Details of the Drug
General Information of Drug (ID: DMMZXIW)
| Drug Name |
Rabeprazole
|
||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Synonyms |
Dexrabeprazole; Eraloc; Rebeprazole sodium; CL23619; LY307640; Aciphex (TN); Eraloc (TN); Pariet (TN); Rabeprazole (INN); Rabeprazole [BAN:INN]; Rabeprazole [INN:BAN]; Rablet (TN); 2-(((4-(3-Methoxypropoxy)-3-methyl-2-pyridinyl)methyl)sulfinyl)-1H-benzimidazole; 2-((4-(3-methoxypropoxy)-3-methylpyridin-2-yl)methylsulfinyl)-1H-benzimidazole; 2-({[4-(3-methoxypropoxy)-3-methylpyridin-2-yl]methyl}sulfinyl)-1H-benzimidazole; 2-[[4-(3-methoxypropoxy)-3-methylpyridin-2-yl]methylsulfinyl]-1H-benzimidazole
|
||||||||||||||||||||||||||||||
| Indication |
|
||||||||||||||||||||||||||||||
| Therapeutic Class |
Antiulcer Agents
|
||||||||||||||||||||||||||||||
| ATC Code |
|
||||||||||||||||||||||||||||||
| Drug Type |
Small molecular drug
|
||||||||||||||||||||||||||||||
| Structure |
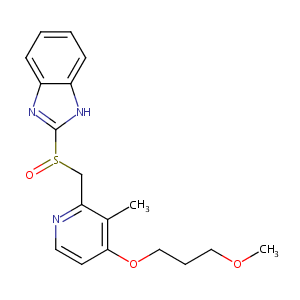 |
||||||||||||||||||||||||||||||
| 3D MOL | 2D MOL | ||||||||||||||||||||||||||||||
| #Ro5 Violations (Lipinski): 0 | Molecular Weight (mw) | 359.4 | |||||||||||||||||||||||||||||
| Logarithm of the Partition Coefficient (xlogp) | 1.9 | ||||||||||||||||||||||||||||||
| Rotatable Bond Count (rotbonds) | 8 | ||||||||||||||||||||||||||||||
| Hydrogen Bond Donor Count (hbonddonor) | 1 | ||||||||||||||||||||||||||||||
| Hydrogen Bond Acceptor Count (hbondacc) | 6 | ||||||||||||||||||||||||||||||
| ADMET Property |
|
||||||||||||||||||||||||||||||
| Chemical Identifiers |
|
||||||||||||||||||||||||||||||
| Cross-matching ID | |||||||||||||||||||||||||||||||
| Combinatorial Drugs (CBD) | Click to Jump to the Detailed CBD Information of This Drug | ||||||||||||||||||||||||||||||
| Repurposed Drugs (RPD) | Click to Jump to the Detailed RPD Information of This Drug | ||||||||||||||||||||||||||||||
Molecular Interaction Atlas of This Drug
 Drug Therapeutic Target (DTT) |
|
|||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
 Drug Transporter (DTP) |
|
|||||||||||||||||||||||||||||||
 Drug-Metabolizing Enzyme (DME) |
|
|||||||||||||||||||||||||||||||
| Molecular Interaction Atlas (MIA) | ||||||||||||||||||||||||||||||||
Molecular Expression Atlas of This Drug
| The Studied Disease | Bacterial infection | |||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ICD Disease Classification | 1A00-1C4Z | |||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||
| Molecular Expression Atlas (MEA) | ||||||||||||||||||||||||||||||||||||||||||
Drug-Drug Interaction (DDI) Information of This Drug
|
Coadministration of a Drug Treating the Same Disease as Rabeprazole
Coadministration of a Drug Treating the Disease Different from Rabeprazole (Comorbidity)
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Drug Inactive Ingredient(s) (DIG) and Formulation(s) of This Drug
References
| 1 | URL: http://www.guidetopharmacology.org Nucleic Acids Res. 2015 Oct 12. pii: gkv1037. The IUPHAR/BPS Guide to PHARMACOLOGY in 2016: towards curated quantitative interactions between 1300 protein targets and 6000 ligands. (Ligand id: 7290). | ||||
|---|---|---|---|---|---|
| 2 | Rabeprazole FDA Label | ||||
| 3 | BDDCS applied to over 900 drugs | ||||
| 4 | Trend Analysis of a Database of Intravenous Pharmacokinetic Parameters in Humans for 1352 Drug Compounds | ||||
| 5 | Estimating the safe starting dose in phase I clinical trials and no observed effect level based on QSAR modeling of the human maximum recommended daily dose | ||||
| 6 | Review article: rabeprazole's tolerability profile in clinical trials. Aliment Pharmacol Ther. 1999 Oct;13 Suppl 5:17-23. | ||||
| 7 | Co-administration of proton pump inhibitors delays elimination of plasma methotrexate in high-dose methotrexate therapy. Br J Clin Pharmacol. 2009 Jan;67(1):44-9. | ||||
| 8 | Effects of clarithromycin and verapamil on rabeprazole pharmacokinetics between CYP2C19 genotypes. Eur J Clin Pharmacol. 2006 Aug;62(8):597-603. | ||||
| 9 | FDA. U.S. Food and Drug Administration "FDA Drug Safety Communication: Low magnesium levels can be associated with long-term use of proton pump inhibitor drugs (PPIs).". | ||||
| 10 | Honig PK, Gillespie BK "Clinical significance of pharmacokinetic drug interactions with over-the-counter (OTC) drugs." Clin Pharmacokinet 35 (1998): 167-71. [PMID: 9784931] | ||||
| 11 | Cerner Multum, Inc. "UK Summary of Product Characteristics.". | ||||
| 12 | Alffenaar JW, van Assen S, van der Werf TS, Kosterink JG, Uges DR "Omeprazole significantly reduces posaconazole serum trough level." Clin Infect Dis 48 (2009): 839. [PMID: 19220151] | ||||
| 13 | Cerner Multum, Inc. "Australian Product Information.". | ||||
| 14 | Product Information. Turalio (pexidartinib). Daiichi Sankyo, Inc., Parsippany, NJ. | ||||
| 15 | Product Information. Bosulif (bosutinib). Pfizer U.S. Pharmaceuticals Group, New York, NY. | ||||
| 16 | Cerner Multum, Inc. "Canadian Product Information.". | ||||
| 17 | EMEA. European Medicines Agency "EPARs. European Union Public Assessment Reports.". | ||||
| 18 | Bottiger Y, Tybring G, Gotharson E, Bertilsson L "Inhibition of the sulfoxidation of omeprazole by ketoconazole in poor and extensive metabolizers of S-mephenytoin." Clin Pharmacol Ther 62 (1997): 384-91. [PMID: 9357389] | ||||
| 19 | Product Information. Priftin (rifapentine). Hoechst Marion-Roussel Inc, Kansas City, MO. | ||||
| 20 | Product Information. Lexiva (fosamprenavir). GlaxoSmithKline, Research Triangle Park, NC. | ||||
| 21 | Product Information. Intelence (etravirine). Ortho Biotech Inc, Bridgewater, NJ. | ||||
| 22 | Product Information. Edurant (rilpivirine). Tibotec Pharmaceuticals, Titusville, NJ. | ||||
| 23 | Product Information. Isentress (raltegravir). Merck & Company Inc, West Point, PA. | ||||
| 24 | Hutchison C, Geissler CA, Powell JJ, Bomford A "Proton pump inhibitors suppress absorption of dietary non-haem iron in hereditary haemochromatosis." Gut 56 (2007): 1291-5. [PMID: 17344278] | ||||
| 25 | Product Information. Alunbrig (brigatinib). Ariad Pharmaceuticals Inc, Cambridge, MA. | ||||
| 26 | Product Information. Zykadia (ceritinib). Novartis Pharmaceuticals, East Hanover, NJ. | ||||
| 27 | Product Information. Calquence (acalabrutinib). Astra-Zeneca Pharmaceuticals, Wilmington, DE. | ||||
| 28 | Product Information. Exjade (deferasirox). Novartis Pharmaceuticals, East Hanover, NJ. | ||||
| 29 | Product Information. Addyi (flibanserin). Sprout Pharmaceuticals, Raleigh, NC. | ||||
| 30 | Product Information. Tasigna (nilotinib). Novartis Pharmaceuticals, East Hanover, NJ. | ||||
| 31 | Product Information. Sprycel (dasatinib). Bristol-Myers Squibb, Princeton, NJ. | ||||
| 32 | Product Information. Effient (prasugrel). Lilly, Eli and Company, Indianapolis, IN. | ||||
| 33 | Product Information. Xeglyze (abametapir topical). Dr. Reddy's Laboratories Inc, Upper Saddle River, NJ. | ||||
| 34 | Product Information. Fycompa (perampanel). Eisai Inc, Teaneck, NJ. | ||||
| 35 | Miner PB Jr, Fort JG, Zhang Y. Intragastric acidity and omeprazole exposure during dosing with either PA32540 (enteric-coated aspirin 325?mg + immediate-release omeprazole 40?mg) or enteric-coated aspirin 325?mg + enteric-coated omeprazole 40?mg - a randomised, phase 1, crossover study.?Aliment Pharmacol Ther. 2013;38(1):62-71. [PMID: 23692061] | ||||
| 36 | Benoist G, van Oort I, et al "Drug-drug interaction potential in men treated with enzalutamide: Mind the gap." Br J Clin Pharmacol 0 (2017): epub. [PMID: 28881501] | ||||
| 37 | Product Information. Adempas (riociguat). Bayer Pharmaceutical Inc, West Haven, CT. | ||||
| 38 | Product Information. Inlyta (axitinib). Pfizer U.S. Pharmaceuticals Group, New York, NY. | ||||
| 39 | Product Information. Tavalisse (fostamatinib). Rigel Pharmaceuticals, South San Francisco, CA. | ||||
