Details of the Drug
General Information of Drug (ID: DMO946V)
| Drug Name |
Ticlopidine
|
||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Synonyms |
Ticlid; Ticlopidina; Ticlopidinum; PCR 5332; Ticlid (TN); Ticlopidin-Puren; Ticlopidina [INN-Spanish]; Ticlopidine (INN); Ticlopidine [INN:BAN]; Ticlopidinum [INN-Latin]; Ticlopidin-Puren (TN); 5-((2-Chlorophenyl)methyl)-4,5,6,7-tetrahydrothieno(3,2-c)pyridine; 5-(2-Chlorobenzyl)-4,5,6,7-tetrahydrothieno[3,2-c]pyridine; 5-[(2-chlorophenyl)methyl]-4,5,6,7-tetrahydrothieno[3,2-c]pyridine; 5-[(2-chlorophenyl)methyl]-6,7-dihydro-4H-thieno[3,2-c]pyridine
|
||||||||||||||||||||||||||||||||||||||||
| Indication |
|
||||||||||||||||||||||||||||||||||||||||
| Therapeutic Class |
Fibrinolytic Agents
|
||||||||||||||||||||||||||||||||||||||||
| Affected Organisms |
Humans and other mammals
|
||||||||||||||||||||||||||||||||||||||||
| ATC Code | |||||||||||||||||||||||||||||||||||||||||
| Drug Type |
Small molecular drug
|
||||||||||||||||||||||||||||||||||||||||
| Structure |
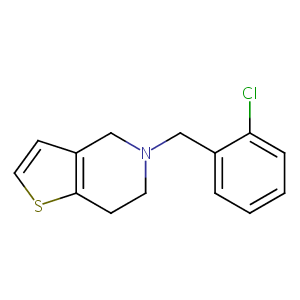 |
||||||||||||||||||||||||||||||||||||||||
| 3D MOL | 2D MOL | ||||||||||||||||||||||||||||||||||||||||
| #Ro5 Violations (Lipinski): 0 | Molecular Weight (mw) | 263.8 | |||||||||||||||||||||||||||||||||||||||
| Logarithm of the Partition Coefficient (xlogp) | 3.6 | ||||||||||||||||||||||||||||||||||||||||
| Rotatable Bond Count (rotbonds) | 2 | ||||||||||||||||||||||||||||||||||||||||
| Hydrogen Bond Donor Count (hbonddonor) | 0 | ||||||||||||||||||||||||||||||||||||||||
| Hydrogen Bond Acceptor Count (hbondacc) | 2 | ||||||||||||||||||||||||||||||||||||||||
| ADMET Property |
|
||||||||||||||||||||||||||||||||||||||||
| Adverse Drug Reaction (ADR) |
|
||||||||||||||||||||||||||||||||||||||||
| Chemical Identifiers |
|
||||||||||||||||||||||||||||||||||||||||
| Cross-matching ID | |||||||||||||||||||||||||||||||||||||||||
| Repurposed Drugs (RPD) | Click to Jump to the Detailed RPD Information of This Drug | ||||||||||||||||||||||||||||||||||||||||
Molecular Interaction Atlas of This Drug
 Drug Therapeutic Target (DTT) |
|
|||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
 Drug-Metabolizing Enzyme (DME) |
|
|||||||||||||||||||||||||||||||||||||||||
 Drug Off-Target (DOT) |
|
|||||||||||||||||||||||||||||||||||||||||
| Molecular Interaction Atlas (MIA) | ||||||||||||||||||||||||||||||||||||||||||
Molecular Expression Atlas of This Drug
| The Studied Disease | Acute coronary syndrome | |||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ICD Disease Classification | BA41 | |||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||
| Molecular Expression Atlas (MEA) | ||||||||||||||||||||||||||||||||||||||||||
Drug-Drug Interaction (DDI) Information of This Drug
|
Coadministration of a Drug Treating the Disease Different from Ticlopidine (Comorbidity)
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Drug Inactive Ingredient(s) (DIG) and Formulation(s) of This Drug
References
| 1 | Ticlopidine FDA Label | ||||
|---|---|---|---|---|---|
| 2 | URL: http://www.guidetopharmacology.org Nucleic Acids Res. 2015 Oct 12. pii: gkv1037. The IUPHAR/BPS Guide to PHARMACOLOGY in 2016: towards curated quantitative interactions between 1300 protein targets and 6000 ligands. (Ligand id: 7307). | ||||
| 3 | BDDCS applied to over 900 drugs | ||||
| 4 | Critical Evaluation of Human Oral Bioavailability for Pharmaceutical Drugs by Using Various Cheminformatics Approaches | ||||
| 5 | Trend Analysis of a Database of Intravenous Pharmacokinetic Parameters in Humans for 1352 Drug Compounds | ||||
| 6 | Estimating the safe starting dose in phase I clinical trials and no observed effect level based on QSAR modeling of the human maximum recommended daily dose | ||||
| 7 | Ticlopidine-induced hepatotoxicity is associated with specific human leukocyte antigen genomic subtypes in Japanese patients: a preliminary case-control study. Pharmacogenomics J. 2008 Feb;8(1):29-33. doi: 10.1038/sj.tpj.6500442. Epub 2007 Mar 6. | ||||
| 8 | Drugs@FDA. U.S. Food and Drug Administration. U.S. Department of Health & Human Services. | ||||
| 9 | Effect of health foods on cytochrome P450-mediated drug metabolism. J Pharm Health Care Sci. 2017 May 10;3:14. | ||||
| 10 | Substrates, inducers, inhibitors and structure-activity relationships of human Cytochrome P450 2C9 and implications in drug development. Curr Med Chem. 2009;16(27):3480-675. | ||||
| 11 | Product Information. Ticlid (ticlopidine). Syntex Laboratories Inc, Palo Alto, CA. | ||||
| 12 | Product Information. Aricept (donepezil). Pfizer US Pharmaceuticals, New York, NY. | ||||
| 13 | Product Information. Ranexa (ranolazine). Calmoseptine Inc, Huntington Beach, CA. | ||||
| 14 | Product Information. Onfi (clobazam). Lundbeck Inc, Deerfield, IL. | ||||
| 15 | Cerner Multum, Inc. "UK Summary of Product Characteristics.". | ||||
| 16 | Belle DJ, Ernest CS, Sauer JM, Smith BP, Thomasson HR, Witcher JW "Effect of potent CYP2D6 inhibition by paroxetine on atomoxetine pharmacokinetics." J Clin Pharmacol 42 (2002): 1219-27. [PMID: 12412820] | ||||
| 17 | Product Information. Brukinsa (zanubrutinib). BeiGene USA, Inc, San Mateo, CA. | ||||
| 18 | Alderman CP, Moritz CK, Ben-Tovim DI "Abnormal platelet aggregation associated with fluoxetine therapy." Ann Pharmacother 26 (1992): 1517-9. [PMID: 1482806] | ||||
| 19 | Gidal BE, Sorkness CA, McGill KA, Larson R, Levine RR "Evaluation of a potential enantioselective interaction between ticlopidine and warfarin in chronically anticoagulated patients." Ther Drug Monit 17 (1995): 33-8. [PMID: 7725374] | ||||
| 20 | Product Information. Ofev (nintedanib). Boehringer Ingelheim, Ridgefield, CT. | ||||
| 21 | Product Information. Fragmin (dalteparin). Pharmacia and Upjohn, Kalamazoo, MI. | ||||
| 22 | Cerner Multum, Inc. "Australian Product Information.". | ||||
| 23 | Product Information. Xarelto (rivaroxaban). Bayer Inc, Toronto, IA. | ||||
| 24 | Alderman CP, Seshadri P, Ben-Tovim DI "Effects of serotonin reuptake inhibitors on hemostasis." Ann Pharmacother 30 (1996): 1232-4. [PMID: 8913401] | ||||
| 25 | Product Information. Panhematin (hemin). Recordati Rare Diseases Inc, Lebanon, NJ. | ||||
| 26 | Heck AM, DeWitt BA, Lukes AL "Potential interactions between alternative therapies and warfarin." Am J Health Syst Pharm 57 (2000): 1221-7 quiz 1228-30. [PMID: 10902065] | ||||
| 27 | FDA. U.S. Food and Drug Administration "FDA Drug Safety Communication: Abnormal heart rhythms associated with high doses of Celexa (citalopram hydrobromide).". | ||||
| 28 | Canadian Pharmacists Association. | ||||
| 29 | Donahue S, Flockhart DA, Abernethy DR "Ticlopidine inhibits phenytoin clearance." Clin Pharmacol Ther 66 (1999): 563-8. [PMID: 10613611] | ||||
| 30 | Ha-Duong NT, Dijols S, Macherey AC, Goldstein JA, Dansette PM, Mansuy D "Ticlopidine as a selective mechanism-based inhibitor of human cytochrome P450 2C19." Biochemistry 40 (2001): 12112-22. [PMID: 11580286] | ||||
| 31 | Klotz U "The role of pharmacogenetics in the metabolism of antiepileptic drugs: pharmacokinetic and therapeutic implications." Clin Pharmacokinet 46 (2007): 271-9. [PMID: 17375979] | ||||
| 32 | Edeki TI, He HB, Wood AJJ "Pharmacogenetic explanation for excessive beta-blockade following timolol eye drops: potential for oral-ophthalmic drug interaction." JAMA 274 (1995): 1611-3. [PMID: 7474246] | ||||
| 33 | Product Information. Acular (ketorolac). Allergan Inc, Irvine, CA. | ||||
| 34 | Brynne N, Svanstrom C, AbergWistedt A, Hallen B, Bertilsson L "Fluoxetine inhibits the metabolism of tolterodin-pharmacokinetic implications and proposed clinical relevance." Br J Clin Pharmacol 48 (1999): 553-63. [PMID: 10583026] | ||||
| 35 | Product Information. Enablex (darifenacin). Novartis Pharmaceuticals, East Hanover, NJ. | ||||
| 36 | Product Information. Aptivus (tipranavir). Boehringer-Ingelheim, Ridgefield, CT. | ||||
| 37 | Product Information. Intelence (etravirine). Ortho Biotech Inc, Bridgewater, NJ. | ||||
| 38 | Product Information. Rozerem (ramelteon). Takeda Pharmaceuticals America, Lincolnshire, IL. | ||||
| 39 | Product Information. Hetlioz (tasimelteon). Vanda Pharmaceuticals Inc, Rockville, MD. | ||||
| 40 | Product Information. Lotronex (alosetron). Glaxo Wellcome, Research Triangle Park, NC. | ||||
| 41 | Product Information. Viberzi (eluxadoline). Actavis Pharma, Inc., Parsippany, NJ. | ||||
| 42 | Caruso V, Iacoviello L, Di Castelnuovo A, et.al "Thrombotic complications in childhood acute lymphoblastic leukemia: a meta-analysis of 17 prospective studies comprising 1752 pediatric patients." Blood 108 (2006): 2216-22. [PMID: 16804111] | ||||
| 43 | Product Information. Calquence (acalabrutinib). Astra-Zeneca Pharmaceuticals, Wilmington, DE. | ||||
| 44 | Agencia Espaola de Medicamentos y Productos Sanitarios Healthcare "Centro de informacion online de medicamentos de la AEMPS - CIMA.". | ||||
| 45 | Product Information. Iclusig (ponatinib). Ariad Pharmaceuticals Inc, Cambridge, MA. | ||||
| 46 | Product Information. Addyi (flibanserin). Sprout Pharmaceuticals, Raleigh, NC. | ||||
| 47 | Product Information. Sprycel (dasatinib). Bristol-Myers Squibb, Princeton, NJ. | ||||
| 48 | Product Information. Zontivity (vorapaxar). Merck & Company Inc, Whitehouse Station, NJ. | ||||
| 49 | Klinkhardt U, Kirchmaier CM, Westrup D, Graff J, Mahnel R, Breddin HK, Harder S "Ex vivo-in vitro interaction between aspirin, clopidogrel, and the glycoprotein IIb/IIIa inhibitors abciximab and SR121566A." Clin Pharmacol Ther 67 (2000): 305-13. [PMID: 10741635] | ||||
| 50 | Bannister SJ, Houser VP, Hulse JD, Kisicki JC, Rasmussen JG "Evaluation of the potential for interactions of paroxetine with diazepam, cimetidine, warfarin, and digoxin." Acta Psychiatr Scand Suppl 350 (1989): 102-6. [PMID: 2530759] | ||||
| 51 | Caraco Y, Sheller J, Wood AJJ "Impact of ethnic origin and quinidine coadministration on codeine's disposition and pharmacodynamic effects." J Pharmacol Exp Ther 290 (1999): 413-22. [PMID: 10381807] | ||||
| 52 | Product Information. Noroxin (norfloxacin). Merck & Co, Inc, West Point, PA. | ||||
| 53 | Product Information. Flolan (epoprostenol). Glaxo Wellcome, Research Triangle Park, NC. | ||||
| 54 | Carrillo JA, Ramos SI, Herraiz AG, Llerena A, Agundez JAG, Berecz R, Duran M, Benitez J "Pharmacokinetic interaction of fluvoxamine and thioridazine in schizophrenic patients." J Clin Psychopharmacol 19 (1999): 494-9. [PMID: 10587283] | ||||
| 55 | Product Information. Abilify (aripiprazole). Bristol-Myers Squibb, Princeton, NJ. | ||||
| 56 | Product Information. Saphris (asenapine). Schering-Plough Corporation, Kenilworth, NJ. | ||||
| 57 | Krahenbuhl S, Sauter B, Kupferschmidt H, Krause M, Wyss PA, Meier PJ "Case report: reversible QT prolongation with torsades de pointes in a patient with pimozide intoxication." Am J Med Sci 309 (1995): 315-6. [PMID: 7771501] | ||||
| 58 | Abebe W "Herbal medication: potential for adverse interactions with analgesic drugs." J Clin Pharm Ther 27 (2002): 391-401. [PMID: 12472978] | ||||
| 59 | Product Information. Agrylin (anagrelide). Roberts Pharmaceutical Corporation, Eatontown, NJ. | ||||
| 60 | Bodiford AB, Kessler FO, Fermo JD, Ragucci KR "Elevated international normalized ratio with the consumption of grapefruit and use of warfarin." SAGE Open Med Case Rep 0 (2013): 1-3. [PMID: 27489634] | ||||
| 61 | Product Information. Acova (argatroban) SmithKline Beecham, Philadelphia, PA. | ||||
| 62 | Collet JP, Hulot JS, Pena A, et al. "Cytochrome P450 2C19 polymorphism in young patients treated with clopidogrel after myocardial infarction: a cohort study." Lancet 373 (2009): 309-17. [PMID: 19108880] | ||||
| 63 | Product Information. Cometriq (cabozantinib). Exelixis Inc, S San Francisco, CA. | ||||
| 64 | Granfors MT, Backman JT, Laitila J, Neuvonen PJ "Tizanidine is mainly metabolized by cytochrome P450 1A2 in vitro." Br J Clin Pharmacol 57 (2004): 349-53. [PMID: 14998432] | ||||
