Details of the Drug
General Information of Drug (ID: DM6IR3P)
| Drug Name |
Liothyronine
|
|||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Synonyms |
liothyronine; 3,3',5-Triiodo-L-thyronine; 6893/2/3; Liothyronin; Tresitope; 3,5,3'-triiodothyronine; L-Liothyronine; O-(4-Hydroxy-3-iodophenyl)-3,5-diiodo-L-tyrosine; triothyrone; Liothyroninum; Liotironina; 3,5,3'-Triiodo-L-thyronine; L-T3; T3; 3,3',5-Triiodothyronine; Triiodo-L-thyronine; Lyothyronine; 3,5,3'TRIIODOTHYRONINE; L-3,5,3'-Triiodothyronine; (2S)-2-amino-3-[4-(4-hydroxy-3-iodophenoxy)-3,5-diiodophenyl]propanoic acid; T3 (amino acid); T3 (Hormone); L-3,3',5-TriioDOThyronine; Liothyronine [INN:BAN]; Rathyronine; Triothyrone; THYROID HORMONE; Liothyronine I 131; Liothyronine I 131 [USAN]; T3 liothyronine; Cytomel (TN); Euthroid-1; Euthroid-2; Euthroid-3; Liothyronine (INN); Liothyroninum[INN-Latin]; Liotironina [INN-Spanish]; T3 (Triiodothyronine); T3 (VAN); Tertroxin (TN); Thyrolar-1; Thyrolar-2; Thyrolar-3; Tri-Thyrotope; Triiodothyronine (T3); Triomet-131; Euthroid-05; Thyrolar-025; Thyrolar-05; TRIIODOTHYRONINE (T3 OR LIOTHYRONINE, ACTIVE) (6-11%); Thyronine, 3,3',5-triiodo-, L-(6CI); L-3-(4-(4-Hydroxy-3-iodophenoxy)-3,5-diiodophenyl)alanine; L-Tyrosine, O-(4-hydroxy-3-iodophenyl)-3,5-diiodo-(9CI); 3,3'5-Triiodo-L-thyronine; 3,5,3'-Tri-iodo-L-thyronine; 4-(3-Iodo-4-hydroxyphenoxy)-3,5-diiodophenylalanine; 4-(4-Hydroxy-3-iodophenoxy)-3,5-diiodophenylalanine; 4-(4-hydroxy-3-iodophenoxy)-3,5-diiodo-L-phenylalanine
|
|||||||||||||||||||||||||||||||||||||||||||||||
| Indication |
|
|||||||||||||||||||||||||||||||||||||||||||||||
| Therapeutic Class |
Hormone Replacement Agents
|
|||||||||||||||||||||||||||||||||||||||||||||||
| Drug Type |
Small molecular drug
|
|||||||||||||||||||||||||||||||||||||||||||||||
| Structure |
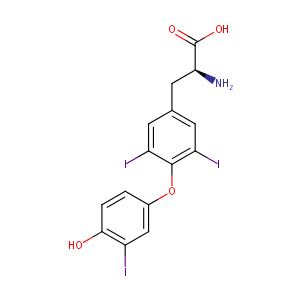 |
|||||||||||||||||||||||||||||||||||||||||||||||
| 3D MOL | 2D MOL | |||||||||||||||||||||||||||||||||||||||||||||||
| #Ro5 Violations (Lipinski): 1 | Molecular Weight (mw) | 650.97 | ||||||||||||||||||||||||||||||||||||||||||||||
| Logarithm of the Partition Coefficient (xlogp) | 1.7 | |||||||||||||||||||||||||||||||||||||||||||||||
| Rotatable Bond Count (rotbonds) | 5 | |||||||||||||||||||||||||||||||||||||||||||||||
| Hydrogen Bond Donor Count (hbonddonor) | 3 | |||||||||||||||||||||||||||||||||||||||||||||||
| Hydrogen Bond Acceptor Count (hbondacc) | 5 | |||||||||||||||||||||||||||||||||||||||||||||||
| ADMET Property | ||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical Identifiers |
|
|||||||||||||||||||||||||||||||||||||||||||||||
| Cross-matching ID | ||||||||||||||||||||||||||||||||||||||||||||||||
| Combinatorial Drugs (CBD) | Click to Jump to the Detailed CBD Information of This Drug | |||||||||||||||||||||||||||||||||||||||||||||||
| Repurposed Drugs (RPD) | Click to Jump to the Detailed RPD Information of This Drug | |||||||||||||||||||||||||||||||||||||||||||||||
Molecular Interaction Atlas of This Drug
 Drug Therapeutic Target (DTT) |
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
 Drug Transporter (DTP) |
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
 Drug-Metabolizing Enzyme (DME) |
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
 Drug Off-Target (DOT) |
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Molecular Interaction Atlas (MIA) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Drug-Drug Interaction (DDI) Information of This Drug
|
Coadministration of a Drug Treating the Disease Different from Liothyronine (Comorbidity)
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Drug Inactive Ingredient(s) (DIG) and Formulation(s) of This Drug
References
| 1 | Liothyronine FDA Label | ||||
|---|---|---|---|---|---|
| 2 | Drugs@FDA. U.S. Food and Drug Administration. U.S. Department of Health & Human Services. 2015 | ||||
| 3 | ClinicalTrials.gov (NCT00790738) Liothyronine (T3) for Bipolar Depression. U.S. National Institutes of Health. | ||||
| 4 | BDDCS predictions, self-correcting aspects of BDDCS assignments, BDDCS assignment corrections, and classification for more than 175 additional drugs | ||||
| 5 | Estimating the safe starting dose in phase I clinical trials and no observed effect level based on QSAR modeling of the human maximum recommended daily dose | ||||
| 6 | Evaluation of thyroid hormone action in a case of generalized resistance to thyroid hormone with chronic thyroiditis: discovery of a novel heterozy... Endocr J. 2007 Dec;54(5):727-32. | ||||
| 7 | Involvement of multispecific organic anion transporter, Oatp14 (Slc21a14), in the transport of thyroxine across the blood-brain barrier. Endocrinology. 2004 Sep;145(9):4384-91. | ||||
| 8 | Isolation and characterization of a digoxin transporter and its rat homologue expressed in the kidney. Proc Natl Acad Sci U S A. 2004 Mar 9;101(10):3569-74. | ||||
| 9 | Identification of thyroid hormone transporters. Biochem Biophys Res Commun. 1999 Jan 19;254(2):497-501. | ||||
| 10 | Identification of thyroid hormone transporters in humans: different molecules are involved in a tissue-specific manner. Endocrinology. 2001 May;142(5):2005-12. | ||||
| 11 | Identification of a novel gene family encoding human liver-specific organic anion transporter LST-1. J Biol Chem. 1999 Jun 11;274(24):17159-63. | ||||
| 12 | LST-2, a human liver-specific organic anion transporter, determines methotrexate sensitivity in gastrointestinal cancers. Gastroenterology. 2001 Jun;120(7):1689-99. | ||||
| 13 | FDA Label of Liothyronine sodium. The 2020 official website of the U.S. Food and Drug Administration. | ||||
| 14 | Characterization of human liver thermostable phenol sulfotransferase (SULT1A1) allozymes with 3,3',5-triiodothyronine as the substrate. J Endocrinol. 2001 Dec;171(3):525-32. | ||||
| 15 | Similarities and differences between two modes of antagonism of the thyroid hormone receptor. ACS Chem Biol. 2011 Oct 21;6(10):1096-106. | ||||
| 16 | Monitoring of deiodinase deficiency based on transcriptomic responses in SH-SY5Y cells. Arch Toxicol. 2013 Jun;87(6):1103-13. doi: 10.1007/s00204-013-1018-4. Epub 2013 Feb 10. | ||||
| 17 | In vitro fluorescence displacement investigation of thyroxine transport disruption by bisphenol A. J Environ Sci (China). 2011;23(2):315-21. doi: 10.1016/s1001-0742(10)60408-1. | ||||
| 18 | Thyroid hormone responsive genes in cultured human fibroblasts. J Clin Endocrinol Metab. 2005 Feb;90(2):936-43. | ||||
| 19 | Havrankova J, Lahaie R "Levothyroxine binding by sucralfate." Ann Intern Med 117 (1992): 445-6. [PMID: 1503346] | ||||
| 20 | Cerner Multum, Inc. "UK Summary of Product Characteristics.". | ||||
| 21 | Cerner Multum, Inc. "Australian Product Information.". | ||||
| 22 | Arafah BM "Increased need for thyroxine in women with hypothyroidism during estrogen therapy." N Engl J Med 344 (2001): 1743-9. [PMID: 11396440] | ||||
| 23 | Costigan DC, Freedman MH, Ehrlich RM "Potentiation of oral anticoagulant effect by l-thyroxine." Clin Pediatr (Phila) 23 (1984): 172-4. [PMID: 6697623] | ||||
| 24 | Frye RL, Braunwald E "Studies on digitalis. III: The influence of triiodothyronine on digitalis requirements." Circulation 23 (1961): 376-82. [PMID: 13702336] | ||||
| 25 | Campbell NR, Hasinoff BB, Stalts H, Rao B, Wong NC "Ferrous sulfate reduces thyroxine efficacy in patients with hypothyroidism." Ann Intern Med 117 (1992): 1010-3. [PMID: 1443969] | ||||
| 26 | Filippatos TD, Derdemezis CS, Gazi IF, Nakou ES, Mikhailidis DP, Elisaf MS "Orlistat-associated adverse effects and drug interactions: a critical review." Drug Saf 31 (2008): 53-65. [PMID: 18095746] | ||||
