Details of the Drug
General Information of Drug (ID: DMA5XGH)
| Drug Name |
Isradipine
|
||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Synonyms |
Clivoten; DynaCirc; DynaCire; Dynacrine; Esradin; Icaz; Isradipin; Isradipino; Isradipinum; Isrodipine; Lomir; Prescal; Rebriden; DynaCire CR; Dynacirc CR; Isradipino [Spanish]; Isradipinum [Latin]; PN 205 033; PN 205 034; PN 205033; PN 205034; PN205033; PN205034; DynaCirc (TN); PN 200-110; PN 205-033; PN 205-034; PN-200110; Prescal (TN); Isradipine (USP/INN); Isradipine [USAN:INN:BAN]; PN-200-110; PN-205-033; PN-205-034; Isradipine, (R)-Isomer; Isradipine, (S)-Isomer; Isradipine, (+-)-Isomer; DynaCirc, Prescal, PN-200-110, Clivoten, Esradin, Isradipine; Isopropyl methyl (+-)-4-(4-benzofurazanyl)-1,4-dihydro-2,6-dimethyl-3,5-pyridinedicarboxylate; Isopropyl 4-(2,1,3-benzoxadiazol-4-yl)-1,4-dihydro-5-methoxycarbonyl-2,6-dimethyl-3-pyridinecarboxylate; Methyl 1-methylethyl 4-(2,1,3-benzoxadiazol-4-yl)-2,6-dimethyl-1,4-dihydropyridine-3,5-dicarboxylate; O5-methyl O3-propan-2-yl 4-(2,1,3-benzoxadiazol-7-yl)-2,6-dimethyl-1,4-dihydropyridine-3,5-dicarboxylate; (+-)-Isradipine; (+/-)-Isradipine; 2,1,3-Benzoxadiazole, 3,5-pyridinedicarboxylic acid deriv.; 3,5-Pyridinedicarboxylic acid, 4-(2,1,3-benzoxadiazol-4-yl)-1,4-dihydro-2,6-dimethyl-, 3-methyl 5-(1-methylethyl) ester; 3,5-Pyridinedicarboxylic acid, 4-(2,1,3-benzoxadiazol-4-yl)-1,4-dihydro-2,6-dimethyl-, methyl 1-methylethyl ester; 3,5-Pyridinedicarboxylic acid, 4-(2,1,3-benzoxadiazol-4-yl)-1,4-dihydro-2,6-dimethyl-, methyl 1-methylethyl ester (9CI); 3,5-Pyridinedicarboxylic acid, 4-(4-benzofurazanyl)-1,4-dihydro-2,6-dimethyl-, methyl 1-methylethyl ester; 3-O-methyl 5-O-propan-2-yl 4-(2,1,3-benzoxadiazol-4-yl)-2,6-dimethyl-1,4-dihydropyridine-3,5-dicarboxylate; 4-(2,1,3-Benzooxadiazol-4-yl)-2.6-dimethyl-1,4-dihydro-3-isopropyloxycarbonylpyridine-5-carboxylic Acid Methyl Ester; 4-(4-Benzofurazanyl)-1,-4-dihydro-2,6-dimethyl-3,5-pyridinedicarboxylic acid methyl 1-methhylethyl ester; 4-(4-Benzofurazanyl)-1,-4-dihydro-2,6-dimethyl-3,5-pyridinedicarboxylic acid methyl 1-methylethyl ester; 4-(4-Benzofurazanyl)-1,4-dihydro-2,-6-dimethyl-3,5-pyridinedicarboxylic acid methyl 1-methylethyl ester
|
||||||||||||||||||||||||||
| Indication |
|
||||||||||||||||||||||||||
| Therapeutic Class |
Antihypertensive Agents
|
||||||||||||||||||||||||||
| Affected Organisms |
Humans and other mammals
|
||||||||||||||||||||||||||
| ATC Code |
|
||||||||||||||||||||||||||
| Drug Type |
Small molecular drug
|
||||||||||||||||||||||||||
| Structure |
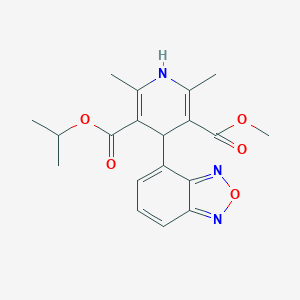 |
||||||||||||||||||||||||||
| 3D MOL | 2D MOL | ||||||||||||||||||||||||||
| #Ro5 Violations (Lipinski): 0 | Molecular Weight (mw) | 371.4 | |||||||||||||||||||||||||
| Logarithm of the Partition Coefficient (xlogp) | 4.3 | ||||||||||||||||||||||||||
| Rotatable Bond Count (rotbonds) | 6 | ||||||||||||||||||||||||||
| Hydrogen Bond Donor Count (hbonddonor) | 1 | ||||||||||||||||||||||||||
| Hydrogen Bond Acceptor Count (hbondacc) | 8 | ||||||||||||||||||||||||||
| ADMET Property |
|
||||||||||||||||||||||||||
| Chemical Identifiers |
|
||||||||||||||||||||||||||
| Cross-matching ID | |||||||||||||||||||||||||||
| Combinatorial Drugs (CBD) | Click to Jump to the Detailed CBD Information of This Drug | ||||||||||||||||||||||||||
| Repurposed Drugs (RPD) | Click to Jump to the Detailed RPD Information of This Drug | ||||||||||||||||||||||||||
Molecular Interaction Atlas of This Drug
 Drug Therapeutic Target (DTT) |
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
 Drug Off-Target (DOT) |
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Molecular Interaction Atlas (MIA) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Drug-Drug Interaction (DDI) Information of This Drug
|
Coadministration of a Drug Treating the Same Disease as Isradipine
Coadministration of a Drug Treating the Disease Different from Isradipine (Comorbidity)
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Drug Inactive Ingredient(s) (DIG) and Formulation(s) of This Drug
References
| 1 | URL: http://www.guidetopharmacology.org Nucleic Acids Res. 2015 Oct 12. pii: gkv1037. The IUPHAR/BPS Guide to PHARMACOLOGY in 2016: towards curated quantitative interactions between 1300 protein targets and 6000 ligands. (Ligand id: 4488). | ||||
|---|---|---|---|---|---|
| 2 | ClinicalTrials.gov (NCT00367211) Study to Evaluate the Incidence of Gastric Ulcers Following Administration of Either PN 200 or Naproxen in Subjects Who Are at Risk for Developing NSAID-Associated Ulcers.. U.S. National Institutes of Health. | ||||
| 3 | BDDCS applied to over 900 drugs | ||||
| 4 | Trend Analysis of a Database of Intravenous Pharmacokinetic Parameters in Humans for 1352 Drug Compounds | ||||
| 5 | Estimating the safe starting dose in phase I clinical trials and no observed effect level based on QSAR modeling of the human maximum recommended daily dose | ||||
| 6 | Drugs@FDA. U.S. Food and Drug Administration. U.S. Department of Health & Human Services. | ||||
| 7 | Optical isomers of dihydropyridine calcium channel blockers display enantiospecific effects on the expression and enzyme activities of human xenobiotics-metabolizing cytochromes P450. Toxicol Lett. 2016 Nov 16;262:173-186. | ||||
| 8 | Early identification of clinically relevant drug interactions with the human bile salt export pump (BSEP/ABCB11). Toxicol Sci. 2013 Dec;136(2):328-43. | ||||
| 9 | Association of CYP1A1 and CYP1B1 inhibition in in vitro assays with drug-induced liver injury. J Toxicol Sci. 2021;46(4):167-176. doi: 10.2131/jts.46.167. | ||||
| 10 | Proliferation of cultured human gingival fibroblasts caused by isradipine, a dihydropyridine-derivative calcium antagonist. Eur J Med Res. 2004 Jun 30;9(6):313-5. | ||||
| 11 | Anastassiades CJ "Nifedipine and beta-blocker drugs." Br Med J 281 (1980): 1251-2. [PMID: 6107167] | ||||
| 12 | Abernethy DR, Egan JM, Dickinson TH, Carrum G "Substrate-selective inhibition by verapamil and diltiazem: differential disposition of antipyrine and theophylline in humans." J Pharmacol Exp Ther 244 (1988): 994-9. [PMID: 3252045] | ||||
| 13 | Product Information. Tibsovo (ivosidenib). Agios Pharmaceuticals, Cambridge, MA. | ||||
| 14 | Product Information. Mycobutin (rifabutin). Pharmacia and Upjohn, Kalamazoo, MI. | ||||
| 15 | Benami M, Giladi Y, Shalev E "The combination of magnesium sulphate and nifedipine - a cause of neuromuscular blockade." Br J Obstet Gynaecol 101 (1994): 262-3. [PMID: 8193107] | ||||
| 16 | Cerner Multum, Inc. "UK Summary of Product Characteristics.". | ||||
| 17 | Hamann SR, Blouin RA, Chang SL, et al "Effects of hemodynamic changes on the elimination kinetics of verapamil and nifedipine." J Pharmacol Exp Ther 231 (1984): 301-5. [PMID: 6491984] | ||||
| 18 | Aronowitz JS, Chakos MH, Safferman AZ, Lieberman JA "Syncope associated with the combination of clozapine and enalapril." J Clin Psychopharmacol 14 (1994): 429-30. [PMID: 7884028] | ||||
| 19 | Heinig R, Adelmann HG, Ahr G "The effect of ketoconazole on the pharmacokinetics, pharmacodynamics and safety of nisoldipine." Eur J Clin Pharmacol 55 (1999): 57-60. [PMID: 10206086] | ||||
| 20 | Product Information. Zylo Filmtab (zileuton). Abbott Pharmaceutical, Abbott Park, IL. | ||||
| 21 | Agencia Espaola de Medicamentos y Productos Sanitarios Healthcare "Centro de informacion online de medicamentos de la AEMPS - CIMA.". | ||||
| 22 | Ban TA "Drug interactions with psychoactive drugs." Dis Nerv Syst 36 (1975): 164-6. [PMID: 1116424] | ||||
| 23 | Product Information. Ketek (telithromycin). Aventis Pharmaceuticals, Bridgewater, NJ. | ||||
| 24 | Product Information. Balversa (erdafitinib). Janssen Products, LP, Horsham, PA. | ||||
| 25 | Product Information. Tykerb (lapatinib). Novartis Pharmaceuticals, East Hanover, NJ. | ||||
| 26 | Product Information. Tukysa (tucatinib). Seattle Genetics Inc, Bothell, WA. | ||||
| 27 | Product Information. Raxar (grepafloxacin). Glaxo Wellcome, Research Triangle Park, NC. | ||||
| 28 | Cerner Multum, Inc. "Australian Product Information.". | ||||
| 29 | Canadian Pharmacists Association. | ||||
| 30 | Product Information. Kalydeco (ivacaftor). Vertex Pharmaceuticals, Cambridge, MA. | ||||
| 31 | Product Information. Prevymis (letermovir). Merck & Company Inc, Whitehouse Station, NJ. | ||||
| 32 | Product Information. Emend (aprepitant). Merck & Company Inc, West Point, PA. | ||||
| 33 | Product Information. Serzone (nefazodone). Bristol-Myers Squibb, Princeton, NJ. | ||||
| 34 | Product Information. Trileptal (oxcarbazepine) Novartis Pharmaceuticals, East Hanover, NJ. | ||||
| 35 | Ahmad S "Nifedipine-phenytoin interaction." J Am Coll Cardiol 3 (1984): 1582. [PMID: 6425385] | ||||
| 36 | Product Information. Xcopri (cenobamate). SK Life Science, Inc., Paramus, NJ. | ||||
| 37 | EMEA. European Medicines Agency "EPARs. European Union Public Assessment Reports.". | ||||
| 38 | Bahls FH, Ozuna J, Ritchie DE "Interactions between calcium channel blockers and the anticonvulsants carbamazepine and phenytoin." Neurology 41 (1991): 740-2. [PMID: 2027492] | ||||
| 39 | Product Information. Tazverik (tazemetostat). Epizyme, Inc, Cambridge, MA. | ||||
| 40 | Henry M, Kay MM, Viccellio P "Cardiogenic shock associated with calcium-channel and beta blockers: reversal with intravenous calcium chloride." Am J Emerg Med 3 (1985): 334-6. [PMID: 2860911] | ||||
| 41 | Product Information. Incivek (telaprevir). Vertex Pharmaceuticals, Cambridge, MA. | ||||
| 42 | Product Information. Olysio (simeprevir). Janssen Pharmaceuticals, Titusville, NJ. | ||||
| 43 | Product Information. Rescriptor (delavirdine). Pharmacia and Upjohn, Kalamazoo, MI. | ||||
| 44 | Product Information. Agenerase (amprenavir). Glaxo Wellcome, Research Triangle Pk, NC. | ||||
| 45 | Product Information. Stribild (cobicistat/elvitegravir/emtricitabine/tenofov). Gilead Sciences, Foster City, CA. | ||||
| 46 | Product Information. Sustiva (efavirenz). DuPont Pharmaceuticals, Wilmington, DE. | ||||
| 47 | Product Information. Fortovase (saquinavir) Roche Laboratories, Nutley, NJ. | ||||
| 48 | Product Information. Intelence (etravirine). Ortho Biotech Inc, Bridgewater, NJ. | ||||
| 49 | Product Information. Prezista (darunavir). Ortho Biotech Inc, Bridgewater, NJ. | ||||
| 50 | Product Information. Reyataz (atazanavir). Bristol-Myers Squibb, Princeton, NJ. | ||||
| 51 | Olkkola KT, Palkama VJ, Neuvonen PJ "Ritonavir's role in reducing fentanyl clearance and prolonging its half-life." Anesthesiology 91 (1999): 681-5. [PMID: 10485779] | ||||
| 52 | Product Information. Vaprisol (conivaptan). Cumberland Pharmaceuticals Inc, Nashville, TN. | ||||
| 53 | Product Information. Zurampic (lesinurad). Astra-Zeneca Pharmaceuticals, Wilmington, DE. | ||||
| 54 | Product Information. Orladeyo (berotralstat). BioCryst Pharmaceuticals Inc, Durham, NC. | ||||
| 55 | Product Information. Lorbrena (lorlatinib). Pfizer U.S. Pharmaceuticals Group, New York, NY. | ||||
| 56 | Product Information. Zydelig (idelalisib). Gilead Sciences, Foster City, CA. | ||||
| 57 | Product Information. Copiktra (duvelisib). Verastem, Inc., Needham, MA. | ||||
| 58 | Product Information. Braftovi (encorafenib). Array BioPharma Inc., Boulder, CO. | ||||
| 59 | Product Information. Exjade (deferasirox). Novartis Pharmaceuticals, East Hanover, NJ. | ||||
| 60 | Product Information. Gilenya (fingolimod). Novartis Pharmaceuticals, East Hanover, NJ. | ||||
| 61 | Product Information. Zeposia (ozanimod). Celgene Corporation, Summit, NJ. | ||||
| 62 | Product Information. Tasigna (nilotinib). Novartis Pharmaceuticals, East Hanover, NJ. | ||||
| 63 | Product Information. Gleevec (imatinib mesylate). Novartis Pharmaceuticals, East Hanover, NJ. | ||||
| 64 | Product Information. Sprycel (dasatinib). Bristol-Myers Squibb, Princeton, NJ. | ||||
| 65 | Product Information. Rozlytrek (entrectinib). Genentech, South San Francisco, CA. | ||||
| 66 | EMA. European Medicines Agency. European Union "EMA - List of medicines under additional monitoring.". | ||||
| 67 | Product Information. Xeglyze (abametapir topical). Dr. Reddy's Laboratories Inc, Upper Saddle River, NJ. | ||||
| 68 | Product Information. Xenleta (lefamulin). Nabriva Therapeutics US, Inc., King of Prussia, PA. | ||||
| 69 | Product Information. Zokinvy (lonafarnib). Eiger BioPharmaceuticals, Palo Alto, CA. | ||||
| 70 | Product Information. Tracleer (bosentan). Acetelion Pharmaceuticals US, Inc, South San Francisco, CA. | ||||
| 71 | Product Information. Prograf (tacrolimus). Fujisawa, Deerfield, IL. | ||||
| 72 | Product Information. Oxbryta (voxelotor). Global Blood Therapeutics, Inc., South San Francisco, CA. | ||||
| 73 | DSouza DL, Levasseur LM, Nezamis J, Robbins DK, Simms L, Koch KM "Effect of alosetron on the pharmacokinetics of alprazolam." J Clin Pharmacol 41 (2001): 452-4. [PMID: 11304902] | ||||
| 74 | Product Information. Tavalisse (fostamatinib). Rigel Pharmaceuticals, South San Francisco, CA. | ||||
| 75 | Product Information. Clozaril (clozapine). Novartis Pharmaceuticals, East Hanover, NJ. | ||||
