| 1 |
ClinicalTrials.gov (NCT01052272) Impact of Diabetes on Left Ventricular Remodeling
|
| 2 |
Allopurinol FDA Label
|
| 3 |
URL: http://www.guidetopharmacology.org Nucleic Acids Res. 2015 Oct 12. pii: gkv1037. The IUPHAR/BPS Guide to PHARMACOLOGY in 2016: towards curated quantitative interactions between 1300 protein targets and 6000 ligands. (Ligand id: 6795).
|
| 4 |
Candesartan FDA Label
|
| 5 |
URL: http://www.guidetopharmacology.org Nucleic Acids Res. 2015 Oct 12. pii: gkv1037. The IUPHAR/BPS Guide to PHARMACOLOGY in 2016: towards curated quantitative interactions between 1300 protein targets and 6000 ligands. (Ligand id: 587).
|
| 6 |
Uric acid-lowering treatment with benzbromarone in patients with heart failure: a double-blind placebo-controlled crossover preliminary study. Circ Heart Fail. 2010 Jan;3(1):73-81.
|
| 7 |
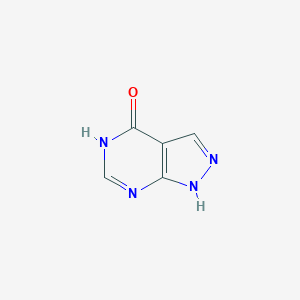
Allopurinol: xanthine oxidase inhibitor. Tex Med. 1966 Jan;62(1):100-1.
|
| 8 |
Isolation, characterization and differential gene expression of multispecific organic anion transporter 2 in mice. Mol Pharmacol. 2002 Jul;62(1):7-14.
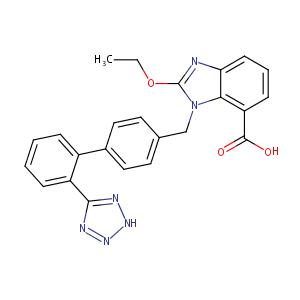
|
| 9 |
Renal transport of organic compounds mediated by mouse organic anion transporter 3 (mOat3): further substrate specificity of mOat3. Drug Metab Dispos. 2004 May;32(5):479-83.
|
| 10 |
Xanthine oxidase inhibition by allopurinol affects the reliability of urinary caffeine metabolic ratios as markers for N-acetyltransferase 2 and CYP1A2 activities. Eur J Clin Pharmacol. 1999 Jan;54(11):869-76.
|
| 11 |
Transport mechanism and substrate specificity of human organic anion transporter 2 (hOat2 [SLC22A7]). J Pharm Pharmacol. 2005 May;57(5):573-8.
|
| 12 |
HLA-B*5801 allele as a genetic marker for severe cutaneous adverse reactions caused by allopurinol. Proc Natl Acad Sci U S A. 2005 Mar 15;102(11):4134-9. doi: 10.1073/pnas.0409500102. Epub 2005 Mar 2.
|
| 13 |
A study of HLA class I and class II 4-digit allele level in Stevens-Johnson syndrome and toxic epidermal necrolysis. Int J Immunogenet. 2011 Aug;38(4):303-9. doi: 10.1111/j.1744-313X.2011.01011.x. Epub 2011 May 4.
|
| 14 |
Positive and negative associations of HLA class I alleles with allopurinol-induced SCARs in Koreans. Pharmacogenet Genomics. 2011 May;21(5):303-7. doi: 10.1097/FPC.0b013e32834282b8.
|
| 15 |
Effect of common medications on the expression of SARS-CoV-2 entry receptors in liver tissue. Arch Toxicol. 2020 Dec;94(12):4037-4041. doi: 10.1007/s00204-020-02869-1. Epub 2020 Aug 17.
|
| 16 |
Drug-induced hepatic steatosis in absence of severe mitochondrial dysfunction in HepaRG cells: proof of multiple mechanism-based toxicity. Cell Biol Toxicol. 2021 Apr;37(2):151-175. doi: 10.1007/s10565-020-09537-1. Epub 2020 Jun 14.
|
| 17 |
Selection of drugs to test the specificity of the Tg.AC assay by screening for induction of the gadd153 promoter in vitro. Toxicol Sci. 2003 Aug;74(2):260-70. doi: 10.1093/toxsci/kfg113. Epub 2003 May 2.
|
| 18 |
Allopurinol induces innate immune responses through mitogen-activated protein kinase signaling pathways in HL-60 cells. J Appl Toxicol. 2016 Sep;36(9):1120-8. doi: 10.1002/jat.3272. Epub 2015 Dec 7.
|
| 19 |
Systemic drugs inducing non-immediate cutaneous adverse reactions and contact sensitizers evoke similar responses in THP-1 cells. J Appl Toxicol. 2015 Apr;35(4):398-406. doi: 10.1002/jat.3033. Epub 2014 Aug 4.
|
| 20 |
Allopurinol Protects Against Cholestatic Liver Injury in Mice Not Through Depletion of Uric Acid. Toxicol Sci. 2021 May 27;181(2):295-305. doi: 10.1093/toxsci/kfab034.
|
| 21 |
ADReCS-Target: target profiles for aiding drug safety research and application. Nucleic Acids Res. 2018 Jan 4;46(D1):D911-D917. doi: 10.1093/nar/gkx899.
|
| 22 |
Clinical Pharmacogenetics Implementation Consortium guidelines for human leukocyte antigen-B genotype and allopurinol dosing. Clin Pharmacol Ther. 2013 Feb;93(2):153-8. doi: 10.1038/clpt.2012.209. Epub 2012 Oct 17.
|
| 23 |
Angiotensin II type 1 receptor antagonist candesartan as an angiogenic inhibitor in a xenograft model of bladder cancer. Clin Cancer Res. 2006 May 1;12(9):2888-93. doi: 10.1158/1078-0432.CCR-05-2213.
|
| 24 |
Candesartan: widening indications for this angiotensin II receptor blocker Expert Opin Pharmacother. 2009 Aug;10(12):1995-2007.
|
| 25 |
The human UDP-glucuronosyltransferase UGT1A3 is highly selective towards N2 in the tetrazole ring of losartan, candesartan, and zolarsartan. Biochem Pharmacol. 2008 Sep 15;76(6):763-72.
|
| 26 |
Product Monograph of Atacand.
|
| 27 |
Substrates, inducers, inhibitors and structure-activity relationships of human Cytochrome P450 2C9 and implications in drug development. Curr Med Chem. 2009;16(27):3480-675.
|
| 28 |
Different hydrolases involved in bioactivation of prodrug-type angiotensin receptor blockers: carboxymethylenebutenolidase and carboxylesterase 1. Drug Metab Dispos. 2013 Nov;41(11):1888-95.
|
| 29 |
A nonpeptide, piperidine renin inhibitor provides renal and cardiac protection in double-transgenic mice expressing human renin and angiotensinogen genes. Cardiovasc Drugs Ther. 2008 Dec;22(6):469-78. doi: 10.1007/s10557-008-6131-x. Epub 2008 Aug 5.
|
| 30 |
The mechanism of action of angiotensin II is dependent on direct activation of vascular smooth muscle carbonic anhydrase I. Int J Clin Lab Res. 2000;30(3):119-25. doi: 10.1007/s005990070010.
|
| 31 |
Candesartan reduces oxidative stress and inflammation in patients with essential hypertension. Hypertens Res. 2003 Sep;26(9):691-7. doi: 10.1291/hypres.26.691.
|
| 32 |
Protective mechanisms of the angiotensin II type 1 receptor blocker candesartan against cerebral ischemia: in-vivo and in-vitro studies. J Hypertens. 2008 Jul;26(7):1435-45. doi: 10.1097/HJH.0b013e3283013b6e.
|
| 33 |
Telmisartan ameliorates lipopolysaccharide-induced innate immune response through peroxisome proliferator-activated receptor- activation in human monocytes. J Hypertens. 2012 Jan;30(1):87-96. doi: 10.1097/HJH.0b013e32834dde5f.
|
| 34 |
Additive beneficial effects of fenofibrate combined with candesartan in the treatment of hypertriglyceridemic hypertensive patients. Diabetes Care. 2006 Feb;29(2):195-201. doi: 10.2337/diacare.29.02.06.dc05-1418.
|
| 35 |
Distinct vascular and metabolic effects of different classes of anti-hypertensive drugs. Int J Cardiol. 2010 Apr 1;140(1):73-81. doi: 10.1016/j.ijcard.2008.11.017. Epub 2008 Dec 6.
|
| 36 |
Variants of the CYP11B2 gene predict response to therapy with candesartan. Eur J Pharmacol. 2002 Jun 7;445(1-2):151-2. doi: 10.1016/s0014-2999(02)01766-1.
|
|
|
|
|
|
|