Details of the Drug
General Information of Drug (ID: DMREUQ6)
| Drug Name |
Valsartan
|
||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Synonyms |
valsartan; 137862-53-4; Diovan; Tareg; Provas; L-Valsartan; CGP 48933; Exforge; CGP-48933; (S)-2-(N-((2'-(1H-Tetrazol-5-yl)-[1,1'-biphenyl]-4-yl)methyl)pentanamido)-3-methylbutanoic acid; UNII-80M03YXJ7I; N-(p-(o-1H-Tetrazol-5-ylphenyl)benzyl)-N-valeryl-L-valine; CHEMBL1069; 80M03YXJ7I; CHEBI:9927; C24H29N5O3; (2S)-3-methyl-2-[pentanoyl-[[4-[2-(2H-tetrazol-5-yl)phenyl]phenyl]methyl]amino]butanoic acid; N-(1-oxopentyl)-N-[[2'-(1H-tetrazol-5-yl)[1,1'-biphenyl]-4-yl]methyl]-L-valine; 137863-60-6; AK-58790; Kalpress; Miten; Nisis; Diovan; Vals; Valsarran; Walsartan; Aventis brand of valsartan; CEPA brand of valsartan; Esteve brand of valsartan; Lacer brand of valsartan; Novartis brand of valsartan; Sanol brand of valsartan; Schwarz brand of valsartan; Diovan (TN); Diovan, Valsartan; Valsartan [USAN:INN]; Valtan (TN); Valzaar (TN); Valsartan (JAN/USAN/INN); N-valeryl-N-((2'-(1H-tetrazol-5-yl)biphenyl-4-yl)methyl)valine; N-pentanoyl-N-{[2'-(1H-tetrazol-5-yl)biphenyl-4-yl]methyl}-L-valine; N-pentanoyl-N-{[2'-(1H-tetrazol-5-yl)[1,1'-biphenyl]-4-yl]methyl}-L-valine; L-Valine, N-(1-oxopentyl)-N-[[2'-(1H-tetrazol-5-yl)[1,1'-biphenyl]-4-yl]methyl]-(9CI); (S)-N-valeryl-N-{[2'-(1H-tetrazol-5-yl)biphenyl-4-yl]-methyl}-valine; (s)-2-(n-((2'-(1h-tetrazol-5-yl)biphenyl-4-yl)methyl)pentanamido)-3-methylbutanoic acid; [3H]valsartan
|
||||||||||||||||||||||||||||||||||
| Indication |
|
||||||||||||||||||||||||||||||||||
| Therapeutic Class |
Antihypertensive Agents
|
||||||||||||||||||||||||||||||||||
| Affected Organisms |
Humans and other mammals
|
||||||||||||||||||||||||||||||||||
| ATC Code | |||||||||||||||||||||||||||||||||||
| Drug Type |
Small molecular drug
|
||||||||||||||||||||||||||||||||||
| Structure |
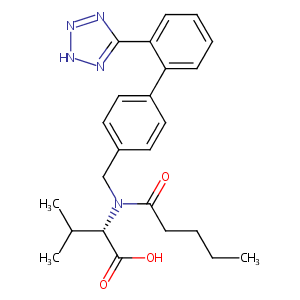 |
||||||||||||||||||||||||||||||||||
| 3D MOL | 2D MOL | ||||||||||||||||||||||||||||||||||
| #Ro5 Violations (Lipinski): 0 | Molecular Weight (mw) | 435.5 | |||||||||||||||||||||||||||||||||
| Logarithm of the Partition Coefficient (xlogp) | 4.4 | ||||||||||||||||||||||||||||||||||
| Rotatable Bond Count (rotbonds) | 10 | ||||||||||||||||||||||||||||||||||
| Hydrogen Bond Donor Count (hbonddonor) | 2 | ||||||||||||||||||||||||||||||||||
| Hydrogen Bond Acceptor Count (hbondacc) | 6 | ||||||||||||||||||||||||||||||||||
| ADMET Property |
|
||||||||||||||||||||||||||||||||||
| Chemical Identifiers |
|
||||||||||||||||||||||||||||||||||
| Cross-matching ID | |||||||||||||||||||||||||||||||||||
| Combinatorial Drugs (CBD) | Click to Jump to the Detailed CBD Information of This Drug | ||||||||||||||||||||||||||||||||||
| Repurposed Drugs (RPD) | Click to Jump to the Detailed RPD Information of This Drug | ||||||||||||||||||||||||||||||||||
Molecular Interaction Atlas of This Drug
 Drug Therapeutic Target (DTT) |
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
 Drug Transporter (DTP) |
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
 Drug-Metabolizing Enzyme (DME) |
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
 Drug Off-Target (DOT) |
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Molecular Interaction Atlas (MIA) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Molecular Expression Atlas of This Drug
| The Studied Disease | Chronic heart failure | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ICD Disease Classification | BD1Z | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Molecular Expression Atlas (MEA) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Drug-Drug Interaction (DDI) Information of This Drug
|
Coadministration of a Drug Treating the Same Disease as Valsartan
Coadministration of a Drug Treating the Disease Different from Valsartan (Comorbidity)
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Drug Inactive Ingredient(s) (DIG) and Formulation(s) of This Drug
References
| 1 | Valsartan FDA Label | ||||
|---|---|---|---|---|---|
| 2 | URL: http://www.guidetopharmacology.org Nucleic Acids Res. 2015 Oct 12. pii: gkv1037. The IUPHAR/BPS Guide to PHARMACOLOGY in 2016: towards curated quantitative interactions between 1300 protein targets and 6000 ligands. (Ligand id: 3937). | ||||
| 3 | Valsartan for Prevention of Acute Respiratory Distress Syndrome in Hospitalized Patients With SARS-COV-2 (COVID-19) Infection Disease | ||||
| 4 | BDDCS applied to over 900 drugs | ||||
| 5 | Trend Analysis of a Database of Intravenous Pharmacokinetic Parameters in Humans for 1352 Drug Compounds | ||||
| 6 | Estimating the safe starting dose in phase I clinical trials and no observed effect level based on QSAR modeling of the human maximum recommended daily dose | ||||
| 7 | Radioligand binding assays: application of [(125)I]angiotensin II receptor binding. Methods Mol Biol. 2009;552:131-41. | ||||
| 8 | High-affinity interaction of sartans with H+/peptide transporters. Drug Metab Dispos. 2009 Jan;37(1):143-9. | ||||
| 9 | Involvement of transporters in the hepatic uptake and biliary excretion of valsartan, a selective antagonist of the angiotensin II AT1-receptor, in humans. Drug Metab Dispos. 2006 Jul;34(7):1247-54. | ||||
| 10 | Regulation of Organic Anion Transporting Polypeptides (OATP) 1B1- and OATP1B3-Mediated Transport: An Updated Review in the Context of OATP-Mediated Drug-Drug Interactions. Int J Mol Sci. 2018 Mar 14;19(3). pii: E855. | ||||
| 11 | Prediction of the overall renal tubular secretion and hepatic clearance of anionic drugs and a renal drug-drug interaction involving organic anion transporter 3 in humans by in vitro uptake experiments. Drug Metab Dispos. 2011 Jun;39(6):1031-8. | ||||
| 12 | In vitro inhibition screening of human hepatic P450 enzymes by five angiotensin-II receptor antagonists. Eur J Clin Pharmacol. 2000 May;56(2):135-40. | ||||
| 13 | Is renoprotection by angiotensin receptor blocker dependent on blood pressure?: the Saitama Medical School, Albuminuria Reduction in Diabetics with Valsartan (STAR) study. Hypertens Res. 2007 Jun;30(6):529-33. doi: 10.1291/hypres.30.529. | ||||
| 14 | Valsartan improves adipose tissue function in humans with impaired glucose metabolism: a randomized placebo-controlled double-blind trial. PLoS One. 2012;7(6):e39930. doi: 10.1371/journal.pone.0039930. Epub 2012 Jun 29. | ||||
| 15 | Angiotensin II receptor blockade in normotensive subjects: A direct comparison of three AT1 receptor antagonists. Hypertension. 1999 Mar;33(3):850-5. doi: 10.1161/01.hyp.33.3.850. | ||||
| 16 | Antiviral effect of Bosentan and Valsartan during coxsackievirus B3 infection of human endothelial cells. J Gen Virol. 2010 Aug;91(Pt 8):1959-1970. doi: 10.1099/vir.0.020065-0. Epub 2010 Apr 14. | ||||
| 17 | Comparing antihypertensive effect and plasma ciclosporin concentration between amlodipine and valsartan regimens in hypertensive renal transplant patients receiving ciclosporin therapy. Am J Cardiovasc Drugs. 2011 Dec 1;11(6):401-9. doi: 10.2165/11593800-000000000-00000. | ||||
| 18 | Health Canada "Potential risks of cardiovascular and renal adverse events in patients with type 2 diabetes treated with aliskiren (RASILEZ) or aliskiren/hydrochlorothiazide (RASILEZ HCT)." . | ||||
| 19 | Product Information. Diovan (valsartan). Novartis Pharmaceuticals, East Hanover, NJ. | ||||
| 20 | Product Information. Micardis (telmisartan). Boehringer-Ingelheim, Ridgefield, CT. | ||||
| 21 | Abad S, Moachon L, Blanche P, Bavoux F, Sicard D, Salmon-Ceron D "Possible interaction between glicazide, fluconazole and sulfamethoxazole resulting in severe hypoglycaemia." Br J Clin Pharmacol 52 (2001): 456-7. [PMID: 11678792] | ||||
| 22 | Asplund K, Wiholm BE, Lithner F "Glibenclamide-associated hypoglycaemia: a report on 57 cases." Diabetologia 24 (1983): 412-7. [PMID: 6411511] | ||||
| 23 | Cerner Multum, Inc. "UK Summary of Product Characteristics.". | ||||
| 24 | Aronowitz JS, Chakos MH, Safferman AZ, Lieberman JA "Syncope associated with the combination of clozapine and enalapril." J Clin Psychopharmacol 14 (1994): 429-30. [PMID: 7884028] | ||||
| 25 | Ban TA "Drug interactions with psychoactive drugs." Dis Nerv Syst 36 (1975): 164-6. [PMID: 1116424] | ||||
| 26 | Cerner Multum, Inc. "Australian Product Information.". | ||||
| 27 | Warrington SJ, Ankier SI, Turner P "Evaluation of possible interactions between ethanol and trazodone or amitriptyline." Neuropsychobiology 15 (1986): 31-7. [PMID: 3725002] | ||||
| 28 | Product Information. Yasmin (drospirenone-ethinyl estradiol) Berlex Laboratories, Richmond, CA. | ||||
| 29 | Product Information. Midamor (amiloride). Merck & Co, Inc, West Point, PA. | ||||
| 30 | Product Information. Inspra (eplerenone). Searle, Chicago, IL. | ||||
| 31 | Walmsley RN, White GH, Cain M, McCarthy PJ, Booth J "Hyperkalemia in the elderly." Clin Chem 30 (1984): 1409-12. [PMID: 6744599] | ||||
| 32 | Product Information. Samsca (tolvaptan). Otsuka American Pharmaceuticals Inc, Rockville, MD. | ||||
| 33 | Blakely KM, Drucker AM, Rosen CF "Drug-induced photosensitivity-an update: Culprit drugs, prevention and management." Drug Saf 42 (2019): 827-47. [PMID: 30888626] | ||||
| 34 | Allred AJ, Bowen CJ, Park JW, et al. "Eltrombopag increases plasma rosuvastatin exposure in healthy volunteers." Br J Clin Pharmacol 72 (2011): 321-9. [PMID: 21434975] | ||||
| 35 | Perazella MA "Drug-induced hyperkalemia: old culprits and new offenders." Am J Med 109 (2000): 307-14. [PMID: 10996582] | ||||
| 36 | Canaday DH, Johnson JR "Hyperkalemia in elderly patients receiving standard doses of trimethoprim-sulfamethoxazole." Ann Intern Med 120 (1994): 438. [PMID: 8304666] | ||||
| 37 | Product Information. Clozaril (clozapine). Novartis Pharmaceuticals, East Hanover, NJ. | ||||
