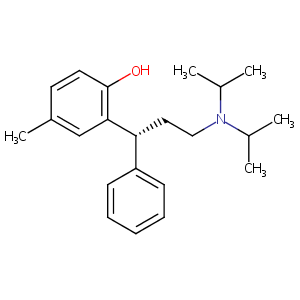
| DDI Drug Name |
DDI Drug ID |
Severity |
Mechanism |
Comorbidity |
REF |
| Arn-509 |
DMT81LZ
|
Moderate |
Increased metabolism of Tolterodine caused by Arn-509 mediated induction of CYP450 enzyme. |
Acute myeloid leukaemia [2A60]
|
[10] |
| Oliceridine |
DM6MDCF
|
Moderate |
Additive CNS depression effects by the combination of Tolterodine and Oliceridine. |
Acute pain [MG31]
|
[11] |
| Mepyramine |
DMB4SFH
|
Moderate |
Additive anticholinergic effects by the combination of Tolterodine and Mepyramine. |
Allergic/hypersensitivity disorder [4A80-4A8Z]
|
[12] |
| Phenyltoloxamine |
DMKAEQW
|
Moderate |
Additive anticholinergic effects by the combination of Tolterodine and Phenyltoloxamine. |
Allergic/hypersensitivity disorder [4A80-4A8Z]
|
[12] |
| Memantine |
DMD9WSC
|
Moderate |
Additive anticholinergic effects by the combination of Tolterodine and Memantine. |
Alzheimer disease [8A20]
|
[9] |
| Dronedarone |
DMA8FS5
|
Moderate |
Decreased metabolism of Tolterodine caused by Dronedarone mediated inhibition of CYP450 enzyme. |
Angina pectoris [BA40]
|
[13] |
| Nifedipine |
DMSVOZT
|
Moderate |
Decreased metabolism of Tolterodine caused by Nifedipine mediated inhibition of CYP450 enzyme. |
Angina pectoris [BA40]
|
[13] |
| Posaconazole |
DMUL5EW
|
Moderate |
Decreased metabolism of Tolterodine caused by Posaconazole mediated inhibition of CYP450 enzyme. |
Aspergillosis [1F20]
|
[13] |
| Desipramine |
DMT2FDC
|
Moderate |
Additive anticholinergic effects by the combination of Tolterodine and Desipramine. |
Attention deficit hyperactivity disorder [6A05]
|
[9] |
| Dalfopristin |
DM4LTKV
|
Moderate |
Decreased metabolism of Tolterodine caused by Dalfopristin mediated inhibition of CYP450 enzyme. |
Bacterial infection [1A00-1C4Z]
|
[13] |
| Clarithromycin |
DM4M1SG
|
Moderate |
Decreased metabolism of Tolterodine caused by Clarithromycin mediated inhibition of CYP450 enzyme. |
Bacterial infection [1A00-1C4Z]
|
[13] |
| Troleandomycin |
DMUZNIG
|
Moderate |
Decreased metabolism of Tolterodine caused by Troleandomycin mediated inhibition of CYP450 enzyme. |
Bacterial infection [1A00-1C4Z]
|
[13] |
| Cariprazine |
DMJYDVK
|
Moderate |
Additive anticholinergic effects by the combination of Tolterodine and Cariprazine. |
Bipolar disorder [6A60]
|
[9] |
| Lapatinib |
DM3BH1Y
|
Moderate |
Decreased metabolism of Tolterodine caused by Lapatinib mediated inhibition of CYP450 enzyme. |
Breast cancer [2C60-2C6Y]
|
[13] |
| Tucatinib |
DMBESUA
|
Moderate |
Decreased metabolism of Tolterodine caused by Tucatinib mediated inhibition of CYP450 enzyme. |
Breast cancer [2C60-2C6Y]
|
[13] |
| Acetylcholine |
DMDF79Z
|
Moderate |
Antagonize the effect of Tolterodine when combined with Acetylcholine. |
Cataract [9B10]
|
[14] |
| Umeclidinium |
DM4E8O9
|
Moderate |
Additive anticholinergic effects by the combination of Tolterodine and Umeclidinium. |
Chronic obstructive pulmonary disease [CA22]
|
[15] |
| Tiotropium |
DMFDC0Q
|
Moderate |
Additive anticholinergic effects by the combination of Tolterodine and Tiotropium. |
Chronic obstructive pulmonary disease [CA22]
|
[15] |
| Revefenacin |
DMMP5SI
|
Moderate |
Additive anticholinergic effects by the combination of Tolterodine and Revefenacin. |
Chronic obstructive pulmonary disease [CA22]
|
[15] |
| Dihydrocodeine |
DMB0FWL
|
Moderate |
Additive CNS depression effects by the combination of Tolterodine and Dihydrocodeine. |
Chronic pain [MG30]
|
[16] |
| Arbutamine |
DMCY8AF
|
Moderate |
Increased risk of rapid heart rate by the combination of Tolterodine and Arbutamine. |
Coronary atherosclerosis [BA80]
|
[17] |
| Osilodrostat |
DMIJC9X
|
Moderate |
Decreased metabolism of Tolterodine caused by Osilodrostat mediated inhibition of CYP450 enzyme. |
Cushing syndrome [5A70]
|
[13] |
| Ivacaftor |
DMZC1HS
|
Moderate |
Decreased metabolism of Tolterodine caused by Ivacaftor mediated inhibition of CYP450 enzyme. |
Cystic fibrosis [CA25]
|
[13] |
| MK-8228 |
DMOB58Q
|
Moderate |
Decreased metabolism of Tolterodine caused by MK-8228 mediated inhibition of CYP450 enzyme. |
Cytomegaloviral disease [1D82]
|
[13] |
| Sertraline |
DM0FB1J
|
Minor |
Decreased metabolism of Tolterodine caused by Sertraline mediated inhibition of CYP450 enzyme. |
Depression [6A70-6A7Z]
|
[18] |
| Nefazodone |
DM4ZS8M
|
Moderate |
Decreased metabolism of Tolterodine caused by Nefazodone mediated inhibition of CYP450 enzyme. |
Depression [6A70-6A7Z]
|
[13] |
| Escitalopram |
DMFK9HG
|
Minor |
Decreased metabolism of Tolterodine caused by Escitalopram mediated inhibition of CYP450 enzyme. |
Depression [6A70-6A7Z]
|
[18] |
| OPC-34712 |
DMHG57U
|
Moderate |
Additive anticholinergic effects by the combination of Tolterodine and OPC-34712. |
Depression [6A70-6A7Z]
|
[9] |
| Clomipramine |
DMINRKW
|
Moderate |
Additive anticholinergic effects by the combination of Tolterodine and Clomipramine. |
Depression [6A70-6A7Z]
|
[9] |
| Doxepin |
DMPI98T
|
Moderate |
Additive anticholinergic effects by the combination of Tolterodine and Doxepin. |
Depression [6A70-6A7Z]
|
[9] |
| Mepenzolate |
DM8YU2F
|
Moderate |
Additive anticholinergic effects by the combination of Tolterodine and Mepenzolate. |
Digestive system disease [DE2Z]
|
[9] |
| Oxybutynine |
DMJPBAX
|
Moderate |
Additive anticholinergic effects by the combination of Tolterodine and Oxybutynine. |
Discovery agent [N.A.]
|
[9] |
| Cenobamate |
DM8KLU9
|
Moderate |
Increased metabolism of Tolterodine caused by Cenobamate mediated induction of CYP450 enzyme. |
Epilepsy/seizure [8A61-8A6Z]
|
[19] |
| Stiripentol |
DMMSDOY
|
Moderate |
Decreased metabolism of Tolterodine caused by Stiripentol mediated inhibition of CYP450 enzyme. |
Epilepsy/seizure [8A61-8A6Z]
|
[13] |
| Rufinamide |
DMWE60C
|
Moderate |
Increased metabolism of Tolterodine caused by Rufinamide mediated induction of CYP450 enzyme. |
Epilepsy/seizure [8A61-8A6Z]
|
[10] |
| Diphenhydramine |
DMKQTBA
|
Moderate |
Additive anticholinergic effects by the combination of Tolterodine and Diphenhydramine. |
Episodic vestibular syndrome [AB31]
|
[9] |
| Nadolol |
DMW6GVL
|
Moderate |
Additive CNS depression effects by the combination of Tolterodine and Nadolol. |
Essential hypertension [BA00]
|
[10] |
| Itraconazole |
DMCR1MV
|
Moderate |
Decreased metabolism of Tolterodine caused by Itraconazole mediated inhibition of CYP450 enzyme. |
Fungal infection [1F29-1F2F]
|
[13] |
| Miconazole |
DMPMYE8
|
Moderate |
Decreased metabolism of Tolterodine caused by Miconazole mediated inhibition of CYP450 enzyme. |
Fungal infection [1F29-1F2F]
|
[13] |
| Pilocarpine |
DMV9ADG
|
Moderate |
Antagonize the effect of Tolterodine when combined with Pilocarpine. |
Glaucoma [9C61]
|
[20] |
| Carvedilol |
DMHTEAO
|
Moderate |
Antagonize the effect of Tolterodine when combined with Carvedilol. |
Heart failure [BD10-BD1Z]
|
[10] |
| Boceprevir |
DMBSHMF
|
Moderate |
Decreased metabolism of Tolterodine caused by Boceprevir mediated inhibition of CYP450 enzyme. |
Hepatitis virus infection [1E50-1E51]
|
[13] |
| Telaprevir |
DMMRV29
|
Moderate |
Decreased metabolism of Tolterodine caused by Telaprevir mediated inhibition of CYP450 enzyme. |
Hepatitis virus infection [1E50-1E51]
|
[13] |
| Rifapentine |
DMCHV4I
|
Moderate |
Increased metabolism of Tolterodine caused by Rifapentine mediated induction of CYP450 enzyme. |
HIV-infected patients with tuberculosis [1B10-1B14]
|
[21] |
| Fosamprenavir |
DM4W9B3
|
Moderate |
Decreased metabolism of Tolterodine caused by Fosamprenavir mediated inhibition of CYP450 enzyme. |
Human immunodeficiency virus disease [1C60-1C62]
|
[13] |
| Cobicistat |
DM6L4H2
|
Moderate |
Decreased metabolism of Tolterodine caused by Cobicistat mediated inhibition of CYP450 enzyme. |
Human immunodeficiency virus disease [1C60-1C62]
|
[13] |
| Saquinavir |
DMG814N
|
Moderate |
Decreased metabolism of Tolterodine caused by Saquinavir mediated inhibition of CYP450 enzyme. |
Human immunodeficiency virus disease [1C60-1C62]
|
[13] |
| Etravirine |
DMGV8QU
|
Moderate |
Increased metabolism of Tolterodine caused by Etravirine mediated induction of CYP450 enzyme. |
Human immunodeficiency virus disease [1C60-1C62]
|
[22] |
| Darunavir |
DMN3GCH
|
Moderate |
Decreased metabolism of Tolterodine caused by Darunavir mediated inhibition of CYP450 enzyme. |
Human immunodeficiency virus disease [1C60-1C62]
|
[13] |
| Atazanavir |
DMSYRBX
|
Moderate |
Decreased metabolism of Tolterodine caused by Atazanavir mediated inhibition of CYP450 enzyme. |
Human immunodeficiency virus disease [1C60-1C62]
|
[13] |
| Acebutolol |
DM0TI4U
|
Moderate |
Antagonize the effect of Tolterodine when combined with Acebutolol. |
Hypertension [BA00-BA04]
|
[10] |
| Penbutolol |
DM4ES8F
|
Moderate |
Additive CNS depression effects by the combination of Tolterodine and Penbutolol. |
Hypertension [BA00-BA04]
|
[10] |
| Nebivolol |
DM7F1PA
|
Moderate |
Antagonize the effect of Tolterodine when combined with Nebivolol. |
Hypertension [BA00-BA04]
|
[10] |
| Givosiran |
DM5PFIJ
|
Minor |
Decreased metabolism of Tolterodine caused by Givosiran mediated inhibition of CYP450 enzyme. |
Inborn porphyrin/heme metabolism error [5C58]
|
[18] |
| Belladonna |
DM2RBWK
|
Moderate |
Additive anticholinergic effects by the combination of Tolterodine and Belladonna. |
Infectious gastroenteritis/colitis [1A40]
|
[9] |
| Berotralstat |
DMWA2DZ
|
Moderate |
Decreased metabolism of Tolterodine caused by Berotralstat mediated inhibition of CYP450 enzyme. |
Innate/adaptive immunodeficiency [4A00]
|
[23] |
| ITI-007 |
DMUQ1DO
|
Moderate |
Additive anticholinergic effects by the combination of Tolterodine and ITI-007. |
Insomnia [7A00-7A0Z]
|
[9] |
| R0-93877 |
DMM4U9G
|
Moderate |
Antagonize the effect of Tolterodine when combined with R0-93877. |
Irritable bowel syndrome [DD91]
|
[24] |
| Glycerol phenylbutyrate |
DMDGRQO
|
Minor |
Decreased metabolism of Tolterodine caused by Glycerol phenylbutyrate mediated inhibition of CYP450 enzyme. |
Liver disease [DB90-DB9Z]
|
[18] |
| Crizotinib |
DM4F29C
|
Moderate |
Decreased metabolism of Tolterodine caused by Crizotinib mediated inhibition of CYP450 enzyme. |
Lung cancer [2C25]
|
[13] |
| Brigatinib |
DM7W94S
|
Moderate |
Increased metabolism of Tolterodine caused by Brigatinib mediated induction of CYP450 enzyme. |
Lung cancer [2C25]
|
[25] |
| Ceritinib |
DMB920Z
|
Moderate |
Decreased metabolism of Tolterodine caused by Ceritinib mediated inhibition of CYP450 enzyme. |
Lung cancer [2C25]
|
[13] |
| PF-06463922 |
DMKM7EW
|
Moderate |
Increased metabolism of Tolterodine caused by PF-06463922 mediated induction of CYP450 enzyme. |
Lung cancer [2C25]
|
[26] |
| Dacomitinib |
DMOH8VY
|
Minor |
Decreased metabolism of Tolterodine caused by Dacomitinib mediated inhibition of CYP450 enzyme. |
Lung cancer [2C25]
|
[18] |
| Selpercatinib |
DMZR15V
|
Moderate |
Decreased metabolism of Tolterodine caused by Selpercatinib mediated inhibition of CYP450 enzyme. |
Lung cancer [2C25]
|
[13] |
| Lumefantrine |
DM29GAD
|
Minor |
Decreased metabolism of Tolterodine caused by Lumefantrine mediated inhibition of CYP450 enzyme. |
Malaria [1F40-1F45]
|
[18] |
| Halofantrine |
DMOMK1V
|
Minor |
Decreased metabolism of Tolterodine caused by Halofantrine mediated inhibition of CYP450 enzyme. |
Malaria [1F40-1F45]
|
[18] |
| Hydroxychloroquine |
DMSIVND
|
Minor |
Decreased metabolism of Tolterodine caused by Hydroxychloroquine mediated inhibition of CYP450 enzyme. |
Malaria [1F40-1F45]
|
[18] |
| Primaquine |
DMWQ16I
|
Minor |
Decreased metabolism of Tolterodine caused by Primaquine mediated inhibition of CYP450 enzyme. |
Malaria [1F40-1F45]
|
[18] |
| Idelalisib |
DM602WT
|
Moderate |
Decreased metabolism of Tolterodine caused by Idelalisib mediated inhibition of CYP450 enzyme. |
Mature B-cell leukaemia [2A82]
|
[13] |
| IPI-145 |
DMWA24P
|
Moderate |
Decreased metabolism of Tolterodine caused by IPI-145 mediated inhibition of CYP450 enzyme. |
Mature B-cell leukaemia [2A82]
|
[13] |
| Vemurafenib |
DM62UG5
|
Minor |
Decreased metabolism of Tolterodine caused by Vemurafenib mediated inhibition of CYP450 enzyme. |
Melanoma [2C30]
|
[18] |
| Dabrafenib |
DMX6OE3
|
Moderate |
Increased metabolism of Tolterodine caused by Dabrafenib mediated induction of CYP450 enzyme. |
Melanoma [2C30]
|
[10] |
| Danazol |
DML8KTN
|
Moderate |
Decreased metabolism of Tolterodine caused by Danazol mediated inhibition of CYP450 enzyme. |
Menstrual cycle bleeding disorder [GA20]
|
[13] |
| Exjade |
DMHPRWG
|
Moderate |
Decreased metabolism of Tolterodine caused by Exjade mediated inhibition of CYP450 enzyme. |
Mineral absorption/transport disorder [5C64]
|
[27] |
| Panobinostat |
DM58WKG
|
Minor |
Decreased metabolism of Tolterodine caused by Panobinostat mediated inhibition of CYP450 enzyme. |
Multiple myeloma [2A83]
|
[18] |
| Neostigmine |
DM6T2J3
|
Moderate |
Antagonize the effect of Tolterodine when combined with Neostigmine. |
Myasthenia gravis [8C6Y]
|
[28] |
| Ambenonium |
DMOP0BL
|
Moderate |
Antagonize the effect of Tolterodine when combined with Ambenonium. |
Myasthenia gravis [8C6Y]
|
[28] |
| Nilotinib |
DM7HXWT
|
Moderate |
Decreased metabolism of Tolterodine caused by Nilotinib mediated inhibition of CYP450 enzyme. |
Myeloproliferative neoplasm [2A20]
|
[29] |
| Phenindamine |
DMDTC7R
|
Moderate |
Additive anticholinergic effects by the combination of Tolterodine and Phenindamine. |
Nasopharyngitis [CA00]
|
[9] |
| Promethazine |
DM6I5GR
|
Moderate |
Additive anticholinergic effects by the combination of Tolterodine and Promethazine. |
Nausea/vomiting [MD90]
|
[9] |
| Rolapitant |
DM8XP26
|
Minor |
Decreased metabolism of Tolterodine caused by Rolapitant mediated inhibition of CYP450 enzyme. |
Nausea/vomiting [MD90]
|
[18] |
| Cyclizine |
DM9G7BS
|
Moderate |
Additive anticholinergic effects by the combination of Tolterodine and Cyclizine. |
Nausea/vomiting [MD90]
|
[9] |
| Netupitant |
DMEKAYI
|
Moderate |
Decreased metabolism of Tolterodine caused by Netupitant mediated inhibition of CYP450 enzyme. |
Nausea/vomiting [MD90]
|
[13] |
| Metoclopramide |
DMFA5MY
|
Moderate |
Antagonize the effect of Tolterodine when combined with Metoclopramide. |
Nausea/vomiting [MD90]
|
[30] |
| Bupropion |
DM5PCS7
|
Minor |
Decreased metabolism of Tolterodine caused by Bupropion mediated inhibition of CYP450 enzyme. |
Nicotine use disorder [6C4A]
|
[18] |
| Entrectinib |
DMMPTLH
|
Moderate |
Decreased metabolism of Tolterodine caused by Entrectinib mediated inhibition of CYP450 enzyme. |
Non-small cell lung cancer [2C25]
|
[13] |
| Lorcaserin |
DMG6OYJ
|
Minor |
Decreased metabolism of Tolterodine caused by Lorcaserin mediated inhibition of CYP450 enzyme. |
Obesity [5B80-5B81]
|
[18] |
| Dexfenfluramine |
DMJ7YDS
|
Minor |
Decreased metabolism of Tolterodine caused by Dexfenfluramine mediated inhibition of CYP450 enzyme. |
Obesity [5B80-5B81]
|
[18] |
| Polythiazide |
DMCH80F
|
Minor |
Altered absorption of Tolterodine due to GI dynamics variation caused by Polythiazide. |
Oedema [MG29]
|
[31] |
| Levomethadyl Acetate |
DM06HG5
|
Moderate |
Additive CNS depression effects by the combination of Tolterodine and Levomethadyl Acetate. |
Opioid use disorder [6C43]
|
[16] |
| Rucaparib |
DM9PVX8
|
Moderate |
Decreased metabolism of Tolterodine caused by Rucaparib mediated inhibition of CYP450 enzyme. |
Ovarian cancer [2C73]
|
[13] |
| Oxymorphone |
DM65AGJ
|
Moderate |
Additive CNS depression effects by the combination of Tolterodine and Oxymorphone. |
Pain [MG30-MG3Z]
|
[11] |
| Dezocine |
DMJDB0Y
|
Moderate |
Additive CNS depression effects by the combination of Tolterodine and Dezocine. |
Pain [MG30-MG3Z]
|
[11] |
| Flavoxate |
DMKV4NL
|
Moderate |
Additive anticholinergic effects by the combination of Tolterodine and Flavoxate. |
Pain [MG30-MG3Z]
|
[9] |
| Levodopa |
DMN3E57
|
Moderate |
Altered absorption of Tolterodine due to GI dynamics variation caused by Levodopa. |
Parkinsonism [8A00]
|
[32] |
| Orphenadrine |
DMW542E
|
Moderate |
Additive anticholinergic effects by the combination of Tolterodine and Orphenadrine. |
Parkinsonism [8A00]
|
[9] |
| Abametapir |
DM2RX0I
|
Moderate |
Decreased metabolism of Tolterodine caused by Abametapir mediated inhibition of CYP450 enzyme. |
Pediculosis [1G00]
|
[33] |
| Methylscopolamine |
DM5VWOB
|
Moderate |
Additive anticholinergic effects by the combination of Tolterodine and Methylscopolamine. |
Peptic ulcer [DA61]
|
[9] |
| Lefamulin |
DME6G97
|
Moderate |
Decreased metabolism of Tolterodine caused by Lefamulin mediated inhibition of CYP450 enzyme. |
Pneumonia [CA40]
|
[34] |
| Lonafarnib |
DMGM2Z6
|
Moderate |
Decreased metabolism of Tolterodine caused by Lonafarnib mediated inhibition of CYP450 enzyme. |
Premature ageing appearance [LD2B]
|
[13] |
| ABIRATERONE |
DM8V75C
|
Minor |
Decreased metabolism of Tolterodine caused by ABIRATERONE mediated inhibition of CYP450 enzyme. |
Prostate cancer [2C82]
|
[18] |
| Enzalutamide |
DMGL19D
|
Moderate |
Increased metabolism of Tolterodine caused by Enzalutamide mediated induction of CYP450 enzyme. |
Prostate cancer [2C82]
|
[35] |
| Bicalutamide |
DMZMSPF
|
Moderate |
Decreased metabolism of Tolterodine caused by Bicalutamide mediated inhibition of CYP450 enzyme. |
Prostate cancer [2C82]
|
[13] |
| Levomepromazine |
DMIKFEL
|
Moderate |
Additive anticholinergic effects by the combination of Tolterodine and Levomepromazine. |
Psychotic disorder [6A20-6A25]
|
[9] |
| Temsirolimus |
DMS104F
|
Moderate |
Increased plasma concentrations of Tolterodine and Temsirolimus due to competitive inhibition of the same metabolic pathway. |
Renal cell carcinoma [2C90]
|
[36] |
| Oxamniquine |
DM2QDX1
|
Minor |
Decreased metabolism of Tolterodine caused by Oxamniquine mediated inhibition of CYP450 enzyme. |
Schistosomiasis [1F86]
|
[18] |
| Quetiapine |
DM1N62C
|
Moderate |
Additive anticholinergic effects by the combination of Tolterodine and Quetiapine. |
Schizophrenia [6A20]
|
[9] |
| Aripiprazole |
DM3NUMH
|
Moderate |
Additive anticholinergic effects by the combination of Tolterodine and Aripiprazole. |
Schizophrenia [6A20]
|
[9] |
| Iloperidone |
DM6AUFY
|
Moderate |
Additive anticholinergic effects by the combination of Tolterodine and Iloperidone. |
Schizophrenia [6A20]
|
[9] |
| Paliperidone |
DM7NPJS
|
Moderate |
Additive anticholinergic effects by the combination of Tolterodine and Paliperidone. |
Schizophrenia [6A20]
|
[9] |
| Molindone |
DMAH70G
|
Moderate |
Additive anticholinergic effects by the combination of Tolterodine and Molindone. |
Schizophrenia [6A20]
|
[9] |
| Thiothixene |
DMDINC4
|
Moderate |
Additive anticholinergic effects by the combination of Tolterodine and Thiothixene. |
Schizophrenia [6A20]
|
[9] |
| Asenapine |
DMSQZE2
|
Moderate |
Additive anticholinergic effects by the combination of Tolterodine and Asenapine. |
Schizophrenia [6A20]
|
[9] |
| Pimozide |
DMW83TP
|
Moderate |
Additive anticholinergic effects by the combination of Tolterodine and Pimozide. |
Schizophrenia [6A20]
|
[9] |
| Voxelotor |
DMCS6M5
|
Moderate |
Decreased metabolism of Tolterodine caused by Voxelotor mediated inhibition of CYP450 enzyme. |
Sickle-cell disorder [3A51]
|
[13] |
| Telotristat ethyl |
DMDIYFZ
|
Moderate |
Increased metabolism of Tolterodine caused by Telotristat ethyl mediated induction of CYP450 enzyme. |
Small intestine developmental anomaly [DA90]
|
[10] |
| Larotrectinib |
DM26CQR
|
Moderate |
Decreased metabolism of Tolterodine caused by Larotrectinib mediated inhibition of CYP450 enzyme. |
Solid tumour/cancer [2A00-2F9Z]
|
[13] |
| Armodafinil |
DMGB035
|
Minor |
Increased metabolism of Tolterodine caused by Armodafinil mediated induction of CYP450 enzyme. |
Solid tumour/cancer [2A00-2F9Z]
|
[37] |
| LEE011 |
DMMX75K
|
Moderate |
Decreased metabolism of Tolterodine caused by LEE011 mediated inhibition of CYP450 enzyme. |
Solid tumour/cancer [2A00-2F9Z]
|
[13] |
| Pitolisant |
DM8RFNJ
|
Moderate |
Increased metabolism of Tolterodine caused by Pitolisant mediated induction of CYP450 enzyme. |
Somnolence [MG42]
|
[10] |
| Fostamatinib |
DM6AUHV
|
Moderate |
Decreased metabolism of Tolterodine caused by Fostamatinib mediated inhibition of CYP450 enzyme. |
Thrombocytopenia [3B64]
|
[38] |
| Chlorpheniramine |
DM5URA2
|
Moderate |
Additive anticholinergic effects by the combination of Tolterodine and Chlorpheniramine. |
Vasomotor/allergic rhinitis [CA08]
|
[9] |
| Trimeprazine |
DMEMV9D
|
Moderate |
Additive anticholinergic effects by the combination of Tolterodine and Trimeprazine. |
Vasomotor/allergic rhinitis [CA08]
|
[12] |
| Acrivastine |
DMTIGA0
|
Moderate |
Additive anticholinergic effects by the combination of Tolterodine and Acrivastine. |
Vasomotor/allergic rhinitis [CA08]
|
[9] |
| Propafenone |
DMPIBJK
|
Minor |
Decreased metabolism of Tolterodine caused by Propafenone mediated inhibition of CYP450 enzyme. |
Ventricular tachyarrhythmia [BC71]
|
[18] |
| Amiodarone |
DMUTEX3
|
Moderate |
Decreased metabolism of Tolterodine caused by Amiodarone mediated inhibition of CYP450 enzyme. |
Ventricular tachyarrhythmia [BC71]
|
[13] |
| ----------- |
|
|
|
|
|