| 1 |
Recurrent recessive mutation in deoxyguanosine kinase causes idiopathic noncirrhotic portal hypertension.Hepatology. 2016 Jun;63(6):1977-86. doi: 10.1002/hep.28499. Epub 2016 Mar 31.
|
| 2 |
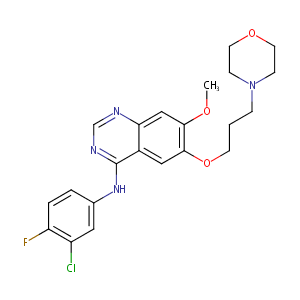
Gefitinib FDA Label
|
| 3 |
URL: http://www.guidetopharmacology.org Nucleic Acids Res. 2015 Oct 12. pii: gkv1037. The IUPHAR/BPS Guide to PHARMACOLOGY in 2016: towards curated quantitative interactions between 1300 protein targets and 6000 ligands. (Ligand id: 4941).
|
| 4 |
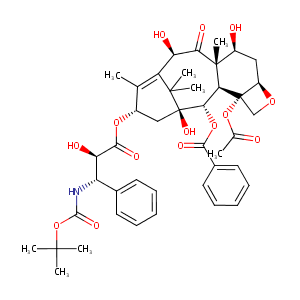
Docetaxel FDA Label
|
| 5 |
URL: http://www.guidetopharmacology.org Nucleic Acids Res. 2015 Oct 12. pii: gkv1037. The IUPHAR/BPS Guide to PHARMACOLOGY in 2016: towards curated quantitative interactions between 1300 protein targets and 6000 ligands. (Ligand id: 6809).
|
| 6 |
Synergistic antitumor effect of S-1 and the epidermal growth factor receptor inhibitor gefitinib in non-small cell lung cancer cell lines: role of gefitinib-induced down-regulation of thymidylate synthase. Mol Cancer Ther. 2008 Mar;7(3):599-606.
|
| 7 |
Frankincense myrrh attenuates hepatocellular carcinoma by regulating tumor blood vessel development through multiple epidermal growth factor receptor-mediated signaling pathways. World J Gastrointest Oncol. 2022 Feb 15;14(2):450-477. doi: 10.4251/wjgo.v14.i2.450.
|
| 8 |
EGFR inhibitors enhanced the susceptibility to NK cell-mediated lysis of lung cancer cells. J Immunother. 2011 May;34(4):372-81. doi: 10.1097/CJI.0b013e31821b724a.
|
| 9 |
Overcoming acquired resistance of gefitinib in lung cancer cells without T790M by AZD9291 or Twist1 knockdown in vitro and in vivo. Arch Toxicol. 2019 Jun;93(6):1555-1571. doi: 10.1007/s00204-019-02453-2. Epub 2019 Apr 16.
|
| 10 |
DNA repair gene polymorphisms and benefit from gefitinib in never-smokers with lung adenocarcinoma. Cancer. 2011 Jul 15;117(14):3201-8. doi: 10.1002/cncr.25863. Epub 2011 Jan 24.
|
| 11 |
Gefitinib ('Iressa', ZD1839) and new epidermal growth factor receptor inhibitors. Br J Cancer. 2004 Feb 9;90(3):566-72.
|
| 12 |
Gefitinib-phenytoin interaction is not correlated with the C-erythromycin breath test in healthy male volunteers. Br J Clin Pharmacol. 2009 Aug;68(2):226-37.
|
| 13 |
Mammalian drug efflux transporters of the ATP binding cassette (ABC) family in multidrug resistance: A review of the past decade. Cancer Lett. 2016 Jan 1;370(1):153-64.
|
| 14 |
Contribution of OATP1B1 and OATP1B3 to the disposition of sorafenib and sorafenib-glucuronide. Clin Cancer Res. 2013 Mar 15;19(6):1458-66.
|
| 15 |
Differential metabolism of gefitinib and erlotinib by human cytochrome P450 enzymes. Clin Cancer Res. 2007 Jun 15;13(12):3731-7.
|
| 16 |
Pharmacokinetic drug interactions of gefitinib with rifampicin, itraconazole and metoprolol. Clin Pharmacokinet. 2005;44(10):1067-81.
|
| 17 |
Pharmacogenetics of ABCG2 and adverse reactions to gefitinib. J Natl Cancer Inst. 2006 Dec 6;98(23):1739-42.
|
| 18 |
Dasatinib (BMS-354825) selectively induces apoptosis in lung cancer cells dependent on epidermal growth factor receptor signaling for survival. Cancer Res. 2006 Jun 1;66(11):5542-8. doi: 10.1158/0008-5472.CAN-05-4620.
|
| 19 |
Identification of genes linked to gefitinib treatment in prostate cancer cell lines with or without resistance to androgen: a clue to application of gefitinib to hormone-resistant prostate cancer. Oncol Rep. 2006 Jun;15(6):1453-60.
|
| 20 |
Interference with bile salt export pump function is a susceptibility factor for human liver injury in drug development. Toxicol Sci. 2010 Dec; 118(2):485-500.
|
| 21 |
Effects and mechanisms of betulinic acid on improving EGFR TKI-resistance of lung cancer cells. Environ Toxicol. 2018 Nov;33(11):1153-1159.
|
| 22 |
Antiproliferative effects of gefitinib are associated with suppression of E2F-1 expression and telomerase activity. Anticancer Res. 2006 Sep-Oct;26(5A):3387-91.
|
| 23 |
Leptomycin B reduces primary and acquired resistance of gefitinib in lung cancer cells. Toxicol Appl Pharmacol. 2017 Nov 15;335:16-27. doi: 10.1016/j.taap.2017.09.017. Epub 2017 Sep 21.
|
| 24 |
EGFR tyrosine kinase inhibitors activate autophagy as a cytoprotective response in human lung cancer cells. PLoS One. 2011;6(6):e18691. doi: 10.1371/journal.pone.0018691. Epub 2011 Jun 2.
|
| 25 |
ZD1839 induces p15INK4b and causes G1 arrest by inhibiting the mitogen-activated protein kinase/extracellular signal-regulated kinase pathway. Mol Cancer Ther. 2007 May;6(5):1579-87. doi: 10.1158/1535-7163.MCT-06-0814.
|
| 26 |
Reactive metabolite of gefitinib activates inflammasomes: implications for gefitinib-induced idiosyncratic reaction. J Toxicol Sci. 2020;45(11):673-680. doi: 10.2131/jts.45.673.
|
| 27 |
Systems pharmacological analysis of drugs inducing stevens-johnson syndrome and toxic epidermal necrolysis. Chem Res Toxicol. 2015 May 18;28(5):927-34. doi: 10.1021/tx5005248. Epub 2015 Apr 3.
|
| 28 |
Bidirectional cross talk between ERalpha and EGFR signalling pathways regulates tamoxifen-resistant growth. Breast Cancer Res Treat. 2006 Mar;96(2):131-46. doi: 10.1007/s10549-005-9070-2. Epub 2005 Oct 27.
|
| 29 |
Epidermal growth factor receptor activity determines response of colorectal cancer cells to gefitinib alone and in combination with chemotherapy. Clin Cancer Res. 2005 Oct 15;11(20):7480-9. doi: 10.1158/1078-0432.CCR-05-0328.
|
| 30 |
Implication of the insulin-like growth factor-IR pathway in the resistance of non-small cell lung cancer cells to treatment with gefitinib. Clin Cancer Res. 2007 May 1;13(9):2795-803. doi: 10.1158/1078-0432.CCR-06-2077.
|
| 31 |
Evidence of securin-mediated resistance to gefitinib-induced apoptosis in human cancer cells. Chem Biol Interact. 2013 Apr 25;203(2):412-22. doi: 10.1016/j.cbi.2013.03.011. Epub 2013 Mar 22.
|
| 32 |
Crosstalk between alveolar macrophages and alveolar epithelial cells/fibroblasts contributes to the pulmonary toxicity of gefitinib. Toxicol Lett. 2021 Mar 1;338:1-9. doi: 10.1016/j.toxlet.2020.11.011. Epub 2020 Nov 25.
|
| 33 |
Cytotoxicity of 34 FDA approved small-molecule kinase inhibitors in primary rat and human hepatocytes. Toxicol Lett. 2018 Jul;291:138-148. doi: 10.1016/j.toxlet.2018.04.010. Epub 2018 Apr 12.
|
| 34 |
Growth of hormone-dependent MCF-7 breast cancer cells is promoted by constitutive caveolin-1 whose expression is lost in an EGF-R-mediated manner during development of tamoxifen resistance. Breast Cancer Res Treat. 2010 Feb;119(3):575-91. doi: 10.1007/s10549-009-0355-8. Epub 2009 Mar 15.
|
| 35 |
Dihydromyricetin suppresses tumor growth via downregulation of the EGFR/Akt/survivin signaling pathway. J Biochem Mol Toxicol. 2023 Jun;37(6):e23328. doi: 10.1002/jbt.23328. Epub 2023 Feb 19.
|
| 36 |
The anti-cancer drug gefitinib accelerates Fas-mediated apoptosis by enhancing caspase-8 activation in cancer cells. J Toxicol Sci. 2019;44(6):435-440. doi: 10.2131/jts.44.435.
|
| 37 |
Autophagy Inhibition Overcomes the Antagonistic Effect Between Gefitinib and Cisplatin in Epidermal Growth Factor Receptor Mutant Non--Small-Cell Lung Cancer Cells. Clin Lung Cancer. 2015 Sep;16(5):e55-66. doi: 10.1016/j.cllc.2015.03.006. Epub 2015 Apr 2.
|
| 38 |
Nrf2 but not autophagy inhibition is associated with the survival of wild-type epidermal growth factor receptor non-small cell lung cancer cells. Toxicol Appl Pharmacol. 2016 Nov 1;310:140-149. doi: 10.1016/j.taap.2016.09.010. Epub 2016 Sep 14.
|
| 39 |
Association of CYP1A1 and CYP1B1 inhibition in in vitro assays with drug-induced liver injury. J Toxicol Sci. 2021;46(4):167-176. doi: 10.2131/jts.46.167.
|
| 40 |
Susceptibility to natural killer cell-mediated lysis of colon cancer cells is enhanced by treatment with epidermal growth factor receptor inhibitors through UL16-binding protein-1 induction. Cancer Sci. 2012 Jan;103(1):7-16. doi: 10.1111/j.1349-7006.2011.02109.x. Epub 2011 Nov 15.
|
| 41 |
Combined tamoxifen and gefitinib in non-small cell lung cancer shows antiproliferative effects. Biomed Pharmacother. 2010 Feb;64(2):88-92. doi: 10.1016/j.biopha.2009.06.010. Epub 2009 Oct 23.
|
| 42 |
Inorganic arsenic exposure promotes malignant progression by HDAC6-mediated down-regulation of HTRA1. J Appl Toxicol. 2023 Aug;43(8):1214-1224. doi: 10.1002/jat.4457. Epub 2023 Mar 11.
|
| 43 |
A high-throughput screen for teratogens using human pluripotent stem cells. Toxicol Sci. 2014 Jan;137(1):76-90. doi: 10.1093/toxsci/kft239. Epub 2013 Oct 23.
|
| 44 |
Inhibition of SREBP increases gefitinib sensitivity in non-small cell lung cancer cells. Oncotarget. 2016 Aug 9;7(32):52392-52403.
|
| 45 |
The K-Ras effector p38 MAPK confers intrinsic resistance to tyrosine kinase inhibitors by stimulating EGFR transcription and EGFR dephosphorylation. J Biol Chem. 2017 Sep 8;292(36):15070-15079. doi: 10.1074/jbc.M117.779488. Epub 2017 Jul 24.
|
| 46 |
Slug confers resistance to the epidermal growth factor receptor tyrosine kinase inhibitor. Am J Respir Crit Care Med. 2011 Apr 15;183(8):1071-9. doi: 10.1164/rccm.201009-1440OC. Epub 2010 Oct 29.
|
| 47 |
Identification of protein expression alterations in gefitinib-resistant human lung adenocarcinoma: PCNT and mPR play key roles in the development of gefitinib-associated resistance. Toxicol Appl Pharmacol. 2015 Nov 1;288(3):359-73. doi: 10.1016/j.taap.2015.08.008. Epub 2015 Aug 20.
|
| 48 |
Induction of CYP1A1 increases gefitinib-induced oxidative stress and apoptosis in A549 cells. Toxicol In Vitro. 2017 Oct;44:36-43.
|
| 49 |
Prediction of sensitivity of advanced non-small cell lung cancers to gefitinib (Iressa, ZD1839). Hum Mol Genet. 2004 Dec 15;13(24):3029-43. doi: 10.1093/hmg/ddh331. Epub 2004 Oct 20.
|
| 50 |
The transcription factor FOXO3a is a crucial cellular target of gefitinib (Iressa) in breast cancer cells. Mol Cancer Ther. 2007 Dec;6(12 Pt 1):3169-79. doi: 10.1158/1535-7163.MCT-07-0507.
|
| 51 |
Aurora-A promotes gefitinib resistance via a NF-B signaling pathway in p53 knockdown lung cancer cells. Biochem Biophys Res Commun. 2011 Feb 11;405(2):168-72. doi: 10.1016/j.bbrc.2011.01.001. Epub 2011 Jan 7.
|
| 52 |
BIM induction of apoptosis triggered by EGFR-sensitive and resistance cell lines of non-small-cell lung cancer. Med Oncol. 2011 Jun;28(2):572-7. doi: 10.1007/s12032-010-9470-y. Epub 2010 Mar 17.
|
| 53 |
E3 ubiquitin ligase RNF180 reduces sensitivity of triple-negative breast cancer cells to Gefitinib by downregulating RAD51. Chem Biol Interact. 2022 Feb 25;354:109798. doi: 10.1016/j.cbi.2022.109798. Epub 2022 Jan 6.
|
| 54 |
Increases of amphiregulin and transforming growth factor-alpha in serum as predictors of poor response to gefitinib among patients with advanced non-small cell lung cancers. Cancer Res. 2005 Oct 15;65(20):9176-84. doi: 10.1158/0008-5472.CAN-05-1556.
|
| 55 |
Synergistic effects of docetaxel and S-1 by modulating the expression of metabolic enzymes of 5-fluorouracil in human gastric cancer cell lines. Int J Cancer. 2006 Aug 15;119(4):783-91.
|
| 56 |
Induction of tubulin by docetaxel is associated with p53 status in human non small cell lung cancer cell lines. Int J Cancer. 2006 Jan 15;118(2):317-25. doi: 10.1002/ijc.21372.
|
| 57 |
Cyclooxygenase-2 inhibitor celecoxib augments chemotherapeutic drug-induced apoptosis by enhancing activation of caspase-3 and -9 in prostate cancer cells. Int J Cancer. 2005 Jun 20;115(3):484-92. doi: 10.1002/ijc.20878.
|
| 58 |
Docetaxel: a review of its use in metastatic breast cancer. Drugs. 2005;65(17):2513-31.
|
| 59 |
Human intestinal transporter database: QSAR modeling and virtual profiling of drug uptake, efflux and interactions. Pharm Res. 2013 Apr;30(4):996-1007.
|
| 60 |
Transport of diclofenac by breast cancer resistance protein (ABCG2) and stimulation of multidrug resistance protein 2 (ABCC2)-mediated drug transport by diclofenac and benzbromarone. Drug Metab Dispos. 2009 Jan;37(1):129-36.
|
| 61 |
Effect of ABCB1 C3435T polymorphism on docetaxel pharmacokinetics according to menopausal status in breast cancer patients. Br J Cancer. 2010 Aug 10;103(4):560-6.
|
| 62 |
Ixabepilone, a novel microtubule-targeting agent for breast cancer, is a substrate for P-glycoprotein (P-gp/MDR1/ABCB1) but not breast cancer resistance protein (BCRP/ABCG2). J Pharmacol Exp Ther. 2011 May;337(2):423-32.
|
| 63 |
FDA Drug Development and Drug Interactions
|
| 64 |
Rapid screening of antineoplastic candidates for the human organic anion transporter OATP1B3 substrates using fluorescent probes. Cancer Lett. 2008 Feb 18;260(1-2):163-9.
|
| 65 |
RNA-sequencing dissects the transcriptome of polyploid cancer cells that are resistant to combined treatments of cisplatin with paclitaxel and docetaxel. Mol Biosyst. 2017 Sep 26;13(10):2125-2134.
|
| 66 |
Modulation of the ATPase and transport activities of broad-acting multidrug resistance factor ABCC10 (MRP7). Cancer Res. 2012 Dec 15;72(24):6457-67.
|
| 67 |
Randomized pharmacokinetic and pharmacodynamic study of docetaxel: dosing based on body-surface area compared with individualized dosing based on cytochrome P450 activity estimated using a urinary metabolite of exogenous cortisol. J Clin Oncol. 2005 Feb 20;23(6):1061-9.
|
| 68 |
Drug Interactions Flockhart Table
|
| 69 |
Concise prediction models of anticancer efficacy of 8 drugs using expression data from 12 selected genes. Int J Cancer. 2004 Sep 10;111(4):617-26. doi: 10.1002/ijc.20289.
|
| 70 |
Association of genetic polymorphisms in SLCO1B3 and ABCC2 with docetaxel-induced leukopenia. Cancer Sci. 2008 May;99(5):967-72.
|
| 71 |
Enhanced in vitro invasiveness and drug resistance with altered gene expression patterns in a human lung carcinoma cell line after pulse selection with anticancer drugs. Int J Cancer. 2004 Sep 10;111(4):484-93. doi: 10.1002/ijc.20230.
|
| 72 |
Enhanced chemotherapeutic efficacy of docetaxel in human lung cancer cell line via GLUT1 inhibitor. J Biochem Mol Toxicol. 2023 Jun;37(6):e23348. doi: 10.1002/jbt.23348. Epub 2023 Mar 31.
|
| 73 |
Comparison of burst of reactive oxygen species and activation of caspase-3 in apoptosis of K562 and HL-60 cells induced by docetaxel. Cancer Lett. 2004 Oct 8;214(1):103-13. doi: 10.1016/j.canlet.2004.03.047.
|
| 74 |
Down-regulation of intratumoral aromatase messenger RNA levels by docetaxel in human breast cancers. Clin Cancer Res. 2004 Dec 15;10(24):8163-9.
|
| 75 |
Pretreatment with paclitaxel enhances apo-2 ligand/tumor necrosis factor-related apoptosis-inducing ligand-induced apoptosis of prostate cancer cells by inducing death receptors 4 and 5 protein levels. Cancer Res. 2001 Jan 15;61(2):759-63.
|
| 76 |
Docetaxel-induced apoptosis of human melanoma is mediated by activation of c-Jun NH2-terminal kinase and inhibited by the mitogen-activated protein kinase extracellular signal-regulated kinase 1/2 pathway. Clin Cancer Res. 2007 Feb 15;13(4):1308-14. doi: 10.1158/1078-0432.CCR-06-2216.
|
| 77 |
PXR-mediated induction of P-glycoprotein by anticancer drugs in a human colon adenocarcinoma-derived cell line. Cancer Chemother Pharmacol. 2010 Sep;66(4):765-71. doi: 10.1007/s00280-009-1221-4. Epub 2009 Dec 30.
|
| 78 |
Docetaxel-induced prostate cancer cell death involves concomitant activation of caspase and lysosomal pathways and is attenuated by LEDGF/p75. Mol Cancer. 2009 Aug 28;8:68. doi: 10.1186/1476-4598-8-68.
|
| 79 |
Docetaxel induces p53-dependent apoptosis and synergizes with farnesyl transferase inhibitor r115777 in human epithelial cancer cells. Front Biosci. 2005 Sep 1;10:2566-75. doi: 10.2741/1720.
|
| 80 |
Kaempferol and quercetin stimulate granulocyte-macrophage colony-stimulating factor secretion in human prostate cancer cells. Mol Cell Endocrinol. 2008 Jun 11;287(1-2):57-64. doi: 10.1016/j.mce.2008.01.015. Epub 2008 Feb 3.
|
| 81 |
All-trans retinoic acid potentiates Taxotere-induced cell death mediated by Jun N-terminal kinase in breast cancer cells. Oncogene. 2004 Jan 15;23(2):426-33. doi: 10.1038/sj.onc.1207040.
|
| 82 |
Docetaxel Facilitates Endothelial Dysfunction through Oxidative Stress via Modulation of Protein Kinase C Beta: The Protective Effects of Sotrastaurin. Toxicol Sci. 2015 May;145(1):59-67. doi: 10.1093/toxsci/kfv017. Epub 2015 Jan 28.
|
| 83 |
Docetaxel induces apoptosis in hormone refractory prostate carcinomas during multiple treatment cycles. Br J Cancer. 2006 Jun 5;94(11):1592-8. doi: 10.1038/sj.bjc.6603129.
|
| 84 |
The proteasome inhibitor bortezomib enhances the activity of docetaxel in orthotopic human pancreatic tumor xenografts. Mol Cancer Ther. 2004 Jan;3(1):59-70.
|
| 85 |
Tubulin-targeting chemotherapy impairs androgen receptor activity in prostate cancer. Cancer Res. 2010 Oct 15;70(20):7992-8002. doi: 10.1158/0008-5472.CAN-10-0585. Epub 2010 Aug 31.
|
| 86 |
Anti-cancer effects of novel flavonoid vicenin-2 as a single agent and in synergistic combination with docetaxel in prostate cancer. Biochem Pharmacol. 2011 Nov 1;82(9):1100-9. doi: 10.1016/j.bcp.2011.07.078. Epub 2011 Jul 23.
|
| 87 |
Replication-dependent -H2AX formation is involved in docetaxel-induced apoptosis in NSCLC A549 cells. Oncol Rep. 2010 Nov;24(5):1297-305. doi: 10.3892/or_00000986.
|
| 88 |
Docetaxel induced cardiotoxicity. Heart. 2001 Aug;86(2):219. doi: 10.1136/heart.86.2.219.
|
| 89 |
Development and validation of the TGx-HDACi transcriptomic biomarker to detect histone deacetylase inhibitors in human TK6 cells. Arch Toxicol. 2021 May;95(5):1631-1645. doi: 10.1007/s00204-021-03014-2. Epub 2021 Mar 26.
|
| 90 |
[The mechanism of docetaxel-induced apoptosis in human lung cancer cells]. Zhonghua Zhong Liu Za Zhi. 2000 May;22(3):208-11.
|
| 91 |
Sensitization to docetaxel in prostate cancer cells by green tea and quercetin. J Nutr Biochem. 2015 Apr;26(4):408-15. doi: 10.1016/j.jnutbio.2014.11.017. Epub 2015 Jan 15.
|
| 92 |
Azidothymidine and cisplatin increase p14ARF expression in OVCAR-3 ovarian cancer cell line. Toxicol Appl Pharmacol. 2006 Oct 1;216(1):89-97. doi: 10.1016/j.taap.2006.04.015. Epub 2006 May 19.
|
| 93 |
Role of delta-like ligand-4 in chemoresistance against docetaxel in MCF-7 cells. Hum Exp Toxicol. 2017 Apr;36(4):328-338. doi: 10.1177/0960327116650006. Epub 2016 Jun 22.
|
| 94 |
Roles of CYP3A4, CYP3A5 and CYP2C8 drug-metabolizing enzymes in cellular cytostatic resistance. Chem Biol Interact. 2021 May 1;340:109448. doi: 10.1016/j.cbi.2021.109448. Epub 2021 Mar 26.
|
| 95 |
Focal adhesion kinase silencing augments docetaxel-mediated apoptosis in ovarian cancer cells. Clin Cancer Res. 2005 Dec 15;11(24 Pt 1):8829-36. doi: 10.1158/1078-0432.CCR-05-1728.
|
| 96 |
Enhanced Bax in oral SCC in relation to antitumor effects of chemotherapy. J Oral Pathol Med. 2005 Feb;34(2):93-9. doi: 10.1111/j.1600-0714.2004.00257.x.
|
| 97 |
Pharmacogenomics variation in drug metabolizing enzymes and transporters in relation to docetaxel toxicity in Lebanese breast cancer patients: paving the way for OMICs in low and middle income countries. OMICS. 2013 Jul;17(7):353-67. doi: 10.1089/omi.2013.0019. Epub 2013 Jun 11.
|
| 98 |
Specific kinesin expression profiles associated with taxane resistance in basal-like breast cancer. Breast Cancer Res Treat. 2012 Feb;131(3):849-58. doi: 10.1007/s10549-011-1500-8. Epub 2011 Apr 9.
|
| 99 |
PAR1-mediated NFkappaB activation promotes survival of prostate cancer cells through a Bcl-xL-dependent mechanism. J Cell Biochem. 2005 Oct 15;96(3):641-52. doi: 10.1002/jcb.20533.
|
| 100 |
Regulators of G-Protein signaling RGS10 and RGS17 regulate chemoresistance in ovarian cancer cells. Mol Cancer. 2010 Nov 2;9:289. doi: 10.1186/1476-4598-9-289.
|
| 101 |
Mitotic checkpoint genes, hsMAD2 and BubR1, in oesophageal squamous cancer cells and their association with 5-fluorouracil and cisplatin-based radiochemotherapy. Clin Oncol (R Coll Radiol). 2008 Oct;20(8):639-46. doi: 10.1016/j.clon.2008.06.010. Epub 2008 Aug 8.
|
| 102 |
EGFR mediates docetaxel resistance in human castration-resistant prostate cancer through the Akt-dependent expression of ABCB1 (MDR1). Arch Toxicol. 2015 Apr;89(4):591-605. doi: 10.1007/s00204-014-1275-x. Epub 2014 Jun 3.
|
| 103 |
[Antisense RNA targeting survivin enhances the chemosensitivity of LOVO/Adr cells to taxotere]. Zhonghua Wei Chang Wai Ke Za Zhi. 2005 Sep;8(5):455-8.
|
| 104 |
Manganese superoxide dismutase is a promising target for enhancing chemosensitivity of basal-like breast carcinoma. Antioxid Redox Signal. 2014 May 20;20(15):2326-46. doi: 10.1089/ars.2013.5295. Epub 2013 Nov 14.
|
| 105 |
HER-2/neu as a predictive marker in a population of advanced breast cancer patients randomly treated either with single-agent doxorubicin or single-agent docetaxel. Breast Cancer Res Treat. 2004 Aug;86(3):197-206. doi: 10.1023/B:BREA.0000036783.88387.47.
|
| 106 |
Epigenetic inactivation of the CHFR gene in cervical cancer contributes to sensitivity to taxanes. Int J Oncol. 2007 Oct;31(4):713-20.
|
| 107 |
Differential effect of anti-apoptotic genes Bcl-xL and c-FLIP on sensitivity of MCF-7 breast cancer cells to paclitaxel and docetaxel. Anticancer Res. 2005 May-Jun;25(3c):2367-79.
|
| 108 |
CD146 expression in human breast cancer cell lines induces phenotypic and functional changes observed in Epithelial to Mesenchymal Transition. PLoS One. 2012;7(8):e43752. doi: 10.1371/journal.pone.0043752. Epub 2012 Aug 30.
|
| 109 |
Proteomic identification of differentially expressed proteins associated with the multiple drug resistance in methotrexate-resistant human breast cancer cells. Int J Oncol. 2014 Jul;45(1):448-58.
|
| 110 |
KIF14 promotes AKT phosphorylation and contributes to chemoresistance in triple-negative breast cancer. Neoplasia. 2014 Mar;16(3):247-56, 256.e2. doi: 10.1016/j.neo.2014.03.008.
|
| 111 |
The prognostic gene CRABP2 affects drug sensitivity by regulating docetaxel-induced apoptosis in breast invasive carcinoma: A pan-cancer analysis. Chem Biol Interact. 2023 Mar 1;373:110372. doi: 10.1016/j.cbi.2023.110372. Epub 2023 Feb 2.
|
| 112 |
Association of the CYP1B1*3 allele with survival in patients with prostate cancer receiving docetaxel. Mol Cancer Ther. 2008 Jan;7(1):19-26. doi: 10.1158/1535-7163.MCT-07-0557. Epub 2008 Jan 9.
|
| 113 |
ClinicalTrials.gov (NCT00086957) Gefitinib, Trastuzumab, and Docetaxel in Treating Patients With Metastatic Breast Cancer
|
| 114 |
ClinicalTrials.gov (NCT00048087) Iressa/Docetaxel in Non-Small-Cell Lung Cancer
|
| 115 |
Biologically active neutrophil chemokine pattern in tonsillitis.Clin Exp Immunol. 2004 Mar;135(3):511-8. doi: 10.1111/j.1365-2249.2003.02390.x.
|
|
|
|
|
|
|