Details of the Drug
General Information of Drug (ID: DMF8DNE)
| Drug Name |
Framycetin
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Synonyms |
Neomycin; neomycin; NEOMYCIN B; Soframycin; Actiline; Mycifradin; Fradiomycin; Soframycine; Framycetinum; Framycetine; Framicetina; Neomas; Fradiomycinum; Antibiotique; Nivemycin; Actilin; Neolate; Enterfram; Myacyne; Framygen; Caswell No 595; Vonamycin powder V; Neomycin B sulfate; Neomin; Neomcin; Fradiomycin B; Neo-Rx; Neomicina [DCIT]; Framycetinum [INN-Latin]; PIMAVECORT; Neobrettin; Neo-Fradin; 119-04-0; Neomycine [INN-French]; Neomycinum [INN-Latin]; Framycetine [INN-French]; Framicetina [INN-Spanish]; USAF CB-19; Endomixin; Fraquinol; Myacine; Myciguent; NMY; Neobiotic; Neomicina; Neomycinum; Tuttomycin; VONAMYCIN; NEOMYCIN AND POLYMYXIN B SULFATES; NEOMYCIN SULFATE; Neomycin solution; Soframycin Ophthalmic; Antibiotic 10676; Antibiotic produced by Streptomyces decaris Neomycin B; Framycetin (INN); Soframycin (TN); Framycetin [INN:BAN:DCF]; Sofra-Tulle (TN); BDG-(1-4)CYY-(5-1)RIB-(3-1)IDG; BDG-(1-4)NEB-(5-1)RIB-(3-1)NED; (1R,2R,3S,4R,6S)-4,6-diamino-2-{[3-O-(2,6-diamino-2,6-dideoxy-beta-L-idopyranosyl)-beta-D-ribofuranosyl]oxy}-3-hydroxycyclohexyl 2,6-diamino-2,6-dideoxy-alpha-D-glucopyranoside; (2R,3S,4R,5R,6R)-5-amino-2-(aminomethyl)-6-[(1R,2R,3S,4R,6S)-4,6-diamino-2-[(2S,3R,4S,5R)-4-[(2R,3R,4R,5S,6S)-3-amino-6-(aminomethyl)-4,5-dihydroxyoxan-2-yl]oxy-3-hydroxy-5-(hydroxymethyl)oxolan-2-yl]oxy-3-hydroxycyclohexyl]oxyoxane-3,4-diol; D-Streptamine, O-2,6-diamino-2,6-dideoxy-beta-L-idopyranosyl-(1->3)-O-beta-D-ribofuranosyl-(1->5)]-O-[2,6-diamino-2,6-dideoxy-alpha-D-glucopyranosyl-(1->4)]-2-deoxy
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Indication |
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Therapeutic Class |
Antibiotics
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Affected Organisms |
Enteric bacteria and other eubacteriaEscherichia coliKlebsiellaEnterobacterBacteroidesPseudomonas aeruginosaHaemophilus influenzaeEnterobacter spp.
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ATC Code |
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Drug Type |
Small molecular drug
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Structure |
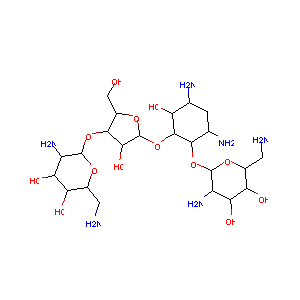 |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 3D MOL | 2D MOL | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| #Ro5 Violations (Lipinski): 4 | Molecular Weight (mw) | 614.6 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Logarithm of the Partition Coefficient (xlogp) | -9 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Rotatable Bond Count (rotbonds) | 9 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hydrogen Bond Donor Count (hbonddonor) | 13 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hydrogen Bond Acceptor Count (hbondacc) | 19 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ADMET Property |
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Adverse Drug Reaction (ADR) |
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical Identifiers |
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Cross-matching ID | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Combinatorial Drugs (CBD) | Click to Jump to the Detailed CBD Information of This Drug | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Repurposed Drugs (RPD) | Click to Jump to the Detailed RPD Information of This Drug | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Molecular Interaction Atlas of This Drug
 Drug Therapeutic Target (DTT) |
|
||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
 Drug-Metabolizing Enzyme (DME) |
|
||||||||||||||||||||||||||||||||||||
 Drug Off-Target (DOT) |
|
||||||||||||||||||||||||||||||||||||
| Molecular Interaction Atlas (MIA) | |||||||||||||||||||||||||||||||||||||
Drug-Drug Interaction (DDI) Information of This Drug
|
Coadministration of a Drug Treating the Disease Different from Framycetin (Comorbidity)
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Drug Inactive Ingredient(s) (DIG) and Formulation(s) of This Drug
References
| 1 | URL: http://www.guidetopharmacology.org Nucleic Acids Res. 2015 Oct 12. pii: gkv1037. The IUPHAR/BPS Guide to PHARMACOLOGY in 2016: towards curated quantitative interactions between 1300 protein targets and 6000 ligands. (Ligand id: 709). | ||||
|---|---|---|---|---|---|
| 2 | Drug information of Framycetin, 2008. eduDrugs. | ||||
| 3 | A standard database for drug repositioning. Sci Data. 2017 Mar 14;4:170029. | ||||
| 4 | Bacterial isolates, antimicrobial susceptibility, and clinical characteristics of bacterial keratitis in dogs presenting to referral practice in Australia. Vet Ophthalmol. 2016 Sep;19(5):418-26. | ||||
| 5 | Prostatectomy and infection. J Urol. 1971 Dec;106(6):910-2. | ||||
| 6 | BDDCS applied to over 900 drugs | ||||
| 7 | Estimating the safe starting dose in phase I clinical trials and no observed effect level based on QSAR modeling of the human maximum recommended daily dose | ||||
| 8 | ADReCS-Target: target profiles for aiding drug safety research and application. Nucleic Acids Res. 2018 Jan 4;46(D1):D911-D917. doi: 10.1093/nar/gkx899. | ||||
| 9 | Antistaphylococcal activity of gentamicin. Minerva Med. 1975 Dec 8;66(84):4505-26. | ||||
| 10 | Characterization of a 30S ribosomal subunit assembly intermediate found in Escherichia coli cells growing with neomycin or paromomycin. Arch Microbiol. 2008 May;189(5):441-9. | ||||
| 11 | Relationship between antimicrobial resistance and aminoglycoside-modifying enzyme gene expressions in Acinetobacter baumannii. Chin Med J (Engl). 2005 Jan 20;118(2):141-5. | ||||
| 12 | A plasmacytoid dendritic cell (CD123+/CD11c-) based assay system to predict contact allergenicity of chemicals. Toxicology. 2009 Oct 1;264(1-2):1-9. doi: 10.1016/j.tox.2009.07.021. Epub 2009 Aug 7. | ||||
| 13 | Cerner Multum, Inc. "Australian Product Information.". | ||||
| 14 | Agencia Espaola de Medicamentos y Productos Sanitarios Healthcare "Centro de informacion online de medicamentos de la AEMPS - CIMA.". | ||||
| 15 | Cerner Multum, Inc. "UK Summary of Product Characteristics.". | ||||
| 16 | FDA. U.S. Food and Drug Administration "FDA Drug Safety Communication: Low magnesium levels can be associated with long-term use of proton pump inhibitor drugs (PPIs).". | ||||
| 17 | Engle JE, Drago J, Carlin B, Schoolwerth AC "Letter: Reversible acute renal failure after cephalothin." Ann Intern Med 83 (1975): 232-3. [PMID: 1147461] | ||||
| 18 | Chang JT, Green L, Beitz J "Renal failure with the use of zoledronic acid." N Engl J Med 349 (2003): 1676-9 discussion 1676-9. [PMID: 14573746] | ||||
| 19 | Wong GT, Lee EY, Irwin MG. Contrast induced nephropathy in vascular surgery.?Br J Anaesth. 2016;117 Suppl 2:ii63-ii73. [PMID: 27566809] | ||||
| 20 | Burkett L, Bikhazi GB, Thomas KC Jr, Rosenthal DA, Wirta MG, Foldes FF "Mutual potentiation of the neuromuscular effects of antibiotics and relaxants." Anesth Analg 58 (1979): 107-15. [PMID: 571233] | ||||
| 21 | Product Information. CellCept (mycophenolate mofetil). Roche Laboratories, Nutley, NJ. | ||||
| 22 | Novis BH, Korzets Z, Chen P, Bernheim J "Nephrotic syndrome after treatment with 5-aminosalicylic acid." Br Med J (Clin Res Ed) 296 (1988): 1442. [PMID: 3132281] | ||||
| 23 | Product Information. Exjade (deferasirox). Novartis Pharmaceuticals, East Hanover, NJ. | ||||
| 24 | Product Information. Prograf (tacrolimus). Fujisawa, Deerfield, IL. | ||||
