Details of the Drug
General Information of Drug (ID: DMQVRZH)
| Drug Name |
Vitamin B3
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Synonyms | Niacin (TN); Niacinamide (TN); Nicotinamide (TN) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Indication |
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Affected Organisms |
Humans and other mammals
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Drug Type |
Small molecular drug
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Structure |
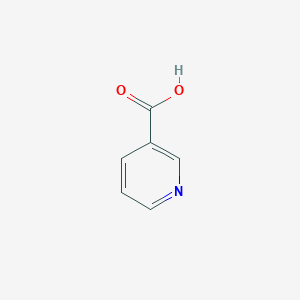 |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 3D MOL | 2D MOL | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| #Ro5 Violations (Lipinski): 0 | Molecular Weight (mw) | 123.11 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Logarithm of the Partition Coefficient (xlogp) | 0.4 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Rotatable Bond Count (rotbonds) | 1 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hydrogen Bond Donor Count (hbonddonor) | 1 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hydrogen Bond Acceptor Count (hbondacc) | 3 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ADMET Property |
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical Identifiers |
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Cross-matching ID | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Combinatorial Drugs (CBD) | Click to Jump to the Detailed CBD Information of This Drug | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Repurposed Drugs (RPD) | Click to Jump to the Detailed RPD Information of This Drug | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Molecular Interaction Atlas of This Drug
 Drug Therapeutic Target (DTT) |
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
 Drug-Metabolizing Enzyme (DME) |
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
 Drug Off-Target (DOT) |
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Molecular Interaction Atlas (MIA) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Drug-Drug Interaction (DDI) Information of This Drug
|
Coadministration of a Drug Treating the Disease Different from Vitamin B3 (Comorbidity)
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Drug Inactive Ingredient(s) (DIG) and Formulation(s) of This Drug
References
| 1 | Nicotinamide for photoprotection and skin cancer chemoprevention: A review of efficacy and safety. Exp Dermatol. 2019 Feb;28 Suppl 1:15-22. | ||||
|---|---|---|---|---|---|
| 2 | Vitamin B3 impairs reverse cholesterol transport in Apolipoprotein E-deficient mice. Clin Investig Arterioscler. 2019 Nov-Dec;31(6):251-260. | ||||
| 3 | Vitamin B3. 2024 Feb 29. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2024 JanC. | ||||
| 4 | A standard database for drug repositioning. Sci Data. 2017 Mar 14;4:170029. | ||||
| 5 | Niacin: chemical forms, bioavailability, and health effects. Nutr Rev. 2012 Jun;70(6):357-66. | ||||
| 6 | Hakozaki T, Minwalla L, Zhuang J, Chhoa M, Matsubara A, Miyamoto K, Greatens A, Hillebrand GG, Bissett DL, Boissy RE: The effect of niacinamide on reducing cutaneous pigmentation and suppression of melanosome transfer. Br J Dermatol. 2002 Jul;147(1):20-31. | ||||
| 7 | Extended-release niacin (nicotinic acid)/laropiprant. Drugs. 2009 Aug 20;69(12):1665-79. | ||||
| 8 | Cosmeceutical Aptitudes of Niacinamide: A Review. Recent Adv Antiinfect Drug Discov. 2021;16(3):196-208. | ||||
| 9 | Hydrosoluble vitamins. Handb Clin Neurol. 2014;120:891-914. | ||||
| 10 | Reiche I, Westphal S, Martens-Lobenhoffer J, Troger U, Luley C, Bode-Boger SM: Pharmacokinetics and dose recommendations of Niaspan(R) in chronic kidney disease and dialysis patients. Nephrol Dial Transplant. 2011 Jan;26(1):276-82. doi: 10.1093/ndt/gfq344. Epub 2010 Jun 17. | ||||
| 11 | BDDCS applied to over 900 drugs | ||||
| 12 | Plasma and urine pharmacokinetics of niacin and its metabolites from an extended-release niacin formulation. Int J Clin Pharmacol Ther. 2007 Aug;45(8):448-54. doi: 10.5414/cpp45448. | ||||
| 13 | Estimating the safe starting dose in phase I clinical trials and no observed effect level based on QSAR modeling of the human maximum recommended daily dose | ||||
| 14 | ADP-ribosyl cyclase and CD38 catalyze the synthesis of a calcium-mobilizing metabolite from NADP. J Biol Chem. 1995 Dec 22;270(51):30327-33. | ||||
| 15 | Reduced plasma FFA availability increases net triacylglycerol degradation, but not GPAT or HSL activity, in human skeletal muscle. Am J Physiol Endocrinol Metab. 2004 Jul;287(1):E120-7. doi: 10.1152/ajpendo.00542.2003. Epub 2004 Jan 28. | ||||
| 16 | Effects of extended-release niacin on lipid profile and adipocyte biology in patients with impaired glucose tolerance. Atherosclerosis. 2009 Jul;205(1):207-13. doi: 10.1016/j.atherosclerosis.2008.11.026. Epub 2008 Dec 3. | ||||
| 17 | Niacin, but not gemfibrozil, selectively increases LP-AI, a cardioprotective subfraction of HDL, in patients with low HDL cholesterol. Arterioscler Thromb Vasc Biol. 2001 Nov;21(11):1783-9. doi: 10.1161/hq1001.096624. | ||||
| 18 | Niacin treatment of the atherogenic lipid profile and Lp(a) in diabetes. Diabetes Obes Metab. 2002 Jul;4(4):255-61. doi: 10.1046/j.1463-1326.2002.00205.x. | ||||
| 19 | Niacin inhibits surface expression of ATP synthase beta chain in HepG2 cells: implications for raising HDL. J Lipid Res. 2008 Jun;49(6):1195-201. doi: 10.1194/jlr.M700426-JLR200. Epub 2008 Mar 3. | ||||
| 20 | Niacin inhibits vascular oxidative stress, redox-sensitive genes, and monocyte adhesion to human aortic endothelial cells. Atherosclerosis. 2009 Jan;202(1):68-75. doi: 10.1016/j.atherosclerosis.2008.04.044. Epub 2008 May 9. | ||||
| 21 | Effects of extended-release niacin on lipoprotein particle size, distribution, and inflammatory markers in patients with coronary artery disease. Am J Cardiol. 2006 Sep 15;98(6):743-5. doi: 10.1016/j.amjcard.2006.04.011. Epub 2006 Jul 26. | ||||
| 22 | Cerner Multum, Inc. "Australian Product Information.". | ||||
| 23 | Product Information. Sirturo (bedaquiline). Janssen Pharmaceuticals, Titusville, NJ. | ||||
| 24 | Product Information. Turalio (pexidartinib). Daiichi Sankyo, Inc., Parsippany, NJ. | ||||
| 25 | Cerner Multum, Inc. "UK Summary of Product Characteristics.". | ||||
| 26 | Gershon SL, Fox IH "Pharmacologic effects of nicotinic acid on human purine metabolism." J Lab Clin Med 84 (1974): 179-86. [PMID: 4367231] | ||||
| 27 | Product Information. Adcetris (brentuximab vedotin). Seattle Genetics Inc, Bothell, WA. | ||||
| 28 | Product Information. Kynamro (mipomersen). Genzyme Corporation, Cambridge, MA. | ||||
| 29 | Canadian Pharmacists Association. | ||||
| 30 | Product Information. Juxtapid (lomitapide). Aegerion Pharmaceuticals Inc, Cambridge, MA. | ||||
| 31 | Al-Nawakil C, Willems L, Mauprivez C, et.al "Successful treatment of l-asparaginase-induced severe acute hepatotoxicity using mitochondrial cofactors." Leuk Lymphoma 55 (2014): 1670-4. [PMID: 24090500] | ||||
| 32 | Product Information. Zydelig (idelalisib). Gilead Sciences, Foster City, CA. | ||||
| 33 | Product Information. Clolar (clofarabine). sanofi-aventis, Bridgewater, NJ. | ||||
| 34 | Product Information. ReVia (naltrexone). DuPont Pharmaceuticals, Wilmington, DE. | ||||
