Details of the Drug
General Information of Drug (ID: DMUTEX3)
| Drug Name |
Amiodarone
|
||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Synonyms |
Amidorone; Aminodarone; Amiobeta; Amiodarex; Amiodarona; Amiodarons; Amiodaronum; Amiohexal; Amjodaronum; Ancar; Aratac;Arycor; Braxan; Corbionax; Cordarex; Cordarone; Kordaron; Labaz; Ortacrone; Pacerone; Rytmarone; Sedacoron; Sedacorone; Tachydaron; ASTA Medica Brand of Amiodarone Hydrochloride; Alphapharm Brand of Amiodarone Hydrochloride; Amiodarone Base; Armstrong Brand of Amiodarone Hydrochloride; Berenguer Infale Brand of Amiodarone Hydrochloride; Betapharm Brand of Amiodarone Hydrochloride; Cordarone Intravenous; G Gam Brand of Amiodarone Hydrochloride; Hexal Brand of Amiodarone Hydrochloride; Leurquin Brand of Amiodarone Hydrochloride; Pharma Investi Brand of Amiodarone Hydrochloride; Sanofi Winthrop Brand of Amiodarone Hydrochloride; Wyeth Brand of Amiodarone Hydrochloride; Ethylaminoethoxy)benzoyl]benzofuran; L 3428; L3428; SKF 33134A; Amio-Aqueous IV; Amiodarona [INN-Spanish]; Amiodaronum [INN-Latin]; Aratac (TN); Arycor (TN); Atlansil (TN); Cordarone (TN); L-3428; Pacerone (TN); Pms-Amiodarone; SKF 33134-A; Amiodarone (USAN/INN); Amiodarone [USAN:BAN:INN]; Ketone, 2-butyl-3-benzofuranyl 4-(2-(diethylamino)ethoxy)-3,5-diiodophenyl; Ketone, 2-butyl-3-benzofuranyl 4-[2-(diethylamino)ethoxy]-3,5-diiodophenyl (7CI,8CI); (2-Butyl-3-benzofuranyl)(4-(2-(diethylamino)ethoxy)-3,5-diidophenyl)methanone; (2-butyl-1-benzofuran-3-yl)(4-{[2-(diethylamino)ethyl]oxy}-3,5-diiodophenyl)methanone; (2-butyl-1-benzofuran-3-yl)-[4-(2-diethylaminoethyloxy)-3,5-diiodophenyl]methanone; (2-butyl-1-benzofuran-3-yl){4-[2-(diethylamino)ethoxy]-3,5-diiodophenyl}methanone; (2-butylbenzofuran-3-yl)-[4-(2-diethylaminoethoxy)-3,5-diiodo-phenyl]-methanone; (2-{4-[(2-butyl-1-benzofuran-3-yl)carbonyl]-2,6-diiodophenoxy}ethyl)diethylamine; 2-Butyl-3-(3,5-diiodo-4-(2-diethylaminoethoxy)benzoyl)benzofuran; 2-Butyl-3-(3,5-diiodo-4-(beta-diethylaminoethoxy)benzoyl)benzofuran; 2-Butyl-3-(4'-beta-N-diethylaminoethoxy-3',5'-diiodobenzoyl)benzofuran; 2-Butyl-3-[3,5-diiodo-4-(2-di; 2-Butyl-3-benzofuranyl 4-(2-(diethylamino)ethoxy)-3,5-diiodophenyl ketone; 2-Butyl-3-benzofuranyl p-((2-diethylamino)ethoxy)-m,m-diiodophenyl ketone; 2-Butyl-3-benzofuranyl p-[(2-diethylamino)ethoxy]-m,m-diiodophenyl ketone; 2-n-Butyl-3',5'-diiodo-4'-N-diethylaminoethoxy-3-benzoylbenzofuran
|
||||||||||||||||||||||||||||||
| Indication |
|
||||||||||||||||||||||||||||||
| Therapeutic Class |
Antiarrhythmic Agents
|
||||||||||||||||||||||||||||||
| Affected Organisms |
Humans and other mammals
|
||||||||||||||||||||||||||||||
| ATC Code | |||||||||||||||||||||||||||||||
| Drug Type |
Small molecular drug
|
||||||||||||||||||||||||||||||
| Structure |
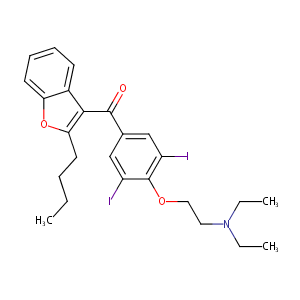 |
||||||||||||||||||||||||||||||
| 3D MOL | 2D MOL | ||||||||||||||||||||||||||||||
| #Ro5 Violations (Lipinski): 3 | Molecular Weight (mw) | 645.3 | |||||||||||||||||||||||||||||
| Logarithm of the Partition Coefficient (xlogp) | 7.6 | ||||||||||||||||||||||||||||||
| Rotatable Bond Count (rotbonds) | 11 | ||||||||||||||||||||||||||||||
| Hydrogen Bond Donor Count (hbonddonor) | 0 | ||||||||||||||||||||||||||||||
| Hydrogen Bond Acceptor Count (hbondacc) | 4 | ||||||||||||||||||||||||||||||
| ADMET Property |
|
||||||||||||||||||||||||||||||
| Adverse Drug Reaction (ADR) |
|
||||||||||||||||||||||||||||||
| Chemical Identifiers |
|
||||||||||||||||||||||||||||||
| Cross-matching ID | |||||||||||||||||||||||||||||||
| Combinatorial Drugs (CBD) | Click to Jump to the Detailed CBD Information of This Drug | ||||||||||||||||||||||||||||||
| Repurposed Drugs (RPD) | Click to Jump to the Detailed RPD Information of This Drug | ||||||||||||||||||||||||||||||
Molecular Interaction Atlas of This Drug
 Drug Therapeutic Target (DTT) |
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
 Drug Transporter (DTP) |
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
 Drug-Metabolizing Enzyme (DME) |
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
 Drug Off-Target (DOT) |
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Molecular Interaction Atlas (MIA) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Drug-Drug Interaction (DDI) Information of This Drug
|
Coadministration of a Drug Treating the Same Disease as Amiodarone
Coadministration of a Drug Treating the Disease Different from Amiodarone (Comorbidity)
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Drug Inactive Ingredient(s) (DIG) and Formulation(s) of This Drug
References
| 1 | URL: http://www.guidetopharmacology.org Nucleic Acids Res. 2015 Oct 12. pii: gkv1037. The IUPHAR/BPS Guide to PHARMACOLOGY in 2016: towards curated quantitative interactions between 1300 protein targets and 6000 ligands. (Ligand id: 2566). | ||||
|---|---|---|---|---|---|
| 2 | FDA Approved Drug Products from FDA Official Website. 2019. Application Number: (ANDA) 204742. | ||||
| 3 | ClinicalTrials.gov (NCT04351763) Amiodarone or Verapamil in COVID-19 Hospitalized Patients With Symptoms. U.S. National Institutes of Health. | ||||
| 4 | BDDCS applied to over 900 drugs | ||||
| 5 | Trend Analysis of a Database of Intravenous Pharmacokinetic Parameters in Humans for 1352 Drug Compounds | ||||
| 6 | Estimating the safe starting dose in phase I clinical trials and no observed effect level based on QSAR modeling of the human maximum recommended daily dose | ||||
| 7 | ADReCS-Target: target profiles for aiding drug safety research and application. Nucleic Acids Res. 2018 Jan 4;46(D1):D911-D917. doi: 10.1093/nar/gkx899. | ||||
| 8 | Drugs@FDA. U.S. Food and Drug Administration. U.S. Department of Health & Human Services. | ||||
| 9 | Characterization of spike glycoprotein of SARS-CoV-2 on virus entry and its immune cross-reactivity with SARS-CoV. Nat Commun. 2020 Mar 27;11(1):1620. | ||||
| 10 | Mammalian drug efflux transporters of the ATP binding cassette (ABC) family in multidrug resistance: A review of the past decade. Cancer Lett. 2016 Jan 1;370(1):153-64. | ||||
| 11 | The metabolism of amiodarone by various CYP isoenzymes of human and rat, and the inhibitory influence of ketoconazole. J Pharm Pharm Sci. 2008;11(1):147-59. | ||||
| 12 | Potentially significant drug interactions of class III antiarrhythmic drugs. Drug Saf. 2003;26(6):421-38. | ||||
| 13 | Substrates, inducers, inhibitors and structure-activity relationships of human Cytochrome P450 2C9 and implications in drug development. Curr Med Chem. 2009;16(27):3480-675. | ||||
| 14 | A significant role of human cytochrome P450 2C8 in amiodarone N-deethylation: an approach to predict the contribution with relative activity factor. Drug Metab Dispos. 2000 Nov;28(11):1303-10. | ||||
| 15 | Role of desethylamiodarone in the anticoagulant effect of concurrent amiodarone and warfarin therapy. J Cardiovasc Pharmacol Ther. 2001 Oct;6(4):363-7. | ||||
| 16 | Functional characterization of five novel CYP2C8 variants, G171S, R186X, R186G, K247R, and K383N, found in a Japanese population. Drug Metab Dispos. 2005 May;33(5):630-6. | ||||
| 17 | Identification by automated screening of a small molecule that selectively eliminates neural stem cells derived from hESCs but not dopamine neurons. PLoS One. 2009 Sep 23;4(9):e7155. | ||||
| 18 | Activation of autophagy rescues amiodarone-induced apoptosis of lung epithelial cells and pulmonary toxicity in rats. Toxicol Sci. 2013 Nov;136(1):193-204. doi: 10.1093/toxsci/kft168. Epub 2013 Aug 2. | ||||
| 19 | A toxicogenomic approach to drug-induced phospholipidosis: analysis of its induction mechanism and establishment of a novel in vitro screening system. Toxicol Sci. 2005 Feb;83(2):282-92. | ||||
| 20 | Buss J, Neuss H, Bilgin Y, Schlepper M "Malignant ventricular tachyarrhythmias in association with propafenone treatment." Eur Heart J 6 (1985): 424-8. [PMID: 4043097] | ||||
| 21 | Agencia Espaola de Medicamentos y Productos Sanitarios Healthcare "Centro de informacion online de medicamentos de la AEMPS - CIMA.". | ||||
| 22 | Cerner Multum, Inc. "Australian Product Information.". | ||||
| 23 | Canadian Pharmacists Association. | ||||
| 24 | Cerner Multum, Inc. "UK Summary of Product Characteristics.". | ||||
| 25 | Fabre G, Julian B, Saint-Aubert B, Joyeux H, Berger Y "Evidence for CYP3A-mediated N-deethylation of amiodarone in human liver microsomal fractions." Drug Metab Dispos 21 (1993): 978-85. [PMID: 7905403] | ||||
| 26 | Product Information. Arcapta Neohaler (indacaterol). Novartis Pharmaceuticals, East Hanover, NJ. | ||||
| 27 | Bengtsson B, Fagerstrom PO "Extrapulmonary effects of terbutaline during prolonged administration." Clin Pharmacol Ther 31 (1982): 726-32. [PMID: 7042176] | ||||
| 28 | Bolland MJ, Bagg W, Thomas MG, Lucas JA, Ticehurst R, Black PN "Cushing's syndrome due to interaction between inhaled corticosteroids and itraconazole." Ann Pharmacother 38 (2004): 46-9. [PMID: 14742792] | ||||
| 29 | Soto J, Sacristan JA, Arellano F, Hazas J "Possible theophylline-amiodarone interaction." DICP 24 (1990): 1115. [PMID: 2275239] | ||||
| 30 | Ansari SR, Chopra N "Gatifloxacin and Prolonged QT Interval." Am J Med Sci 327 (2004): 55-6. [PMID: 14722399] | ||||
| 31 | Bailey DG, Dresser GR, Kreeft JH, Munoz C, Freeman DJ, Bend JR "Grapefruit-felodipine interaction: Effect of unprocessed fruit and probable active ingredients." Clin Pharmacol Ther 68 (2000): 468-77. [PMID: 11103749] | ||||
| 32 | Nattel S, Ranger S, Talajic M, et al "Erythromycin-induced long QT syndrome: concordance with quinidine and underlying cellular electrophysiologic mechanism." Am J Med 89 (1990): 235-8. [PMID: 2382671] | ||||
| 33 | Product Information. Turalio (pexidartinib). Daiichi Sankyo, Inc., Parsippany, NJ. | ||||
| 34 | Product Information. Tukysa (tucatinib). Seattle Genetics Inc, Bothell, WA. | ||||
| 35 | Product Information. Jevtana (cabazitaxel). sanofi-aventis , Bridgewater, NJ. | ||||
| 36 | Product Information. Opsumit (macitentan). Actelion Pharmaceuticals US Inc, South San Francisco, CA. | ||||
| 37 | Goernig M, Kirmeier T, Krack A, Hartog CS, Figulla HR, Leder U "Iohexol contrast medium induces QT prolongation in amiodarone patients." Br J Clin Pharmacol 58 (2004): 96-98. [PMID: 15206999] | ||||
| 38 | Antonelli D, Atar S, Freedberg NA, Rosenfeld T "Torsade de pointes in patients on chronic amiodarone treatment: contributing factors and drug interactions." Isr Med Assoc J 7 (2005): 163-5. [PMID: 15792261] | ||||
| 39 | Caraco Y, Chajek-Shaul T "The incidence and clinical significance of amiodarone and acenocoumarol interaction." Thromb Haemost 62 (1989): 906-8. [PMID: 2595664] | ||||
| 40 | Product Information. Ofev (nintedanib). Boehringer Ingelheim, Ridgefield, CT. | ||||
| 41 | Product Information. Kalydeco (ivacaftor). Vertex Pharmaceuticals, Cambridge, MA. | ||||
| 42 | Product Information. Xarelto (rivaroxaban). Bayer Inc, Toronto, IA. | ||||
| 43 | Product Information. Viibryd (vilazodone). Trovis Pharmaceuticals LLC, New Haven, CT. | ||||
| 44 | Product Information. Serzone (nefazodone). Bristol-Myers Squibb, Princeton, NJ. | ||||
| 45 | Product Information. Osphena (ospemifene). Shionogi USA Inc, Florham Park, NJ. | ||||
| 46 | Product Information. Xcopri (cenobamate). SK Life Science, Inc., Paramus, NJ. | ||||
| 47 | Product Information. Cordarone (amiodarone). Wyeth-Ayerst Laboratories, Philadelphia, PA. | ||||
| 48 | Leor J, Levartowsky D, Sharon C, Farfel Z "Amiodarone and beta-adrenergic blockers: an interaction with metoprolol but not with atenolol." Am Heart J 16 (1988): 206-7. [PMID: 3394625] | ||||
| 49 | Product Information. Tazverik (tazemetostat). Epizyme, Inc, Cambridge, MA. | ||||
| 50 | Product Information. Myrbetriq (mirabegron). Astellas Pharma US, Inc, Deerfield, IL. | ||||
| 51 | Bajaj BP, Baig MW, Perrins EJ "Amiodarone-induced torsades de pointes: the possible facilitatory role of digoxin." Int J Cardiol 33 (1991): 335-7. [PMID: 1743800] | ||||
| 52 | Product Information. Victrelis (boceprevir). Schering-Plough Corporation, Kenilworth, NJ. | ||||
| 53 | Product Information. Olysio (simeprevir). Janssen Pharmaceuticals, Titusville, NJ. | ||||
| 54 | Product Information. VFEND (voriconazole). Pfizer U.S. Pharmaceuticals, New York, NY. | ||||
| 55 | Product Information. Pifeltro (doravirine). Merck & Company Inc, Whitehouse Station, NJ. | ||||
| 56 | Product Information. Stribild (cobicistat/elvitegravir/emtricitabine/tenofov). Gilead Sciences, Foster City, CA. | ||||
| 57 | Product Information. Tivicay (dolutegravir). ViiV Healthcare, Research Triangle Park, NC. | ||||
| 58 | Product Information. Intelence (etravirine). Ortho Biotech Inc, Bridgewater, NJ. | ||||
| 59 | Product Information. Prezista (darunavir). Ortho Biotech Inc, Bridgewater, NJ. | ||||
| 60 | Product Information. Selzentry (maraviroc). Pfizer U.S. Pharmaceuticals Group, New York, NY. | ||||
| 61 | Product Information. Kynamro (mipomersen). Genzyme Corporation, Cambridge, MA. | ||||
| 62 | Product Information. Juxtapid (lomitapide). Aegerion Pharmaceuticals Inc, Cambridge, MA. | ||||
| 63 | Product Information. Samsca (tolvaptan). Otsuka American Pharmaceuticals Inc, Rockville, MD. | ||||
| 64 | Product Information. Altabax (retapamulin topical). GlaxoSmithKline, Research Triangle Park, NC. | ||||
| 65 | Product Information. Zurampic (lesinurad). Astra-Zeneca Pharmaceuticals, Wilmington, DE. | ||||
| 66 | Product Information. Hetlioz (tasimelteon). Vanda Pharmaceuticals Inc, Rockville, MD. | ||||
| 67 | Product Information. Caplyta (lumateperone). Intra-Cellular Therapies, Inc., New York, NY. | ||||
| 68 | Kerin NZ, Aragon E, Faitel K, Frumin H, Rubenfire M "Long-term efficacy and toxicity of high- and low-dose amiodarone regimens." J Clin Pharmacol 29 (1989): 418-23. [PMID: 2661600] | ||||
| 69 | Hill AG, Parry BR "Hypokalaemia following bowel cleansing with sodium phosphate." N Z Med J 109 (1996): 347. [PMID: 8862361] | ||||
| 70 | Product Information. Movantik (naloxegol). Astra-Zeneca Pharmaceuticals, Wilmington, DE. | ||||
| 71 | Product Information. Alunbrig (brigatinib). Ariad Pharmaceuticals Inc, Cambridge, MA. | ||||
| 72 | Product Information. Zepzelca (lurbinectedin). Jazz Pharmaceuticals, Palo Alto, CA. | ||||
| 73 | Product Information. Lorbrena (lorlatinib). Pfizer U.S. Pharmaceuticals Group, New York, NY. | ||||
| 74 | Product Information. Gavreto (pralsetinib). Blueprint Medicines Corporation, Cambridge, MA. | ||||
| 75 | Abernethy DR, Wesche DL, Barbey JT, et al. "Stereoselective halofantrine disposition and effect: concentration-related QTc prolongation." Br J Clin Pharmacol 51 (2001): 231-7. [PMID: 11298069] | ||||
| 76 | Al-Nawakil C, Willems L, Mauprivez C, et.al "Successful treatment of l-asparaginase-induced severe acute hepatotoxicity using mitochondrial cofactors." Leuk Lymphoma 55 (2014): 1670-4. [PMID: 24090500] | ||||
| 77 | Product Information. Zydelig (idelalisib). Gilead Sciences, Foster City, CA. | ||||
| 78 | Product Information. Copiktra (duvelisib). Verastem, Inc., Needham, MA. | ||||
| 79 | Ohnishi K, Yoshida H, Shigeno K, et al. "Prolongation of the QT interval and ventricular tachycardia in patients treated with arsenic trioxide for acute promyelocytic leukemia." Ann Intern Med 133 (2000): 881-5. [PMID: 11103058] | ||||
| 80 | Product Information. Calquence (acalabrutinib). Astra-Zeneca Pharmaceuticals, Wilmington, DE. | ||||
| 81 | Product Information. Imbruvica (ibrutinib). Pharmacyclics Inc, Sunnyvale, CA. | ||||
| 82 | Product Information. Iclusig (ponatinib). Ariad Pharmaceuticals Inc, Cambridge, MA. | ||||
| 83 | Product Information. Exjade (deferasirox). Novartis Pharmaceuticals, East Hanover, NJ. | ||||
| 84 | Product Information. Addyi (flibanserin). Sprout Pharmaceuticals, Raleigh, NC. | ||||
| 85 | Product Information. Lynparza (olaparib). Astra-Zeneca Pharmaceuticals, Wilmington, DE. | ||||
| 86 | Product Information. Zejula (niraparib). Tesaro Inc., Waltham, MA. | ||||
| 87 | Product Information. Xeglyze (abametapir topical). Dr. Reddy's Laboratories Inc, Upper Saddle River, NJ. | ||||
| 88 | Product Information. Macrilen (macimorelin). Aeterna Zentaris, Charleston, SC. | ||||
| 89 | Product Information. Xenleta (lefamulin). Nabriva Therapeutics US, Inc., King of Prussia, PA. | ||||
| 90 | Francis H, Tyndall A, Webb J "Severe vascular spasm due to erythromycin-ergotamine interaction." Clin Rheumatol 3 (1984): 243-6. [PMID: 6236021] | ||||
| 91 | Product Information. Zokinvy (lonafarnib). Eiger BioPharmaceuticals, Palo Alto, CA. | ||||
| 92 | Product Information. Nubeqa (darolutamide). Bayer HealthCare Pharmaceuticals Inc., Whippany, NJ. | ||||
| 93 | Product Information. Rapaflo (silodosin). Watson Pharmaceuticals, Corona, CA. | ||||
| 94 | Product Information. Duagen (dutasteride). GlaxoSmithKline Healthcare, Pittsburgh, PA. | ||||
| 95 | Product Information. Afinitor (everolimus). Novartis Pharmaceuticals, East Hanover, NJ. | ||||
| 96 | Product Information. Torisel (temsirolimus). Wyeth-Ayerst Laboratories, Philadelphia, PA. | ||||
| 97 | Product Information. Oxbryta (voxelotor). Global Blood Therapeutics, Inc., South San Francisco, CA. | ||||
| 98 | Product Information. Odomzo (sonidegib). Novartis Pharmaceuticals, East Hanover, NJ. | ||||
| 99 | Aronson JK, Grahame-Smith DG "Clinical pharmacology: adverse drug interactions." Br Med J 282 (1981): 288-91. [PMID: 6779990] | ||||
| 100 | Product Information. Vandetanib (vandetanib). Astra-Zeneca Pharmaceuticals, Wilmington, DE. | ||||
| 101 | Bergmann TK, Filppula AM, Launiainen T, Nielsen F, Backman J, Brosen K "Neurotoxicity and low paclitaxel clearance associated with concomitant clopidogrel therapy in a 60 year old Caucasian woman with ovarian carcinoma." Br J Clin Pharmacol (2015):. [PMID: 26446447] | ||||
| 102 | EMA. European Medicines Agency. European Union "EMA - List of medicines under additional monitoring.". | ||||
