCI-04B
|
GDSC1; GDSC2
|
-0.2905
|
1.707
|
-0.3241
|
36.413
|
|
JHU-011
|
GDSC1; GDSC2
|
-0.1883
|
3.3579
|
-0.5188
|
36.6743
|
|
Ca9-22
|
GDSC1; GDSC2
|
0.3314
|
1.853
|
0.3132
|
30.1021
|
|
SAS
|
GDSC1; GDSC2
|
0.3358
|
2.9385
|
0.1592
|
30.852
|
|
SAT [Human HNSCC]
|
GDSC1; GDSC2
|
0.6641
|
4.4091
|
0.0657
|
29.7027
|
|
HO-1-N-1
|
GDSC1; GDSC2
|
0.7361
|
3.2961
|
0.507
|
27.1341
|
|
JHU-022
|
GDSC1; GDSC2
|
0.7425
|
4.9695
|
-0.0672
|
29.9609
|
|
JHU-029
|
GDSC1; GDSC2
|
0.7693
|
3.084
|
0.5973
|
26.5049
|
|
HO-1-u-1
|
GDSC1; GDSC2
|
0.7983
|
2.696
|
0.71
|
25.778
|
|
PE/CA-PJ49
|
CTRP2
|
0.9012
|
2.6333
|
0.893
|
27.6297
|
|
BICR 56
|
CTRP2
|
0.9685
|
3.4852
|
0.905
|
27.368
|
|
SCC-4
|
GDSC1; GDSC2; CTRP2
|
1.0026
|
4.184
|
0.8351
|
27.4961
|
|
PCI-38
|
GDSC1
|
1.14
|
5.4403
|
0.1529
|
32.9926
|
|
KON
|
GDSC1; GDSC2
|
1.4061
|
3.7965
|
1.0847
|
20.95
|
|
BICR 10
|
GDSC1; GDSC2
|
1.45
|
4.2487
|
0.968
|
21.4085
|
|
KOSC-2
|
GDSC1; GDSC2
|
1.5228
|
3.5268
|
1.2989
|
19.2456
|
|
PCI-30
|
GDSC1; GDSC2
|
1.5429
|
7.5023
|
-0.388
|
27.931
|
|
SNU-899
|
CTRP2
|
1.648
|
4.4907
|
1.473
|
23.229
|
|
DOK
|
GDSC1; GDSC2
|
1.6686
|
3.8811
|
1.339
|
18.3769
|
|
Detroit 562
|
GDSC1; GDSC2; CTRP2
|
1.7404
|
4.0727
|
1.6499
|
22.3192
|
|
OSC-19
|
GDSC1; GDSC2
|
1.755
|
5.554
|
0.731
|
21.4778
|
|
BB30-HNC
|
GDSC1; GDSC2
|
1.7951
|
3.677
|
1.5467
|
16.6578
|
|
SNU-1076
|
CTRP2
|
2.0006
|
3.9823
|
1.9409
|
20.4801
|
|
HSC-3
|
GDSC1; GDSC2; CTRP2
|
2.0673
|
4.3547
|
1.9543
|
20.2204
|
|
SNU-1214
|
CTRP2
|
2.0825
|
4.4832
|
1.9461
|
20.2011
|
|
SCC-25
|
GDSC1; GDSC2; CTRP2
|
2.0966
|
4.4923
|
1.9596
|
20.1093
|
|
HSC-2
|
GDSC1; GDSC2; CTRP2; CTRP1
|
2.1264
|
4.8092
|
1.9451
|
20.9353
|
|
SNU-46
|
CTRP2
|
2.221
|
4.8502
|
2.0139
|
19.5294
|
|
YD-8
|
CTRP2
|
2.3582
|
4.1538
|
2.2993
|
18.0952
|
|
SCC-15
|
GDSC1; GDSC2
|
2.448
|
6.3841
|
1.013
|
17.2632
|
|
CAL-27
|
GDSC1; GDSC2; CTRP2
|
2.4667
|
4.1376
|
2.417
|
17.3412
|
|
BICR 6
|
CTRP2
|
2.5323
|
5.175
|
2.2662
|
17.671
|
|
HSC-4
|
GDSC1; GDSC2; CTRP2
|
2.5601
|
5.1853
|
2.2935
|
17.4879
|
|
SNU-1041
|
CTRP2
|
2.5856
|
5.6937
|
2.1594
|
17.9057
|
|
BICR 18
|
CTRP2
|
2.5889
|
5.0367
|
2.3667
|
17.1356
|
|
CAL-33
|
GDSC1; GDSC2; CTRP2
|
2.6226
|
4.5553
|
2.5171
|
16.4961
|
|
BHY
|
GDSC1; GDSC2; CTRP2
|
2.6235
|
5.8155
|
2.1579
|
17.8014
|
|
SCC-9
|
GDSC1; GDSC2; CTRP2; CTRP1
|
2.629
|
4.6245
|
2.5287
|
17.344
|
|
BICR 22
|
GDSC1; GDSC2; CTRP2
|
2.6346
|
5.1164
|
2.3945
|
16.8967
|
|
FaDu
|
GDSC1; GDSC2; CTRP2
|
2.7315
|
5.0787
|
2.5109
|
16.1816
|
|
BB49-HNC
|
GDSC1; GDSC2
|
2.8127
|
5.2901
|
1.8868
|
10.6816
|
|
OSC-20
|
GDSC1; GDSC2
|
2.9643
|
8.9542
|
0.2024
|
20.0212
|
|
UPCI-SCC-090
|
GDSC1; GDSC2
|
3.0916
|
6.2369
|
1.6683
|
11.1436
|
|
BICR 31
|
GDSC2; CTRP2
|
3.1552
|
5.7174
|
2.7663
|
13.9889
|
|
PE/CA-PJ34 (clone C12)
|
CTRP2
|
3.3
|
5.1508
|
3.1106
|
12.288
|
|
LB771-HNC
|
GDSC1; GDSC2
|
3.3165
|
4.9096
|
2.5235
|
5.0709
|
|
PE/CA-PJ41 (clone D2)
|
CTRP2
|
3.4801
|
4.9996
|
3.3492
|
10.8781
|
|
PCI-15A
|
GDSC1; GDSC2
|
3.6683
|
5.1489
|
2.6786
|
3.17
|
|
BICR 16
|
CTRP2
|
3.8619
|
7.71
|
2.6321
|
12.675
|
|
PE/CA-PJ15
|
GDSC1; GDSC2; CTRP2
|
4.0704
|
8.0732
|
2.6576
|
12.0802
|
|
SNU-1066
|
CTRP2
|
4.3005
|
6.7666
|
3.5054
|
8.064
|
| ----------- |
|
|
|
|
|
| ⏷ Show the Full List of 55 Cancer Drug Sensitivity Data of This Drug |
|
Cancer Drug Sensitivity Data Curated from 25 Cell Line(s) in Urinary Tract Tissue
| Cell Line |
Original Dataset |
IC50 |
IC90 |
EC50 |
AUC |
|
253J
|
CTRP2
|
0.9409
|
3.7999
|
0.8343
|
27.6986
|
|
5637
|
GDSC1; GDSC2; CTRP2; CTRP1
|
1.0916
|
2.9108
|
1.0802
|
27.1729
|
|
U-BLC1
|
CTRP2
|
1.1666
|
2.8998
|
1.1551
|
25.8731
|
|
KU-19-19
|
GDSC1; GDSC2; CTRP2
|
1.3335
|
2.4526
|
1.3328
|
24.7246
|
|
TCCSUP
|
GDSC1; GDSC2; CTRP2
|
1.4104
|
4.2566
|
1.2625
|
24.7157
|
|
JMSU-1
|
CTRP2
|
1.4559
|
3.6091
|
1.4068
|
24.0729
|
|
RT-112
|
GDSC1; GDSC2; CTRP2
|
1.4992
|
4.5978
|
1.2875
|
24.3474
|
|
CAL-29
|
GDSC1; GDSC2; CTRP2
|
1.5183
|
3.2846
|
1.4986
|
23.5574
|
|
BFTC-905
|
GDSC1; GDSC2
|
1.6868
|
3.648
|
1.4388
|
17.7382
|
|
UM-UC-1
|
CTRP2
|
1.8632
|
6.5401
|
1.0742
|
24.0097
|
|
SCaBER
|
CTRP2
|
2.0304
|
4.1188
|
1.9538
|
20.3401
|
|
KMBC-2
|
CTRP2; CTRP1
|
2.0526
|
4.9555
|
1.8302
|
21.5651
|
|
647V
|
GDSC1; GDSC2; CTRP2
|
2.2312
|
4.6857
|
2.0642
|
19.3189
|
|
RT-4
|
GDSC1; GDSC2; CTRP2; CTRP1
|
2.3661
|
4.4925
|
2.2686
|
19.0662
|
|
VM-CUB-1
|
CTRP2
|
2.3729
|
4.1481
|
2.3158
|
17.9913
|
|
253J-BV
|
CTRP2
|
2.4281
|
4.4416
|
2.3282
|
17.7719
|
|
HT-1376
|
GDSC1; GDSC2; CTRP2; CTRP1
|
2.4444
|
4.1553
|
2.401
|
18.3659
|
|
SW780
|
GDSC1; GDSC2
|
2.6249
|
4.5402
|
2.0658
|
10.242
|
|
SW1710
|
GDSC1; GDSC2; CTRP2
|
2.7989
|
4.9439
|
2.6206
|
15.5808
|
|
HT-1197
|
GDSC1; GDSC2; CTRP2
|
3.0744
|
4.0627
|
3.0632
|
13.1619
|
|
T24
|
GDSC1; GDSC2; CTRP2
|
3.1411
|
6.5207
|
2.4373
|
15.2883
|
|
BC-3C
|
CTRP2
|
3.387
|
5.29
|
3.163
|
11.8359
|
|
UM-UC-3
|
GDSC1; GDSC2; CTRP2; CTRP1
|
3.5916
|
6.9957
|
2.766
|
13.7381
|
|
639V
|
GDSC1; GDSC2; CTRP2
|
4.0794
|
6.2394
|
3.5296
|
8.5132
|
|
J82
|
GDSC1; GDSC2; CTRP2
|
4.3817
|
9.1725
|
2.405
|
12.4233
|
| ----------- |
|
|
|
|
|
| ⏷ Show the Full List of 25 Cancer Drug Sensitivity Data of This Drug |
|
Cancer Drug Sensitivity Data Curated from 39 Cell Line(s) in Urogenital System Tissue
| Cell Line |
Original Dataset |
IC50 |
IC90 |
EC50 |
AUC |
|
NEC8
|
GDSC1; GDSC2
|
-1.3399
|
-0.0098
|
-1.3404
|
46.7835
|
|
PA-1
|
GDSC1; GDSC2
|
-0.8528
|
1.2746
|
-0.8784
|
41.9987
|
|
SW954
|
GDSC1; GDSC2
|
0.1163
|
2.2827
|
0.0427
|
32.5348
|
|
TC-YIK
|
GDSC1; GDSC2
|
0.131
|
2.2406
|
0.0642
|
32.3544
|
|
NT2-D1
|
GDSC1; GDSC2
|
0.4629
|
2.7355
|
0.3401
|
29.3147
|
|
OV17R
|
GDSC1; GDSC2
|
0.4682
|
4.1612
|
-0.0562
|
31.2298
|
|
PWR-1E
|
GDSC1; GDSC2
|
0.5119
|
1.8768
|
0.4986
|
28.2659
|
|
HEY
|
GDSC1; GDSC2
|
0.5817
|
2.3402
|
0.5312
|
27.7548
|
|
C-33 A
|
GDSC1; GDSC2
|
0.902
|
2.6421
|
0.8307
|
24.6495
|
|
SiSo
|
GDSC1; GDSC2
|
1.008
|
3.5966
|
0.7167
|
24.7544
|
|
MS751
|
GDSC1; GDSC2
|
1.0319
|
3.7583
|
0.6914
|
24.7804
|
|
C-4-I
|
GDSC1; GDSC2
|
1.1149
|
2.997
|
0.9936
|
22.7769
|
|
CAL-39
|
GDSC1; GDSC2
|
1.133
|
3.2624
|
0.9491
|
22.9289
|
|
SKN
|
GDSC1; GDSC2
|
1.2272
|
3.2925
|
1.0438
|
21.9836
|
|
NCC-IT
|
GDSC2
|
1.2871
|
4.1015
|
0.8484
|
22.782
|
|
DSH1
|
GDSC1; GDSC2
|
1.3144
|
3.2442
|
1.1518
|
20.9982
|
|
Ca Ski
|
GDSC1; GDSC2
|
1.3371
|
3.7779
|
1.0162
|
21.6355
|
|
SKG-IIIa
|
GDSC1; GDSC2
|
1.3498
|
3.1047
|
1.2249
|
20.441
|
|
JAR
|
GDSC1; GDSC2
|
1.4377
|
2.6952
|
1.3967
|
19.118
|
|
DoTc2 4510
|
GDSC1; GDSC2
|
1.4986
|
5.6089
|
0.4468
|
24.0664
|
|
HEC-1
|
GDSC1; GDSC2
|
1.508
|
2.9513
|
1.4298
|
18.6073
|
|
JEG-3
|
GDSC1; GDSC2
|
1.62
|
2.5277
|
1.6068
|
17.1459
|
|
BPH-1
|
GDSC1; GDSC2
|
1.6624
|
4.0697
|
1.2631
|
18.8327
|
|
ACC-OV7
|
GDSC1; GDSC2
|
1.728
|
4.385
|
1.2096
|
18.8608
|
|
OVMIU
|
GDSC1; GDSC2
|
1.7479
|
4.6065
|
1.1393
|
19.1827
|
|
OVCA433
|
GDSC1; GDSC2
|
1.8489
|
3.2773
|
1.7264
|
15.4255
|
|
OVCA420
|
GDSC1; GDSC2
|
1.9826
|
3.629
|
1.7662
|
14.6045
|
|
ME-180
|
GDSC1; GDSC2
|
1.9845
|
3.575
|
1.7862
|
14.4842
|
|
HeLa
|
GDSC1; GDSC2; CTRP2
|
2.1767
|
4.3748
|
2.0689
|
19.4738
|
|
HT-3
|
GDSC1; GDSC2
|
2.1781
|
4.1739
|
1.7697
|
13.7555
|
|
UWB1.289
|
GDSC1; GDSC2
|
2.2565
|
4.9284
|
1.5198
|
14.9316
|
|
SW756
|
GDSC1; GDSC2
|
2.4734
|
5.8964
|
1.2737
|
15.6213
|
|
KGN
|
GDSC1; GDSC2
|
2.5367
|
5.5862
|
1.4834
|
14.1256
|
|
OMC-1 [Human cervical carcinoma]
|
GDSC1; GDSC2
|
2.6159
|
4.374
|
2.1334
|
9.8656
|
|
PEO1
|
GDSC1; GDSC2
|
2.6911
|
4.2372
|
2.2713
|
8.7462
|
|
SW962
|
GDSC1; GDSC2
|
2.7708
|
4.7847
|
2.0924
|
9.5499
|
|
LB831-BLC
|
GDSC1; GDSC2
|
2.9061
|
5.8722
|
1.6856
|
11.6252
|
|
SiHa
|
GDSC1; GDSC2
|
3.3446
|
4.7184
|
2.6425
|
4.2361
|
|
JHOS-3
|
GDSC1; GDSC2
|
3.7544
|
5.5636
|
2.5379
|
3.8393
|
| ----------- |
|
|
|
|
|
| ⏷ Show the Full List of 39 Cancer Drug Sensitivity Data of This Drug |
|
|