| 1 |
Biologically active neutrophil chemokine pattern in tonsillitis.Clin Exp Immunol. 2004 Mar;135(3):511-8. doi: 10.1111/j.1365-2249.2003.02390.x.
|
| 2 |
New drugs for the treatment of cancer, 1990-2001. Isr Med Assoc J. 2002 Dec;4(12):1124-31.
|
| 3 |
URL: http://www.guidetopharmacology.org Nucleic Acids Res. 2015 Oct 12. pii: gkv1037. The IUPHAR/BPS Guide to PHARMACOLOGY in 2016: towards curated quantitative interactions between 1300 protein targets and 6000 ligands. (Ligand id: 5692).
|
| 4 |
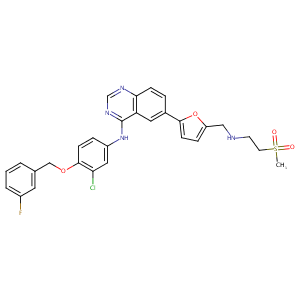
Lapatinib FDA Label
|
| 5 |
Drugs@FDA. U.S. Food and Drug Administration. U.S. Department of Health & Human Services.
|
| 6 |
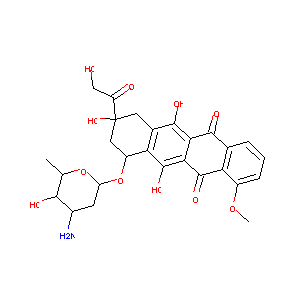
Sulindac sulfide selectively increases sensitivity of ABCC1 expressing tumor cells to doxorubicin and glutathione depletion. J Biomed Res. 2016 Mar;30(2):120-133.
|
| 7 |
Mammalian drug efflux transporters of the ATP binding cassette (ABC) family in multidrug resistance: A review of the past decade. Cancer Lett. 2016 Jan 1;370(1):153-64.
|
| 8 |
Epirubicin glucuronidation and UGT2B7 developmental expression. Drug Metab Dispos. 2006 Dec;34(12):2097-101.
|
| 9 |
Co-encapsulation of chrysophsin-1 and epirubicin in PEGylated liposomes circumvents multidrug resistance in HeLa cells. Chem Biol Interact. 2015 Dec 5;242:13-23. doi: 10.1016/j.cbi.2015.08.023. Epub 2015 Sep 1.
|
| 10 |
Camptothecin resistance: role of the ATP-binding cassette (ABC), mitoxantrone-resistance half-transporter (MXR), and potential for glucuronidation in MXR-expressing cells. Cancer Res. 1999 Dec 1;59(23):5938-46.
|
| 11 |
Preliminary study on behaviour of atrial natriuretic factor in anthracycline-related cardiac toxicity. Int J Clin Pharmacol Res. 1991;11(2):75-81.
|
| 12 |
7,3',4'-Trihydroxyisoflavone modulates multidrug resistance transporters and induces apoptosis via production of reactive oxygen species. Toxicology. 2012 Dec 16;302(2-3):221-32. doi: 10.1016/j.tox.2012.08.003. Epub 2012 Aug 15.
|
| 13 |
Early epirubicin-induced myocardial dysfunction revealed by serial tissue Doppler echocardiography: correlation with inflammatory and oxidative stress markers. Oncologist. 2007 Sep;12(9):1124-33. doi: 10.1634/theoncologist.12-9-1124.
|
| 14 |
Persistence, up to 18 months of follow-up, of epirubicin-induced myocardial dysfunction detected early by serial tissue Doppler echocardiography: correlation with inflammatory and oxidative stress markers. Oncologist. 2008 Dec;13(12):1296-305. doi: 10.1634/theoncologist.2008-0151. Epub 2008 Dec 5.
|
| 15 |
Evidence for direct involvement of epirubicin in the formation of chromosomal translocations in t(15;17) therapy-related acute promyelocytic leukemia. Blood. 2010 Jan 14;115(2):326-30. doi: 10.1182/blood-2009-07-235051. Epub 2009 Nov 2.
|
| 16 |
(-)-Gossypol enhances the anticancer activity of epirubicin via downregulating survivin in hepatocellular carcinoma. Chem Biol Interact. 2022 Sep 1;364:110060. doi: 10.1016/j.cbi.2022.110060. Epub 2022 Jul 22.
|
| 17 |
The differential effects of cyclophosphamide, epirubicin and 5-fluorouracil on apoptotic marker (CPP-32), pro-apoptotic protein (p21(WAF-1)) and anti-apoptotic protein (bcl-2) in breast cancer cells. Breast Cancer Res Treat. 2003 Aug;80(3):239-44. doi: 10.1023/A:1024995202135.
|
| 18 |
Genome-wide association study of chemotherapeutic agent-induced severe neutropenia/leucopenia for patients in Biobank Japan. Cancer Sci. 2013 Aug;104(8):1074-82. doi: 10.1111/cas.12186. Epub 2013 Jun 10.
|
| 19 |
MARVELD1 attenuates arsenic trioxide-induced apoptosis in liver cancer cells by inhibiting reactive oxygen species production. Ann Transl Med. 2019 May;7(9):200. doi: 10.21037/atm.2019.04.38.
|
| 20 |
Genome-wide association study of epirubicin-induced leukopenia in Japanese patients. Pharmacogenet Genomics. 2011 Sep;21(9):552-8. doi: 10.1097/FPC.0b013e328348e48f.
|
| 21 |
[Knock-down of apollon gene by antisense oligodeoxynucleotide inhibits the proliferation of Lovo cells and enhances chemo-sensitivity]. Yao Xue Xue Bao. 2011 Feb;46(2):138-45.
|
| 22 |
[Antisense oligonucleotide targeting survivin induces apoptosis of renal clear-cell carcinoma cells and enhances their sensitivity to epirubicin in vitro]. Zhonghua Zhong Liu Za Zhi. 2005 Aug;27(8):468-70.
|
| 23 |
Endogenous antioxidant enzymes and glutathione S-transferase in protection of mesothelioma cells against hydrogen peroxide and epirubicin toxicity. Br J Cancer. 1998 Apr;77(7):1097-102. doi: 10.1038/bjc.1998.182.
|
| 24 |
S100P contributes to chemosensitivity of human ovarian cancer cell line OVCAR3. Oncol Rep. 2008 Aug;20(2):325-32.
|
| 25 |
TSSC3 overexpression associates with growth inhibition, apoptosis induction and enhances chemotherapeutic effects in human osteosarcoma. Carcinogenesis. 2012 Jan;33(1):30-40. doi: 10.1093/carcin/bgr232. Epub 2011 Oct 21.
|
| 26 |
UGT-dependent regioselective glucuronidation of ursodeoxycholic acid and obeticholic acid and selective transport of the consequent acyl glucuronides by OATP1B1 and 1B3. Chem Biol Interact. 2019 Sep 1;310:108745. doi: 10.1016/j.cbi.2019.108745. Epub 2019 Jul 9.
|
| 27 |
Triple negative breast cancer--current status and prospective targeted treatment based on HER1 (EGFR), TOP2A and C-MYC gene assessment. Biomed Pap Med Fac Univ Palacky Olomouc Czech Repub. 2009 Mar;153(1):13-7.
|
| 28 |
Inhibition of eEF-2 kinase sensitizes human nasopharyngeal carcinoma cells to lapatinib-induced apoptosis through the Src and Erk pathways.BMC Cancer. 2016 Oct 19;16(1):813.
|
| 29 |
Tarascon Pocket Pharmacopoeia 2018 Classic Shirt-Pocket Edition.
|
| 30 |
The role of efflux and uptake transporters in [N-{3-chloro-4-[(3-fluorobenzyl)oxy]phenyl}-6-[5-({[2-(methylsulfonyl)ethyl]amino}methyl)-2-furyl]-4-quinazolinamine (GW572016, lapatinib) disposition and drug interactions. Drug Metab Dispos. 2008 Apr;36(4):695-701.
|
| 31 |
Mechanism-based inactivation of cytochrome P450 3A4 by lapatinib. Mol Pharmacol. 2010 Oct;78(4):693-703.
|
| 32 |
Clinical pharmacokinetics of tyrosine kinase inhibitors. Cancer Treat Rev. 2009 Dec;35(8):692-706.
|
| 33 |
The dual ErbB1/ErbB2 inhibitor, lapatinib (GW572016), cooperates with tamoxifen to inhibit both cell proliferation- and estrogen-dependent gene expression in antiestrogen-resistant breast cancer. Cancer Res. 2005 Jan 1;65(1):18-25.
|
| 34 |
Interference with bile salt export pump function is a susceptibility factor for human liver injury in drug development. Toxicol Sci. 2010 Dec; 118(2):485-500.
|
| 35 |
P450 3A-catalyzed O-dealkylation of lapatinib induces mitochondrial stress and activates Nrf2. Chem Res Toxicol. 2016 May 16;29(5):784-96.
|
| 36 |
Combining lapatinib (GW572016), a small molecule inhibitor of ErbB1 and ErbB2 tyrosine kinases, with therapeutic anti-ErbB2 antibodies enhances apoptosis of ErbB2-overexpressing breast cancer cells. Oncogene. 2005 Sep 15;24(41):6213-21. doi: 10.1038/sj.onc.1208774.
|
| 37 |
CDK4/6 inhibition provides a potent adjunct to Her2-targeted therapies in preclinical breast cancer models. Genes Cancer. 2014 Jul;5(7-8):261-72. doi: 10.18632/genesandcancer.24.
|
| 38 |
Effects of lapatinib on cell proliferation and apoptosis in NB4 cells. Oncol Lett. 2018 Jan;15(1):235-242. doi: 10.3892/ol.2017.7342. Epub 2017 Nov 3.
|
| 39 |
The involvement of hepatic cytochrome P450s in the cytotoxicity of lapatinib. Toxicol Sci. 2023 Dec 21;197(1):69-78. doi: 10.1093/toxsci/kfad099.
|
| 40 |
Evaluating the Role of Multidrug Resistance Protein 3 (MDR3) Inhibition in Predicting Drug-Induced Liver Injury Using 125 Pharmaceuticals. Chem Res Toxicol. 2017 May 15;30(5):1219-1229. doi: 10.1021/acs.chemrestox.7b00048. Epub 2017 May 4.
|
| 41 |
Suppression of HER2/HER3-mediated growth of breast cancer cells with combinations of GDC-0941 PI3K inhibitor, trastuzumab, and pertuzumab. Clin Cancer Res. 2009 Jun 15;15(12):4147-56. doi: 10.1158/1078-0432.CCR-08-2814. Epub 2009 Jun 9.
|
| 42 |
Cytotoxicity of 34 FDA approved small-molecule kinase inhibitors in primary rat and human hepatocytes. Toxicol Lett. 2018 Jul;291:138-148. doi: 10.1016/j.toxlet.2018.04.010. Epub 2018 Apr 12.
|
| 43 |
Association of CYP1A1 and CYP1B1 inhibition in in vitro assays with drug-induced liver injury. J Toxicol Sci. 2021;46(4):167-176. doi: 10.2131/jts.46.167.
|
| 44 |
The K-Ras effector p38 MAPK confers intrinsic resistance to tyrosine kinase inhibitors by stimulating EGFR transcription and EGFR dephosphorylation. J Biol Chem. 2017 Sep 8;292(36):15070-15079. doi: 10.1074/jbc.M117.779488. Epub 2017 Jul 24.
|
| 45 |
HLA-DQA1*02:01 is a major risk factor for lapatinib-induced hepatotoxicity in women with advanced breast cancer. J Clin Oncol. 2011 Feb 20;29(6):667-73. doi: 10.1200/JCO.2010.31.3197. Epub 2011 Jan 18.
|
| 46 |
Niclosamide inhibits epithelial-mesenchymal transition and tumor growth in lapatinib-resistant human epidermal growth factor receptor 2-positive breast cancer. Int J Biochem Cell Biol. 2016 Feb;71:12-23. doi: 10.1016/j.biocel.2015.11.014. Epub 2015 Nov 28.
|
| 47 |
Recurrent recessive mutation in deoxyguanosine kinase causes idiopathic noncirrhotic portal hypertension.Hepatology. 2016 Jun;63(6):1977-86. doi: 10.1002/hep.28499. Epub 2016 Mar 31.
|
| 48 |
Loss of function mutations in VARS encoding cytoplasmic valyl-tRNA synthetase cause microcephaly, seizures, and progressive cerebral atrophy.Hum Genet. 2018 Apr;137(4):293-303. doi: 10.1007/s00439-018-1882-3. Epub 2018 Apr 24.
|
|
|
|
|
|
|