Details of the Drug
General Information of Drug (ID: DM9PLRH)
| Drug Name |
Doxazosin
|
||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Synonyms |
Doxazosina; Doxazosine; Doxazosinum; Normothen; Doxazosina [Spanish]; Doxazosine [French]; Doxazosinum [Latin]; UK 33274; Cardura (TN); Cardura XL (TN); Cardura-1; Cardura-2; Cardura-4; Carduran (TN); Doxazosin (INN); Doxazosin [INN:BAN]; UK-33274; [4-(4-amino-6,7-dimethoxyquinazolin-2-yl)piperazin-1-yl]-(2,3-dihydro-1,4-benzodioxin-3-yl)methanone; 1-(4-Amino-6,7-Dimethoxy-2-quinazolinyl)-4-(1,4-benzodioxan-2-ylcarbonyl)piperazin; 1-(4-Amino-6,7-dimethoxy-2-chinazolinyl)-4-(2,3-dihydro-1,4-benzodioxixin-2-ylcarbonyl)piperazin; 1-(4-Amino-6,7-dimethoxy-2-quinazolinyl)-4-[4-(1,4-benzodioxan-2-yl)carpiperazin-1-yl)]-6,7-dimethoxyquinazoline mesylate; 2-[4-(2,3-dihydro-1,4-benzodioxin-2-ylcarbonyl)piperazin-1-yl]-6,7-bis(methyloxy)quinazolin-4-amine; 2-[4-(2,3-dihydro-1,4-benzodioxin-2-ylcarbonyl)piperazin-1-yl]-6,7-dimethoxyquinazolin-4-amine; 2-{4-[(2,3-dihydro-1,4-benzodioxin-2-yl)carbonyl]piperazin-1-yl}-6,7-dimethoxyquinazolin-4-amine
|
||||||||||||||||||||||||||||||
| Indication |
|
||||||||||||||||||||||||||||||
| Therapeutic Class |
Antihypertensive Agents
|
||||||||||||||||||||||||||||||
| Affected Organisms |
Humans and other mammals
|
||||||||||||||||||||||||||||||
| ATC Code | |||||||||||||||||||||||||||||||
| Drug Type |
Small molecular drug
|
||||||||||||||||||||||||||||||
| Structure |
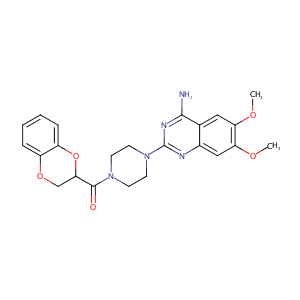 |
||||||||||||||||||||||||||||||
| 3D MOL | 2D MOL | ||||||||||||||||||||||||||||||
| #Ro5 Violations (Lipinski): 0 | Molecular Weight (mw) | 451.5 | |||||||||||||||||||||||||||||
| Logarithm of the Partition Coefficient (xlogp) | 2.5 | ||||||||||||||||||||||||||||||
| Rotatable Bond Count (rotbonds) | 4 | ||||||||||||||||||||||||||||||
| Hydrogen Bond Donor Count (hbonddonor) | 1 | ||||||||||||||||||||||||||||||
| Hydrogen Bond Acceptor Count (hbondacc) | 9 | ||||||||||||||||||||||||||||||
| ADMET Property |
|
||||||||||||||||||||||||||||||
| Chemical Identifiers |
|
||||||||||||||||||||||||||||||
| Cross-matching ID | |||||||||||||||||||||||||||||||
| Combinatorial Drugs (CBD) | Click to Jump to the Detailed CBD Information of This Drug | ||||||||||||||||||||||||||||||
| Repurposed Drugs (RPD) | Click to Jump to the Detailed RPD Information of This Drug | ||||||||||||||||||||||||||||||
Molecular Interaction Atlas of This Drug
 Drug Therapeutic Target (DTT) |
|
|||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
 Drug-Metabolizing Enzyme (DME) |
|
|||||||||||||||||||||||||||||||||||||||||
| Molecular Interaction Atlas (MIA) | ||||||||||||||||||||||||||||||||||||||||||
Drug-Drug Interaction (DDI) Information of This Drug
|
Coadministration of a Drug Treating the Same Disease as Doxazosin
Coadministration of a Drug Treating the Disease Different from Doxazosin (Comorbidity)
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Drug Inactive Ingredient(s) (DIG) and Formulation(s) of This Drug
References
| 1 | Doxazosin FDA Label | ||||
|---|---|---|---|---|---|
| 2 | URL: http://www.guidetopharmacology.org Nucleic Acids Res. 2015 Oct 12. pii: gkv1037. The IUPHAR/BPS Guide to PHARMACOLOGY in 2016: towards curated quantitative interactions between 1300 protein targets and 6000 ligands. (Ligand id: 7170). | ||||
| 3 | BDDCS applied to over 900 drugs | ||||
| 4 | Kaye B, Cussans NJ, Faulkner JK, Stopher DA, Reid JL: The metabolism and kinetics of doxazosin in man, mouse, rat and dog. Br J Clin Pharmacol. 1986;21 Suppl 1:19S-25S. doi: 10.1111/j.1365-2125.1986.tb02849.x. | ||||
| 5 | Doxazosin FDA label | ||||
| 6 | Estimating the safe starting dose in phase I clinical trials and no observed effect level based on QSAR modeling of the human maximum recommended daily dose | ||||
| 7 | Trend Analysis of a Database of Intravenous Pharmacokinetic Parameters in Humans for 1352 Drug Compounds | ||||
| 8 | The role of combination medical therapy in benign prostatic hyperplasia. Int J Impot Res. 2008 Dec;20 Suppl 3:S33-43. | ||||
| 9 | Priapism induced by boceprevir-CYP3A4 inhibition and alpha-adrenergic blockade: case report. Clin Infect Dis. 2014 Jan;58(1):e35-8. | ||||
| 10 | Product monograph: CARDURA (Doxazosin mesylate). | ||||
| 11 | Chrysant SG "Experience with terazosin administered in combination with other antihypertensive agents." Am J Med 80 (1986): 55-61. [PMID: 2872808] | ||||
| 12 | Baba T, Tomiyama T, Takebe K "Enhancement by an ACE inhibitor of first-dose hypotension caused by an alpha-blocker." N Engl J Med 322 (1990): 1237. [PMID: 1970122] | ||||
| 13 | Canadian Pharmacists Association. | ||||
| 14 | Aronowitz JS, Chakos MH, Safferman AZ, Lieberman JA "Syncope associated with the combination of clozapine and enalapril." J Clin Psychopharmacol 14 (1994): 429-30. [PMID: 7884028] | ||||
| 15 | Warrington SJ, Ankier SI, Turner P "Evaluation of possible interactions between ethanol and trazodone or amitriptyline." Neuropsychobiology 15 (1986): 31-7. [PMID: 3725002] | ||||
| 16 | Ban TA "Drug interactions with psychoactive drugs." Dis Nerv Syst 36 (1975): 164-6. [PMID: 1116424] | ||||
| 17 | EMEA. European Medicines Agency "EPARs. European Union Public Assessment Reports.". | ||||
| 18 | Cerner Multum, Inc. "Australian Product Information.". | ||||
| 19 | Product Information. Orladeyo (berotralstat). BioCryst Pharmaceuticals Inc, Durham, NC. | ||||
| 20 | Product Information. Copiktra (duvelisib). Verastem, Inc., Needham, MA. | ||||
| 21 | Product Information. Zulresso (brexanolone). Sage Therapeutics, Inc., Cambridge, MA. | ||||
| 22 | Product Information. Reyvow (lasmiditan). Lilly, Eli and Company, Indianapolis, IN. | ||||
| 23 | Product Information. Xeglyze (abametapir topical). Dr. Reddy's Laboratories Inc, Upper Saddle River, NJ. | ||||
| 24 | Product Information. Xenleta (lefamulin). Nabriva Therapeutics US, Inc., King of Prussia, PA. | ||||
| 25 | Product Information. Flomax (tamsulosin). Boehringer-Ingelheim, Ridgefield, CT. | ||||
| 26 | Product Information. Clozaril (clozapine). Novartis Pharmaceuticals, East Hanover, NJ. | ||||
