Details of the Drug
General Information of Drug (ID: DM9OZWQ)
| Drug Name |
Lovastatin
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Synonyms |
lovastatin; 75330-75-5; mevinolin; Mevacor; Lovalip; Monacolin K; Lovalord; Mevinacor; Altoprev; Nergadan; Artein; Altocor; 6alpha-Methylcompactin; MK-803; Hipovastin; Lovastatine; Lovasterol; Paschol; Lipivas; Closterol; Teroltrat; Tecnolip; Rovacor; Cholestra; Rodatin; Lozutin; Lipofren; Lestatin; Hipolip; Colevix; Sivlor; Taucor; Lipdip; Belvas; Mevlor; Lovastatine [French]; Lovastatinum [Latin]; Lovastatina [Spanish]; 6-alpha-Methylcompactin; Lovastatinum; Lovastatina; Lovastin; MSD 803; MK 803; 6 alpha-Methylcompactin; UNII-9LHU78OQFD; Altocor; Liposcler; Mevinolin; Rextat; Sivlor;Taucor; Monakolin K; MK803; Advicor (TN); Altocor (TN); Altoprev (TN); L-154803; Lovastatin & Primycin; ML-530B; Mevacor (TN); Mevinolin from Aspergillus sp; Statosan (TN); Lovastatin (USP/INN); Lovastatin [USAN:BAN:INN]; Lovastatin, (1 alpha(S*))-Isomer; Lovastatin, 1 alpha-Isomer (without R*/S* notation); 2beta,6alpha-Dimethyl-8alpha-(2-methyl-1-oxobutoxy)-mevinic acid lactone; 6 Methylcompactin; 6-Methylcompactin; Aspirin/lisinopril/ lovastatin fixed-dose combination
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Indication |
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Therapeutic Class |
Anticholesteremic Agents
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Affected Organisms |
Humans and other mammals
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ATC Code | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Drug Type |
Small molecular drug
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Structure |
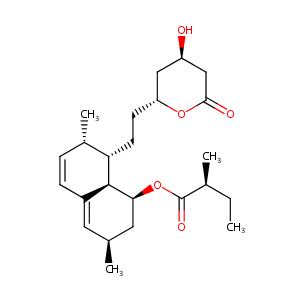 |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 3D MOL | 2D MOL | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| #Ro5 Violations (Lipinski): 0 | Molecular Weight (mw) | 404.5 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Logarithm of the Partition Coefficient (xlogp) | 4.3 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Rotatable Bond Count (rotbonds) | 7 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hydrogen Bond Donor Count (hbonddonor) | 1 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hydrogen Bond Acceptor Count (hbondacc) | 5 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ADMET Property |
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Adverse Drug Reaction (ADR) |
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical Identifiers |
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Cross-matching ID | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Combinatorial Drugs (CBD) | Click to Jump to the Detailed CBD Information of This Drug | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Repurposed Drugs (RPD) | Click to Jump to the Detailed RPD Information of This Drug | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Molecular Interaction Atlas of This Drug
 Drug Therapeutic Target (DTT) |
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
 Drug Transporter (DTP) |
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
 Drug-Metabolizing Enzyme (DME) |
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
 Drug Off-Target (DOT) |
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Molecular Interaction Atlas (MIA) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Molecular Expression Atlas of This Drug
| The Studied Disease | Arteriosclerosis | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ICD Disease Classification | BD40 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Molecular Expression Atlas (MEA) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Drug-Drug Interaction (DDI) Information of This Drug
|
Coadministration of a Drug Treating the Same Disease as Lovastatin
Coadministration of a Drug Treating the Disease Different from Lovastatin (Comorbidity)
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Drug Inactive Ingredient(s) (DIG) and Formulation(s) of This Drug
References
| 1 | Lovastatin FDA Label | ||||
|---|---|---|---|---|---|
| 2 | URL: http://www.guidetopharmacology.org Nucleic Acids Res. 2015 Oct 12. pii: gkv1037. The IUPHAR/BPS Guide to PHARMACOLOGY in 2016: towards curated quantitative interactions between 1300 protein targets and 6000 ligands. (Ligand id: 2739). | ||||
| 3 | Clinical pipeline report, company report or official report of CardioPharma Wilmington. | ||||
| 4 | BDDCS applied to over 900 drugs | ||||
| 5 | Wind S, Schmid U, Freiwald M, Marzin K, Lotz R, Ebner T, Stopfer P, Dallinger C: Clinical Pharmacokinetics and Pharmacodynamics of Nintedanib. Clin Pharmacokinet. 2019 Sep;58(9):1131-1147. doi: 10.1007/s40262-019-00766-0. | ||||
| 6 | Estimating the safe starting dose in phase I clinical trials and no observed effect level based on QSAR modeling of the human maximum recommended daily dose | ||||
| 7 | ADReCS-Target: target profiles for aiding drug safety research and application. Nucleic Acids Res. 2018 Jan 4;46(D1):D911-D917. doi: 10.1093/nar/gkx899. | ||||
| 8 | Microarray and biochemical analysis of lovastatin-induced apoptosis of squamous cell carcinomas. Neoplasia. 2002 Jul-Aug;4(4):337-46. | ||||
| 9 | A novel human hepatic organic anion transporting polypeptide (OATP2). Identification of a liver-specific human organic anion transporting polypeptide and identification of rat and human hydroxymethylglutaryl-CoA reductase inhibitor transporters. J Biol Chem. 1999 Dec 24;274(52):37161-8. | ||||
| 10 | Mammalian drug efflux transporters of the ATP binding cassette (ABC) family in multidrug resistance: A review of the past decade. Cancer Lett. 2016 Jan 1;370(1):153-64. | ||||
| 11 | Metabolic interactions with statins. Tidsskr Nor Laegeforen. 2001 Jan 20;121(2):189-93. | ||||
| 12 | Pharmacogenomics of statins: understanding susceptibility to adverse effects. Pharmgenomics Pers Med. 2016 Oct 3;9:97-106. | ||||
| 13 | Paraoxonases-1, -2 and -3: what are their functions? Chem Biol Interact. 2016 Nov 25;259(Pt B):51-62. | ||||
| 14 | Desmosterol can replace cholesterol in sustaining cell proliferation and regulating the SREBP pathway in a sterol-Delta24-reductase-deficient cell line. Biochem J. 2009 May 13;420(2):305-15. | ||||
| 15 | Simvastatin and lovastatin inhibit breast cell invasion induced by H-Ras. Oncol Rep. 2009 May;21(5):1317-22. doi: 10.3892/or_00000357. | ||||
| 16 | NCI60 cancer cell line panel data and RNAi analysis help identify EAF2 as a modulator of simvastatin and lovastatin response in HCT-116 cells. PLoS One. 2011 Apr 4;6(4):e18306. doi: 10.1371/journal.pone.0018306. | ||||
| 17 | Inhibitory effects of clinical reagents having anti-oxidative activity on transforming growth factor-1-induced expression of -smooth muscle actin in human fetal lung fibroblasts. J Toxicol Sci. 2011;36(6):733-40. doi: 10.2131/jts.36.733. | ||||
| 18 | Protection against the Neurotoxic Effects of -Amyloid Peptide on Cultured Neuronal Cells by Lovastatin Involves Elevated Expression of 7 Nicotinic Acetylcholine Receptors and Activating Phosphorylation of Protein Kinases. Am J Pathol. 2018 Apr;188(4):1081-1093. doi: 10.1016/j.ajpath.2017.11.020. Epub 2018 Jan 16. | ||||
| 19 | Effect of atorvastatin, simvastatin, and lovastatin on the metabolism of cholesterol and triacylglycerides in HepG2 cells. Biochem Pharmacol. 2001 Dec 1;62(11):1545-55. doi: 10.1016/s0006-2952(01)00790-0. | ||||
| 20 | Lovastatin augments apoptosis induced by chemotherapeutic agents in colon cancer cells. Clin Cancer Res. 1999 Aug;5(8):2223-9. | ||||
| 21 | Product Information. Nexletol (bempedoic acid). Esperion Therapeutics, Ann Arbor, MI. | ||||
| 22 | Product Information. Kynamro (mipomersen). Genzyme Corporation, Cambridge, MA. | ||||
| 23 | Canadian Pharmacists Association. | ||||
| 24 | Product Information. Juxtapid (lomitapide). Aegerion Pharmaceuticals Inc, Cambridge, MA. | ||||
| 25 | Cerner Multum, Inc. "Australian Product Information.". | ||||
| 26 | Product Information. Aubagio (teriflunomide). Genzyme Corporation, Cambridge, MA. | ||||
| 27 | Product Information. Tibsovo (ivosidenib). Agios Pharmaceuticals, Cambridge, MA. | ||||
| 28 | Cerner Multum, Inc. "UK Summary of Product Characteristics.". | ||||
| 29 | Multum Information Services, Inc. Expert Review Panel. | ||||
| 30 | Holtzman CW, Wiggins BS, Spinler SA "Role of P-glycoprotein in statin drug interactions." Pharmacotherapy 26 (2006): 1601-7. [PMID: 17064205] | ||||
| 31 | Agbin NE, Brater DC, Hall SD "Interaction of diltiazem with lovastatin and pravastatin." Clin Pharmacol Ther 61 (1997): 201. [PMID: 9797793] | ||||
| 32 | Product Information. Sirturo (bedaquiline). Janssen Pharmaceuticals, Titusville, NJ. | ||||
| 33 | Ayanian JZ, Fuchs CS, Stone RM "Lovastatin and rhabdomyolysis." Ann Intern Med 109 (1988): 682-3. [PMID: 3421582] | ||||
| 34 | Andreou ER, Ledger S "Potential drug interaction between simvastatin and danazol causing rhabdomyolysis." Can J Clin Pharmacol 10 (2003): 172-4. [PMID: 14712320] | ||||
| 35 | Product Information. Ketek (telithromycin). Aventis Pharmaceuticals, Bridgewater, NJ. | ||||
| 36 | Product Information. Balversa (erdafitinib). Janssen Products, LP, Horsham, PA. | ||||
| 37 | Product Information. Turalio (pexidartinib). Daiichi Sankyo, Inc., Parsippany, NJ. | ||||
| 38 | Product Information. Talzenna (talazoparib). Pfizer U.S. Pharmaceuticals Group, New York, NY. | ||||
| 39 | Alderman CP "Possible interaction between nefazodone and pravastatin." Ann Pharmacother 33 (1999): 871. [PMID: 10466919] | ||||
| 40 | He K, Woolf TF, Hollenberg PF "Mechanism-based inactivation of cytochrome P-450-3A4 by mifepristone (RU486)." J Pharmacol Exp Ther 288 (1999): 791-7. [PMID: 9918590] | ||||
| 41 | Product Information. Kalydeco (ivacaftor). Vertex Pharmaceuticals, Cambridge, MA. | ||||
| 42 | Product Information. Prevymis (letermovir). Merck & Company Inc, Whitehouse Station, NJ. | ||||
| 43 | Product Information. Trileptal (oxcarbazepine) Novartis Pharmaceuticals, East Hanover, NJ. | ||||
| 44 | EMEA. European Medicines Agency "EPARs. European Union Public Assessment Reports.". | ||||
| 45 | Akram K, Rao S, Parker M "A lesson for everyone in drug-drug interactions." Int J Cardiol 118 (2007): e19-20. [PMID: 17368833] | ||||
| 46 | Bogman K, Peyer AK, Torok M, Kusters E, Drewe J "HMG-CoA reductase inhibitors and P-glycoprotein modulation." Br J Pharmacol 132 (2001): 1183-92. [PMID: 11250868] | ||||
| 47 | Product Information. Olysio (simeprevir). Janssen Pharmaceuticals, Titusville, NJ. | ||||
| 48 | Product Information. Priftin (rifapentine). Hoechst Marion-Roussel Inc, Kansas City, MO. | ||||
| 49 | Product Information. Accolate (zafirlukast). Zeneca Pharmaceuticals, Wilmington, DE. | ||||
| 50 | Product Information. Rescriptor (delavirdine). Pharmacia and Upjohn, Kalamazoo, MI. | ||||
| 51 | Barry M, Mulcahy F, Merry C, Gibbons S, Back D "Pharmacokinetics and potential interactions amongst antiretroviral agents used to treat patients with HIV infection." Clin Pharmacokinet 36 (1999): 289-304. [PMID: 10320951] | ||||
| 52 | Product Information. Rukobia (fostemsavir). ViiV Healthcare, Research Triangle Park, NC. | ||||
| 53 | Gerber JG, Rosenkranz SL, Fichtenbaum CJ, et al. "Effect of efavirenz on the pharmacokinetics of simvastatin, atorvastatin, and pravastatin: results of AIDS Clinical Trials Group 5108 Study." J Acquir Immune Defic Syndr 39 (2005): 307-12. [PMID: 15980690] | ||||
| 54 | Product Information. Intelence (etravirine). Ortho Biotech Inc, Bridgewater, NJ. | ||||
| 55 | Argov Z, Mastaglia FL "Drug-induced peripheral neuropathies." Br Med J 1 (1979): 663-6. [PMID: 219931] | ||||
| 56 | Product Information. Samsca (tolvaptan). Otsuka American Pharmaceuticals Inc, Rockville, MD. | ||||
| 57 | Product Information. Zurampic (lesinurad). Astra-Zeneca Pharmaceuticals, Wilmington, DE. | ||||
| 58 | Product Information. Alunbrig (brigatinib). Ariad Pharmaceuticals Inc, Cambridge, MA. | ||||
| 59 | Product Information. Lorbrena (lorlatinib). Pfizer U.S. Pharmaceuticals Group, New York, NY. | ||||
| 60 | Al-Nawakil C, Willems L, Mauprivez C, et.al "Successful treatment of l-asparaginase-induced severe acute hepatotoxicity using mitochondrial cofactors." Leuk Lymphoma 55 (2014): 1670-4. [PMID: 24090500] | ||||
| 61 | Product Information. Clolar (clofarabine). sanofi-aventis, Bridgewater, NJ. | ||||
| 62 | Product Information. Braftovi (encorafenib). Array BioPharma Inc., Boulder, CO. | ||||
| 63 | Product Information. Exjade (deferasirox). Novartis Pharmaceuticals, East Hanover, NJ. | ||||
| 64 | Product Information. Xeglyze (abametapir topical). Dr. Reddy's Laboratories Inc, Upper Saddle River, NJ. | ||||
| 65 | Product Information. Xenleta (lefamulin). Nabriva Therapeutics US, Inc., King of Prussia, PA. | ||||
| 66 | Product Information. Zokinvy (lonafarnib). Eiger BioPharmaceuticals, Palo Alto, CA. | ||||
| 67 | Benoist G, van Oort I, et al "Drug-drug interaction potential in men treated with enzalutamide: Mind the gap." Br J Clin Pharmacol 0 (2017): epub. [PMID: 28881501] | ||||
| 68 | Product Information. Tracleer (bosentan). Acetelion Pharmaceuticals US, Inc, South San Francisco, CA. | ||||
| 69 | Product Information. ReVia (naltrexone). DuPont Pharmaceuticals, Wilmington, DE. | ||||
| 70 | Product Information. Tavalisse (fostamatinib). Rigel Pharmaceuticals, South San Francisco, CA. | ||||
| 71 | EMA. European Medicines Agency. European Union "EMA - List of medicines under additional monitoring.". | ||||
| 72 | Barshes NR, Goodpastor SE, Goss JA "Sirolimus-atorvastatin drug interaction in the pancreatic islet transplant recipient." Transplantation 76 (2003): 1649-50. [PMID: 14702546] | ||||
| 73 | Chouhan UM, Chakrabarti S, Millward LJ "Simvastatin interaction with clarithromycin and amiodarone causing myositis." Ann Pharmacother 39 (2005): 1760-1. [PMID: 16159992] | ||||
