Details of the Drug
General Information of Drug (ID: DM2HRKE)
| Drug Name |
Pentosan Polysulfate
|
||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Synonyms |
Elmiron; Pentosan sulfuric polyester; Polypentose sulfate; Elmiron (TN); Xylan,hydrogen sulfate; [(2R,3R,4S,5R)-2-hydroxy-5-[(2S,3R,4S,5R)-5-hydroxy-3,4-disulfooxyoxan-2-yl]oxy-3-sulfooxyoxan-4-yl] hydrogen sulfate
|
||||||||||||||||||||||||||
| Indication |
|
||||||||||||||||||||||||||
| Therapeutic Class |
Anticoagulants
|
||||||||||||||||||||||||||
| Affected Organisms |
Humans and other mammals
|
||||||||||||||||||||||||||
| Drug Type |
Small molecular drug
|
||||||||||||||||||||||||||
| Structure |
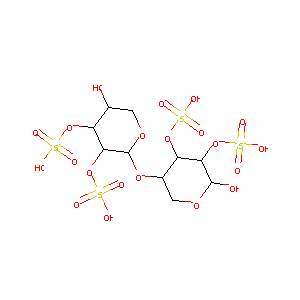 |
||||||||||||||||||||||||||
| 3D MOL | 2D MOL | ||||||||||||||||||||||||||
| #Ro5 Violations (Lipinski): 4 | Molecular Weight (mw) | 602.5 | |||||||||||||||||||||||||
| Logarithm of the Partition Coefficient (xlogp) | -5.6 | ||||||||||||||||||||||||||
| Rotatable Bond Count (rotbonds) | 10 | ||||||||||||||||||||||||||
| Hydrogen Bond Donor Count (hbonddonor) | 6 | ||||||||||||||||||||||||||
| Hydrogen Bond Acceptor Count (hbondacc) | 21 | ||||||||||||||||||||||||||
| ADMET Property |
|
||||||||||||||||||||||||||
| Chemical Identifiers |
|
||||||||||||||||||||||||||
| Cross-matching ID | |||||||||||||||||||||||||||
| Repurposed Drugs (RPD) | Click to Jump to the Detailed RPD Information of This Drug | ||||||||||||||||||||||||||
Molecular Interaction Atlas of This Drug
 Drug Therapeutic Target (DTT) |
|
|||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
 Drug Off-Target (DOT) |
|
|||||||||||||||||||||||||||||||
| Molecular Interaction Atlas (MIA) | ||||||||||||||||||||||||||||||||
Molecular Expression Atlas of This Drug
| ICD Disease Classification | 16 Disease of the genitourinary system | |||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Disease Class | ICD-11: GA10 Endometriosis | |||||||||||||||||||||||
| The Studied Tissue | Bladder tissue | |||||||||||||||||||||||
| The Studied Disease | Interstitial cystitis [ICD-11:GA10] | |||||||||||||||||||||||
|
||||||||||||||||||||||||
| Molecular Expression Atlas (MEA) | ||||||||||||||||||||||||
Drug-Drug Interaction (DDI) Information of This Drug
|
Coadministration of a Drug Treating the Disease Different from Pentosan Polysulfate (Comorbidity)
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Drug Inactive Ingredient(s) (DIG) and Formulation(s) of This Drug
References
| 1 | FDA Approved Drug Products from FDA Official Website. 2009. Application Number: (NDA) 020193. | ||||
|---|---|---|---|---|---|
| 2 | Trend Analysis of a Database of Intravenous Pharmacokinetic Parameters in Humans for 1352 Drug Compounds | ||||
| 3 | Drugs@FDA. U.S. Food and Drug Administration. U.S. Department of Health & Human Services. | ||||
| 4 | Pentosan polysulfate decreases proliferation and extracellular matrix deposition by vascular smooth muscle cells isolated from failed hemodialysis access grafts. Clin Nephrol. 2000 Aug;54(2):121-7. | ||||
| 5 | Product Information. Brukinsa (zanubrutinib). BeiGene USA, Inc, San Mateo, CA. | ||||
| 6 | Product Information. Panhematin (hemin). Recordati Rare Diseases Inc, Lebanon, NJ. | ||||
