Details of the Drug
General Information of Drug (ID: DM2R68E)
| Drug Name |
Ramipril
|
||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Synonyms |
Acovil; Altace; Carasel; Cardace; Delix; Hypren; Hytren; Lostapres; Naprix; Pramace; Quark; Ramace; Ramiprilum; Triatec; Tritace; Vesdil; Zabien; Almirall Brand of Ramipril; Astra Brand of Ramipril; AstraZeneca Brand of Ramipril; Aventis Brand of Ramipril; Aventis Pharma Brand of Ramipril; Hoechst Brand of Ramipril; Monarch Brand of Ramipril; Promed Brand of Ramipril; Ramiprilum [Latin]; HOE 498; HOE498; Altace (TN); Hoe-498; Pramace (discontinued); Tritace (TN); Ramipril (USP/INN); Ramipril [USAN:INN:BAN]; Ramipro, Tritace, Altace, Prilace, Ramipril; N-(1S-carboethoxy-3-phenylpropyl)-S-alanyl-cis,endo-2-azabicyclo[3.3.0]octane-3S-carboxylic Acid; [2s,3as,6as]-1-[(2s)-2-[[(1s)-1-(ethoxycarbonyl)-3-phenylpropyl]amino]-1-oxopropyl]octahydrocyclopen; [2S,3aS,6aS]-1-[(2S)-2-[[(1S)-1-(Ethoxycarbonyl)-3-phenylpropyl]amino]-1-oxopropyl]octahydrocyclopenta[b]pyrrole-2-carboxylic acid; (2 S ,3 aS,6 aS)-1[(S)-N-[(S)-1-Carboxy-3-phenylpropyl] alanyl] octahydrocyclopenta [ b ]pyrrole-2-carboxylic acid, 1-ethyl ester; (2S,3aS,6aS)-1-((S)-2-((S)-1-ethoxy-1-oxo-4-phenylbutan-2-ylamino)propanoyl) octahydrocyclopenta[b]pyrrole-2-carboxylic acid; (2S,3aS,6aS)-1-((S)-N-((S)-1-Carboxy-3-phenylpropyl)alanyl)octahydrocyclopenta(b)pyrrole-2-carboxylic acid, 1-ethyl ester; (2S,3aS,6aS)-1-((S)-N-((S)-1-Ethoxycarbonyl-3-phenylpropyl)alanyl)octahydrocyclopenta(b)pyrrol-2-carbonsaeure; (2S,3aS,6aS)-1-[(2S)-2-[[(2S)-1-ethoxy-1-oxo-4-phenylbutan-2-yl]amino]propanoyl]-3,3a,4,5,6,6a-hexahydro-2H-cyclopenta[b]pyrrole-2-carboxylic acid; (2S,3aS,6aS)-1-[(2S)-2-{[(1S)-1-ethoxycarbonyl-3-phenylpropyl]amino}propanoyl]octahydrocyclopenta[b]pyrrole-2-carboxylic acid; (2S,3aS,6aS)-1-[(2S)-2-{[(2S)-1-ethoxy-1-oxo-4-phenylbutan-2-yl]amino}propanoyl]octahydrocyclopenta[b]pyrrole-2-carboxylic acid (non-preferred name); (2S-(1(R*(R*)),2alpha,3abeta,6abeta))-1-(2-((1-(Ethoxycarbonyl)-3-phenylpropyl)amino)-1-oxopropyl)octahydrocyclopenta(b)pyrrole-2-carboxylic acid; (2s,3as,6as)-1((s)-n-((s)-1-carboxy-3-phenylpropyl)alanyl)octahydrocyclopenta(b)pyrrole-2-carboxylic; (2s,3as,6as)-1-((s)-2-((s)-1-ethoxy-1-oxo-4-phenylbutan-2-ylamino)propanoyl)-octahydrocyclopenta[b]p; (2s,3as,6as)-1-[(s)-2-((s)-1-ethoxycarbonyl-3-phenyl-propylamino)-propionyl]-octahydro-cyclopenta[b]
|
||||||||||||||||||||||||||||||||||||||
| Indication |
|
||||||||||||||||||||||||||||||||||||||
| Therapeutic Class |
Antihypertensive Agents
|
||||||||||||||||||||||||||||||||||||||
| Affected Organisms |
Humans and other mammals
|
||||||||||||||||||||||||||||||||||||||
| ATC Code | |||||||||||||||||||||||||||||||||||||||
| Drug Type |
Small molecular drug
|
||||||||||||||||||||||||||||||||||||||
| Structure |
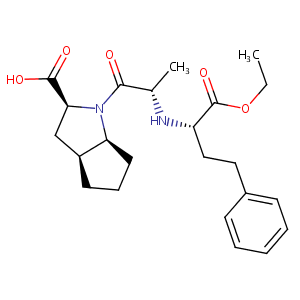 |
||||||||||||||||||||||||||||||||||||||
| 3D MOL | 2D MOL | ||||||||||||||||||||||||||||||||||||||
| #Ro5 Violations (Lipinski): 0 | Molecular Weight (mw) | 416.5 | |||||||||||||||||||||||||||||||||||||
| Logarithm of the Partition Coefficient (xlogp) | 1.4 | ||||||||||||||||||||||||||||||||||||||
| Rotatable Bond Count (rotbonds) | 10 | ||||||||||||||||||||||||||||||||||||||
| Hydrogen Bond Donor Count (hbonddonor) | 2 | ||||||||||||||||||||||||||||||||||||||
| Hydrogen Bond Acceptor Count (hbondacc) | 6 | ||||||||||||||||||||||||||||||||||||||
| ADMET Property |
|
||||||||||||||||||||||||||||||||||||||
| Adverse Drug Reaction (ADR) |
|
||||||||||||||||||||||||||||||||||||||
| Chemical Identifiers |
|
||||||||||||||||||||||||||||||||||||||
| Cross-matching ID | |||||||||||||||||||||||||||||||||||||||
| Combinatorial Drugs (CBD) | Click to Jump to the Detailed CBD Information of This Drug | ||||||||||||||||||||||||||||||||||||||
| Repurposed Drugs (RPD) | Click to Jump to the Detailed RPD Information of This Drug | ||||||||||||||||||||||||||||||||||||||
Molecular Interaction Atlas of This Drug
 Drug Therapeutic Target (DTT) |
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
 Drug Transporter (DTP) |
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
 Drug Off-Target (DOT) |
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Molecular Interaction Atlas (MIA) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Molecular Expression Atlas of This Drug
| The Studied Disease | Acute heart failure | |||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ICD Disease Classification | BD10-BD13 | |||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||
| Molecular Expression Atlas (MEA) | ||||||||||||||||||||||||||||||||||||
Drug-Drug Interaction (DDI) Information of This Drug
|
Coadministration of a Drug Treating the Same Disease as Ramipril
Coadministration of a Drug Treating the Disease Different from Ramipril (Comorbidity)
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Drug Inactive Ingredient(s) (DIG) and Formulation(s) of This Drug
References
| 1 | FDA Approved Drug Products from FDA Official Website. 2019. Application Number: (ANDA) 202392. | ||||
|---|---|---|---|---|---|
| 2 | Ramipril FDA Label | ||||
| 3 | URL: http://www.guidetopharmacology.org Nucleic Acids Res. 2015 Oct 12. pii: gkv1037. The IUPHAR/BPS Guide to PHARMACOLOGY in 2016: towards curated quantitative interactions between 1300 protein targets and 6000 ligands. (Ligand id: 6339). | ||||
| 4 | ClinicalTrials.gov (NCT04366050) Ramipril for the Treatment of COVID-19. U.S. National Institutes of Health. | ||||
| 5 | BDDCS applied to over 900 drugs | ||||
| 6 | Estimating the safe starting dose in phase I clinical trials and no observed effect level based on QSAR modeling of the human maximum recommended daily dose | ||||
| 7 | ADReCS-Target: target profiles for aiding drug safety research and application. Nucleic Acids Res. 2018 Jan 4;46(D1):D911-D917. doi: 10.1093/nar/gkx899. | ||||
| 8 | Angiotensin-converting enzyme gene polymorphism predicts the time-course of blood pressure response to angiotensin converting enzyme inhibition in the AASK trial. J Hypertens. 2007 Oct;25(10):2082-92. doi: 10.1097/HJH.0b013e3282b9720e. | ||||
| 9 | Knockouts model the 100 best-selling drugs--will they model the next 100 Nat Rev Drug Discov. 2003 Jan;2(1):38-51. | ||||
| 10 | Outcomes in Patients with COVID-19 Infection Taking ACEI/ARB. Curr Cardiol Rep. 2020 Apr 14;22(5):31. | ||||
| 11 | Transport of angiotensin-converting enzyme inhibitors by H+/peptide transporters revisited. J Pharmacol Exp Ther. 2008 Nov;327(2):432-41. | ||||
| 12 | Additive beneficial cardiovascular and metabolic effects of combination therapy with ramipril and candesartan in hypertensive patients. Eur Heart J. 2007 Jun;28(12):1440-7. doi: 10.1093/eurheartj/ehm101. Epub 2007 May 5. | ||||
| 13 | Distinct vascular and metabolic effects of different classes of anti-hypertensive drugs. Int J Cardiol. 2010 Apr 1;140(1):73-81. doi: 10.1016/j.ijcard.2008.11.017. Epub 2008 Dec 6. | ||||
| 14 | Contribution of human esterases to the metabolism of selected drugs of abuse. Toxicol Lett. 2015 Jan 5;232(1):159-66. doi: 10.1016/j.toxlet.2014.10.026. Epub 2014 Oct 24. | ||||
| 15 | Ramipril inhibits in vitro human mesangial cell proliferation and platelet-derived growth factor expression. Exp Nephrol. 1999 May-Jun;7(3):229-35. doi: 10.1159/000020606. | ||||
| 16 | Effects of an angiotensin converting enzyme inhibitor, ramipril, on intracranial circulation in healthy volunteers. off. Br J Clin Pharmacol. 1992 Sep;34(3):224-30. doi: 10.1111/j.1365-2125.1992.tb04128.x. | ||||
| 17 | Jarman PR, Mather HM "Diabetes may be independent risk factor for hyperkalaemia." BMJ 327 (2003): 812. [PMID: 14525902] | ||||
| 18 | Baba T, Tomiyama T, Takebe K "Enhancement by an ACE inhibitor of first-dose hypotension caused by an alpha-blocker." N Engl J Med 322 (1990): 1237. [PMID: 1970122] | ||||
| 19 | Product Information. Inspra (eplerenone). Searle, Chicago, IL. | ||||
| 20 | Burnakis TG, Mioduch HJ "Combined therapy with captopril and potassium supplementation: a potential for hyperkalemia." Arch Intern Med 144 (1984): 2371-2. [PMID: 6391404] | ||||
| 21 | Agencia Espaola de Medicamentos y Productos Sanitarios Healthcare "Centro de informacion online de medicamentos de la AEMPS - CIMA.". | ||||
| 22 | Murphy BF, Whitworth JA, Kincaid-Smith P "Renal insufficiency with combinations of angiotensin converting enzyme inhibitors and diuretics." Br Med J 288 (1984): 844-5. [PMID: 6322905] | ||||
| 23 | Cerner Multum, Inc. "Australian Product Information.". | ||||
| 24 | Mantyla R, Mannisto PT, Vuorela A, Sundberg S, Ottoila P "Impairment of captopril bioavailability by concomitant food and antacid intake." Int J Clin Pharmacol Ther Toxicol 22 (1984): 626-9. [PMID: 6389377] | ||||
| 25 | Abad S, Moachon L, Blanche P, Bavoux F, Sicard D, Salmon-Ceron D "Possible interaction between glicazide, fluconazole and sulfamethoxazole resulting in severe hypoglycaemia." Br J Clin Pharmacol 52 (2001): 456-7. [PMID: 11678792] | ||||
| 26 | Asplund K, Wiholm BE, Lithner F "Glibenclamide-associated hypoglycaemia: a report on 57 cases." Diabetologia 24 (1983): 412-7. [PMID: 6411511] | ||||
| 27 | Product Information. Aubagio (teriflunomide). Genzyme Corporation, Cambridge, MA. | ||||
| 28 | Product Information. Sirturo (bedaquiline). Janssen Pharmaceuticals, Titusville, NJ. | ||||
| 29 | Aronowitz JS, Chakos MH, Safferman AZ, Lieberman JA "Syncope associated with the combination of clozapine and enalapril." J Clin Psychopharmacol 14 (1994): 429-30. [PMID: 7884028] | ||||
| 30 | Ban TA "Drug interactions with psychoactive drugs." Dis Nerv Syst 36 (1975): 164-6. [PMID: 1116424] | ||||
| 31 | Product Information. Turalio (pexidartinib). Daiichi Sankyo, Inc., Parsippany, NJ. | ||||
| 32 | Warrington SJ, Ankier SI, Turner P "Evaluation of possible interactions between ethanol and trazodone or amitriptyline." Neuropsychobiology 15 (1986): 31-7. [PMID: 3725002] | ||||
| 33 | Product Information. Yasmin (drospirenone-ethinyl estradiol) Berlex Laboratories, Richmond, CA. | ||||
| 34 | Product Information. Diovan (valsartan). Novartis Pharmaceuticals, East Hanover, NJ. | ||||
| 35 | Katz RJ, Levy WS, Buff L, Wasserman AG "Prevention of nitrate tolerance with angiotension converting enzyme inhibitors." Circulation 83 (1991): 1271-7. [PMID: 1901528] | ||||
| 36 | Andrews C, Pinner G "Postural hypotension induced by paroxetine." BMJ 316 (1998): 595. [PMID: 9518913] | ||||
| 37 | Cerner Multum, Inc. "UK Summary of Product Characteristics.". | ||||
| 38 | Product Information. Adcetris (brentuximab vedotin). Seattle Genetics Inc, Bothell, WA. | ||||
| 39 | Elsharkawy AM, Schwab U, McCarron B, et al. "Efavirenz induced acute liver failure requiring liver transplantation in a slow drug metaboliser." J Clin Virol 58 (2013): 331-3. [PMID: 23763943] | ||||
| 40 | Product Information. Kynamro (mipomersen). Genzyme Corporation, Cambridge, MA. | ||||
| 41 | Canadian Pharmacists Association. | ||||
| 42 | Product Information. Juxtapid (lomitapide). Aegerion Pharmaceuticals Inc, Cambridge, MA. | ||||
| 43 | Health Canada "Potential risks of cardiovascular and renal adverse events in patients with type 2 diabetes treated with aliskiren (RASILEZ) or aliskiren/hydrochlorothiazide (RASILEZ HCT)." . | ||||
| 44 | DeQuattro V "Comparison of benazepril and other antihypertensive agents alone and in combination with the diuretic hydrochlorothiazide." Clin Cardiol 14 (1991): iv28-32. [PMID: 1893639] | ||||
| 45 | Product Information. Micardis (telmisartan). Boehringer-Ingelheim, Ridgefield, CT. | ||||
| 46 | Product Information. Samsca (tolvaptan). Otsuka American Pharmaceuticals Inc, Rockville, MD. | ||||
| 47 | Blakely KM, Drucker AM, Rosen CF "Drug-induced photosensitivity-an update: Culprit drugs, prevention and management." Drug Saf 42 (2019): 827-47. [PMID: 30888626] | ||||
| 48 | Al-Nawakil C, Willems L, Mauprivez C, et.al "Successful treatment of l-asparaginase-induced severe acute hepatotoxicity using mitochondrial cofactors." Leuk Lymphoma 55 (2014): 1670-4. [PMID: 24090500] | ||||
| 49 | Product Information. Zydelig (idelalisib). Gilead Sciences, Foster City, CA. | ||||
| 50 | Product Information. Clolar (clofarabine). sanofi-aventis, Bridgewater, NJ. | ||||
| 51 | Product Information. ReVia (naltrexone). DuPont Pharmaceuticals, Wilmington, DE. | ||||
| 52 | Perazella MA "Drug-induced hyperkalemia: old culprits and new offenders." Am J Med 109 (2000): 307-14. [PMID: 10996582] | ||||
| 53 | Canaday DH, Johnson JR "Hyperkalemia in elderly patients receiving standard doses of trimethoprim-sulfamethoxazole." Ann Intern Med 120 (1994): 438. [PMID: 8304666] | ||||
| 54 | Product Information. Clozaril (clozapine). Novartis Pharmaceuticals, East Hanover, NJ. | ||||
