Details of the Drug
General Information of Drug (ID: DMSIMD8)
| Drug Name |
Cefuroxime
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Synonyms |
Anaptivan; Biociclin; Biofuroksym; Bioxima; CXM; Cefofix; Cefumax; Cefurex; Cefuril; Cefuroxim; Cefuroximesodium; Cefuroximine; Cefuroximo; Cefuroximum; Cephuroxime; Cetroxil; Colifossim; Curocef; Curoxim; Curoxima; Curoxime; Froxal; Furoxil; Kesint; Ketocef; Lifurox; Medoxim; Sharox; Spectrazolr; Ultroxim; Zinacef;CEFUROXIME AND DEXTROSE IN DUPLEX CONTAINER; CEFUROXIME SODIUM; Cefuroxim AJ; Cefuroxim Fresenius; Cefuroxim Genericsn; Cefuroxim Hexal; Cefuroxim Lilly; Cefuroxim MN; Cefuroxim Norcox; Cefuroxim curasan; Cefuroxima Fabra; Cefuroxima Richet; Cefuroxime for Injection and Dextrose for Injection in Duplex Container; Cefuroxime na; Cefuroxime sodium salt; KEFUROX IN PLASTIC CONTAINER; Sodium cefuroxime; ZINACEF IN PLASTIC CONTAINER; Zinacef Danmark; Ceftin (TN); Cefuroxim Norcox [inj.]; Cefuroxime (TN); Cefuroximo [INN-Spanish]; Cefuroximum [INN-Latin]; Cetroxil [inj.]; Froxal [inj.]; KS-1040; Sharox [inj.]; Zinacef (TN); Zinnat (TN); Zinnat [inj.]; Cefuroxime (USAN/INN); Cefuroxime [USAN:INN:BAN]; Cefuroxime sodium (JP15/USP); Cefuroxime sodium [USAN:BAN:JAN]; Sodium (6R-(6alpha,7beta(Z)))-3-(((aminocarbonyl)oxy)methyl)-7-(2-furyl(methoxyimino)acetamido)-8-oxo-5-thia-1-azabicyclo(4.2.0)oct-2-ene-2-carboxylate; Sodium (6R,7R)-7-(2-(2-furyl)glyoxylamido)-3-(hydroxymethyl)-8-oxo-5-thia-1-azabicyclo(4.2.0)oct-2-ene-2-carboxylate, 7(sup 2)-(Z)-(O-methyloxime), carbamate (ester); (6R,7R)-3-(carbamoyloxymethyl)-7-[[(2Z)-2-(furan-2-yl)-2-methoxyiminoacetyl]amino]-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic acid; (6R,7R)-3-[(carbamoyloxy)methyl]-7-[(2Z)-2-(furan-2-yl)-2-(methoxyimino)acetamido]-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic acid; (6R,7R)-3-[(carbamoyloxy)methyl]-7-{[(2Z)-2-furan-2-yl-2-(methoxyimino)acetyl]amino}-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic acid; (6R,7R)-7-(2-(2-Furyl)glyoxylamido)-3-(hydroxymethyl)-8-oxo-5-thia-1-azabicyclo(4.2.0)oct-2-ene-2-carboxylic acid 7(sup 2)-(Z)-(O-methyloxime) carbamate (ester); 3-[(carbamoyloxy)methyl]-7beta-[(2Z)-2-(furan-2-yl)-2-(methoxyimino)acetamido]-3,4-didehydrocepham-4-carboxylic acid
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Indication |
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Therapeutic Class |
Antibiotics
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Affected Organisms |
Enteric bacteria and other eubacteria
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ATC Code |
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Drug Type |
Small molecular drug
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Structure |
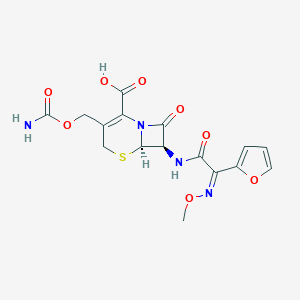 |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 3D MOL | 2D MOL | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| #Ro5 Violations (Lipinski): 0 | Molecular Weight (mw) | 424.4 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Logarithm of the Partition Coefficient (xlogp) | -0.2 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Rotatable Bond Count (rotbonds) | 8 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hydrogen Bond Donor Count (hbonddonor) | 3 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hydrogen Bond Acceptor Count (hbondacc) | 10 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ADMET Property |
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical Identifiers |
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Cross-matching ID | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Combinatorial Drugs (CBD) | Click to Jump to the Detailed CBD Information of This Drug | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Repurposed Drugs (RPD) | Click to Jump to the Detailed RPD Information of This Drug | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Molecular Interaction Atlas of This Drug
 Drug Therapeutic Target (DTT) |
|
|||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
 Drug Off-Target (DOT) |
|
|||||||||||||||||||||||||||||||
| Molecular Interaction Atlas (MIA) | ||||||||||||||||||||||||||||||||
Drug-Drug Interaction (DDI) Information of This Drug
|
Coadministration of a Drug Treating the Disease Different from Cefuroxime (Comorbidity)
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
References
